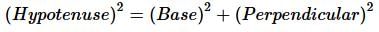VITEEE PCME Mock Test - 4 - JEE MCQ
30 Questions MCQ Test - VITEEE PCME Mock Test - 4
A six-faced unbiased die is thrown twice and the sum of the numbers appearing on the upper face is observed to be 7. The probability that the number 3 has appeared at least once, is
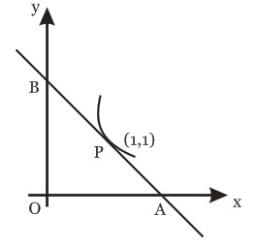
The triangle formed by the tangent to the curve f(x) = x² + bx - b at the point (1, 1) and the coordinate axes lies in the first quadrant. If its area is 2, then the value of the y-intercept is:
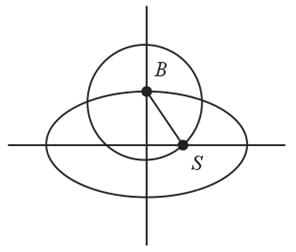
Equation of a circle with its center at the extremity of the positive minor axis and passing through the focus (on the positive x-axis) of the ellipse x²/16 + y²/9 = 1is:
If the foci of the ellipse 16x² + 7y² = 112 and the hyperbola y²/144 - x²/a² = 1/25 coincide, then a =
A bar magnet of magnetic moment 104 J/T is free to rotate in a horizontal plane. The work done in rotating the magnet slowly from a direction parallel to a horizontal magnetic field of 4 x 10-5 T to a direction of 60° from the field will be
As shown in the figure, a magnet is moved with a fast speed towards a coil at rest. Due to this, induced emf, induced current and induced charge in the coil are F, I and Q, respectively. If the speed of the magnet is doubled, the incorrect statement is
If μe and μh are electron and hole mobilities and E is the applied electric field, then the current density for intrinsic semiconductor is equal to
The de-Broglie wavelength of electron falling on the target in an X-ray tube is λ. The cut-off wavelength of the emitted X-ray is
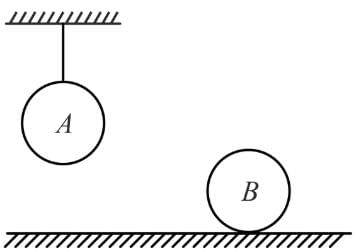
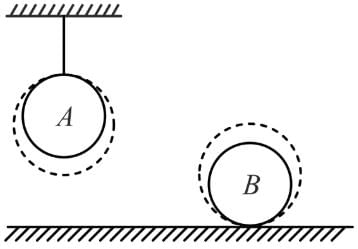
Consider two identical homogeneous balls, A and B, with same initial temperatures. One of them is at rest on fixed horizontal surface, while the second one hangs on a thread as shown. Now the same amount of heat has been supplied to both the balls. We assume that such quantities as specific heat and coefficient of thermal expansion are constant (i.e., do not depend on temperature) and positive. Also we neglect expansion in the length of string and take all kinds of heat loss from balls to be negligible. Considering loss in gravitational potential energy is converted into heat energy and vice versa, the final temperature of the balls, TA and TB will obey the relation.
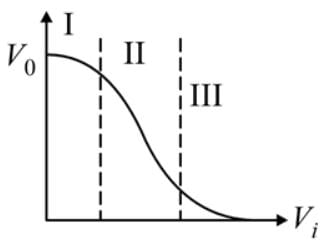
Transfer characteristics [output voltage (V0) vs input voltage (Vi) for a base biased transistor in CE configuration is as shown in the figure. For using transistor as a switch, it is used
The induced emf of a generator when the flux of poles is doubled and speed is doubled then it,
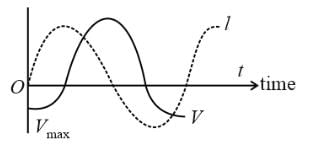
The graphs of AC voltage versus current with time are as shown. The circuit contains
The equation of a sound wave at 0°C is given as y = A sin(1000t - 3x). The speed at some other temperature T is given 336 m/s. The value of T is
A choke is preferred to a resistance for limiting current in AC circuit because
The following mechanism has been proposed for the reaction of NO with Br2 to form NOBr.
NO (g) + Br2 (g)  NOBr2 (g) ..........(fast)
NOBr2 (g) ..........(fast)
NOBr2 (g) + NO (g) → 2NOBr (g) ...........(slow)
The overall order of the reaction is
Using the data provided, calculate the bond energy (kJ mol⁻¹) of a C≡C bond in ethyne. Take the bond energy of a C−H bond as 350 kJ mol⁻¹.
Given data:
- 2C(s) → 2C(g) ; ΔH = 1410 kJ mol⁻¹
- H₂(g) → 2H(g) ; ΔH = 330 kJ mol⁻¹
- Enthalpy of formation of C₂H₂(g) = 225 kJ mol⁻¹
Length of the unit cell is 654 pm, and the density of KBr is 2.75 g cm⁻³. The molecular masses of K and Br are 39 and 80, respectively.
Find out what can be true for the predicted nature of the solid:
What is the entropy change (in J/K·mol) when 1 mole of ice is converted into water at 0°C? (The enthalpy change for the conversion of ice to liquid water is 6.0 kJ/mol at 0°C.)
The number of geometrical isomers of CH3CH = CH − CH = CH − CH = CHCl is
Identify the sentences fom the given options which can be connected to fom a single sentence using 'USUALLY'.
USUALLY
A. The fête was held for the first time on a Saturday.
B. Fleas are a fairly common problem.
C. It is held on a Friday evening.
D. They are brought into the house.
If the hands of a clock coincide every 66 minutes (true time), then how much does it gain or lose every hour?
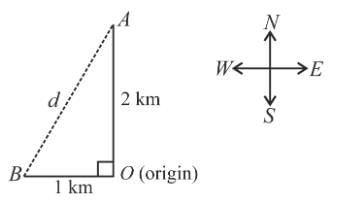
Toy machines A and B walk at an average speed of 0.5 km/hr and 0.25 km/hr, respectively. If toy A walks in the north direction and toy B in the west direction beginning from the same origin at the same time, what will be the distance between two after 4 hours?


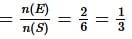
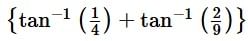 is equal to
is equal to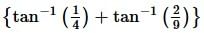
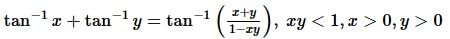
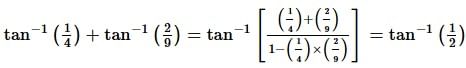

 . Then, f(x)fx is
. Then, f(x)fx is
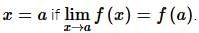
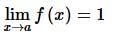
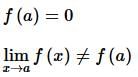
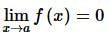
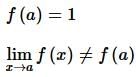
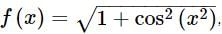 then the value of
then the value of 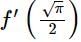 is:
is: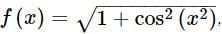 ...........(i)
...........(i)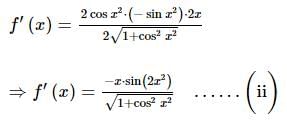
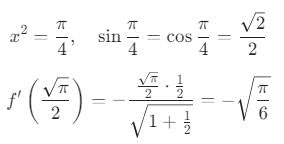
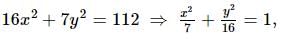
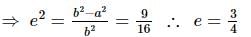
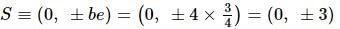
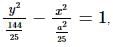
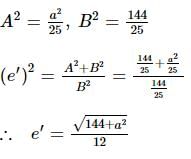
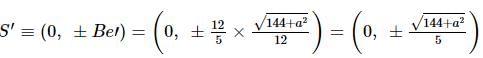
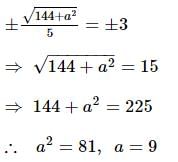



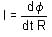

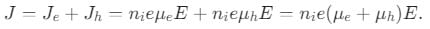
 …(i)
…(i) …(ii)
…(ii)

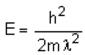 …(iii)
…(iii)



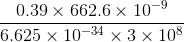
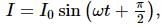
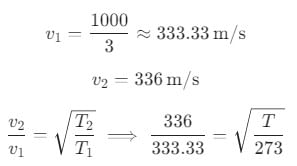


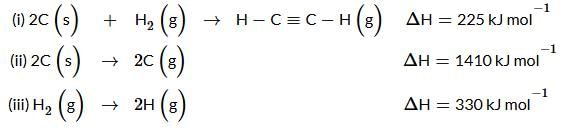
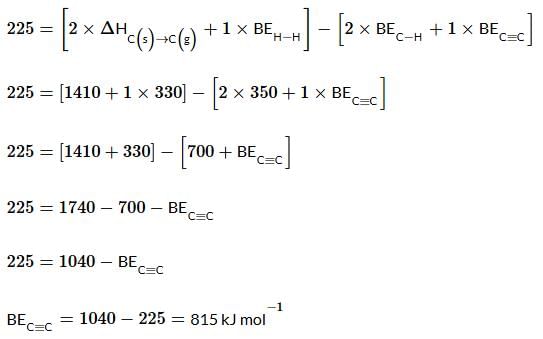
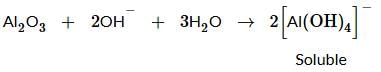
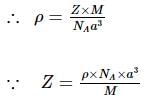
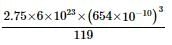
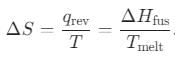
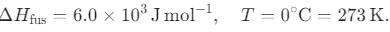
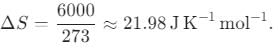
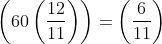
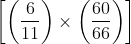 min = 60/121 min
min = 60/121 min