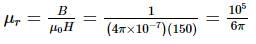AIIMS Full Mock Test 2 - NEET MCQ
30 Questions MCQ Test AIIMS Mock Tests & Previous Year Question Papers - AIIMS Full Mock Test 2
Large transformers, when used for some time, become hot and are cooled by circulating oil. The heating of transformer is due to
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
A capacitor of capacity C₁ charged to V volt is connected to an uncharged capacitor of capacity C₂. The final potential difference across each will be
A 5 Kg body moving at 10 ms-1 strikes a 20Kg body at rest. The collision is inelastic and the 5Kg body comes to rest and the 20Kg body is set into motion. The velocity of the second body is
If a wire of resistance R is melted and recasted to half of its length, then the new resistance of the wire will be
If the no. of turns per unit length of the coil of a solenoid is doubled kepping other dimensions same, then its self-inductance will be
What is the de Broglie wavelength of the α-particle accelerated through a potential difference V
If the kinetic energy of an electron doubles, its de-Broglie's wavelength changes by a factor
A photocell stops emission if it is maintained at 2V negative potential. The energy of most energetic photoelectron is
Two coils are placed close to each other. The mutual inductance of the pair of coils depends upon
What is the value of inductance L for which the current is a maximum in series LCR circuit with
C = 10μF and ω = 1000s−1 ?
When light travels from air to water, which parameter does not change?
The capacities of three capacitors are in ratio 1 : 2 : 3. Their equivalent capacity when connected in parallel is more than that when they are connected in series. The individual capacities of capacitors in μF are
The capacitance of an isolated spherical conductor of radius 15 cm is
Two masses of 1 gm and 4 gm are moving with equal kinetic energies. The ratio of the magnitudes of their linear momentum is
If the stone is thrown up vertically and return to ground, its potential energy is maximum
Consider a compound slab consisting of two different materials having equal thickness and thermal conductivities K and 2K respectively. The equivalent thermal conductivity of the slab is
An electric kettle has two heating elements. One brings it to boil in ten minutes and the other in fifteen minutes. If two heating filament are connected in parallel, the water in kettle will boil in
In a deflection magnetometer experiment in tan A position, a short-bar magnet placed at 18 cm from the centre of the compass needle produces a deflection of 30º. If another magnet of same length but 16 times pole strength as that of first magnet is placed in tan B position at 36cm, the deflection will be
The magnetic induction and the intensity of magnetic field inside an iron core of an electromagnet are 1 wb m-2 and 150 Am-1 respectively. The relative permeability of iron is (μ0 = 4π x 10-7 Henry / m)
An elastic material with Young's modulus 'Y' is subjected to a tensile stress 'σ'. The elastic potential energy stored per unit volume of the material is
The resultant of two forces 3P and 2P is R. If the first force is doubled then the resultant is also doubled. The angle between the two forces is
The radioisotope, tritium (₁H3) has a life-life of 12.3 years. If the initial amount of tritium is 32 mg, how many milligrams of it would remain after 49.2 years?
A spring is stretched by 10 cm when a mass of 1 kg is hanged from it. Now, with the same mass if the spring is pulled and then released, then its time period is 9g = 10 m/s2)
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): Specific gravity of a fluid is a dimensionless quantity.
Reason(R): It is the ratio of density of fluid to the density of water.
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): Sum of the mass moment is zero, if centre of mass is at origin.
Reason(R): Position of centre of mass is given by
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): A wire of length L metre, carrying a current 1 ampere is bent in the form of a circle. Its magnitude of magnetic moment will be 4πIL2.
Reason(R): Magnetic moment = Area of coil X Current .
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): A 4Ω resistance wire is bent in the middle by 180º and both the halves are twisted with each other. The new resistance is 1Ω.
Reason(R): On twisting the parts they will be connected in parallel.
|
3 videos|44 docs|66 tests
|
|
3 videos|44 docs|66 tests
|