VITEEE PCBE Mock Test - 4 - JEE MCQ
30 Questions MCQ Test VITEEE: Subject Wise and Full Length MOCK Tests - VITEEE PCBE Mock Test - 4
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Joining of repeating units to form a macromolecules is called
Use the following characteristics to answer the question: one-way valves thin elastic layer near skeletal muscle These characteristics describe which type of vessel?
Cristae are associated with which of the following organelle?
RNA that picks up specfic amino acid from amino acid pool of cytoplasm to carry it to ribosome during protein synthesis is
Eschirichia coli with completely radioactive DNA was allowed to replicate is non-radioactive medium for two generations. Percentage of bacteria with radioactive DNA is
Had Mendal decided to study those traits together which are determined by linked genes, he would not have found out
In sickle cell anaemia, death occurs when lethal genes are present in
If an albino marries a normal woman and 50% offspring are albino, the woman is
Directions: In this type of questions, some particular words are assigned certain substituted names. Then a question is asked that is to be answered in the substituted code language.
If 'sand' is called 'air', 'air' is called 'plateau', 'plateau' ia called 'well', 'well' is called 'island' and 'island' is called 'sky', then from where will a woman draw water?
Directions: In this type of questions, some particular words are assigned certain substituted names. Then a question is asked that is to be answered in the substituted code language.
If 'cushion' is called 'pillow', 'pillow' is called 'mat', 'mat' is called 'bedsheet' and 'bedsheet' is called 'cover', which will be spread on the floor ?
Directions: In this type of questions, some particular words are assigned certain substituted names. Then a question is asked that is to be answered in the substituted code language.
If 'rain' is 'water', 'water' is 'road', 'road' is 'cloud', 'cloud' is 'sky', 'sky' is 'sea' and 'sea' is 'path', where do aero planes fly ?
Directions: Study the following graph carefully to answer the questions that follow.
Number of Students (In thousands) in Two Different Universities in Six Different Years

Q. What is the sum of the number of students in university 1 in the year 2007 and the number of students in university 2 in the year 2011 together?
Directions: In the question below, the sales of TV and laptop in the subsequent years from 2004 to 2009 is given. The figures given to you is in million. Based on the figure answer the questions below.

Q. How many times do the sales of a laptop less than the TV in the given years?
Directions: In the question below, the sales of TV and laptop in the subsequent years from 2004 to 2009 is given. The figures given to you is in million. Based on the figure answer the questions below.

Q. Which item and for which year shows the highest percentage increase in the sales in the previous year?
Directions: In the question below, the sales of TV and laptop in the subsequent years from 2004 to 2009 is given. The figures given to you is in million. Based on the figure answer the questions below.

Q. Find the ratio of the total number of televisions sold to the total number of laptops sold in all these years.
Directions: Study the following graph carefully and answer the question given below.
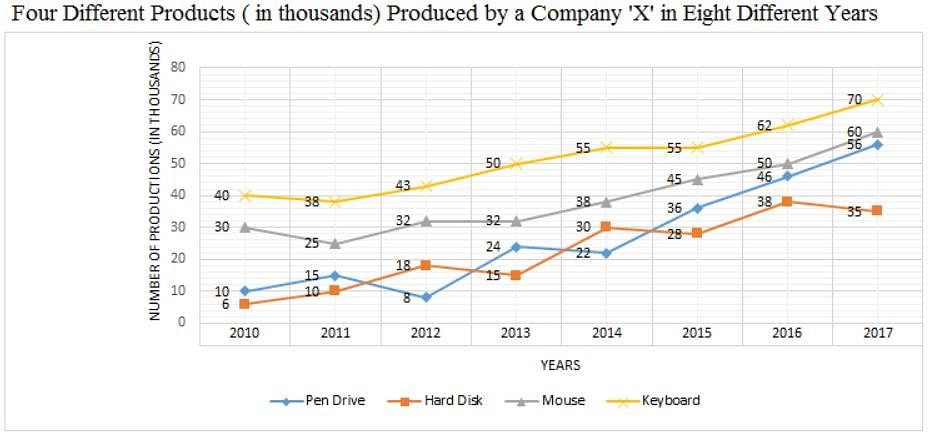
Q. What is the difference between the total number of hard disks and mouses produced by Company 'X' in the year 2010 and the total number of keyboards and pen drives produced by Company 'X' in the year 2015?
Directions: Study the following graph carefully and answer the question given below.
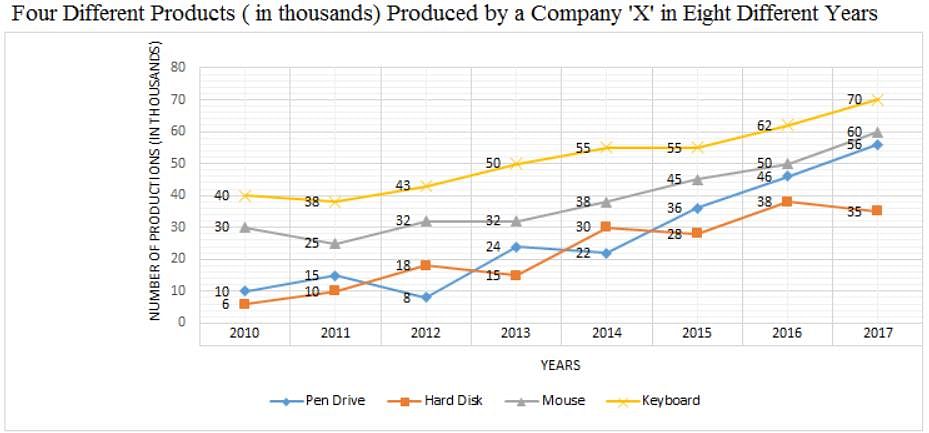
Q. What was the respective ratio between the number of pen drives produced by Company 'X' in the years 2010, 2011, 2014 and 2015 and the number of hard disks produced by Company 'X' in the years 2011, 2012, 2015 and 2017?
290 students of MBA (International Business) in a reputed Business School have to study foreign language in Trimesters IV and V. Suppose the following information are given
(i) 120 students study Spanish
(ii) 100 students study Mandarin
(iii) At least 80 students, who study a foreign language, study neither Spanish nor Mandarin
Then the number of students who study Spanish but not Mandarin could be any number from
|
1 videos|2 docs|73 tests
|
|
1 videos|2 docs|73 tests
|

















