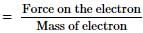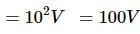AIIMS Full Mock Test - 6 - NEET MCQ
30 Questions MCQ Test AIIMS Mock Tests & Previous Year Question Papers - AIIMS Full Mock Test - 6
A loss free transformer has 500 turns on its primary winding and 2500 in secondary. The meters of the secondary indicate 200 volts at 8 amperes under these conditions. The voltage and current in the primary is
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Two non-ideal batteries are connected in parallel. Consider the following statements:
(i) The equivalent emf is smaller than either of the two emfs
(ii) The equivalent internal resistance is smaller than either of the two internal resistances
A gun fires a bullet of mass 50 gm with a velocity of 30 m sec⁻1 Because of this gun is pushed back with a velocity of 1 m sec⁻1. The mass of the gun is
An electron having charge e and mass m is moving in a uniform electric field E. Its acceleration will be
If particles are moving with same velocity, then maximum de-Broglie wavelength will be for
If a radiation of energy E falls normally on a perfectly reflecting surface, then the momentum transferred to the surface is
Particle nature and wave nature of electromagnetic waves and electrons be shown by
The primary and secondary coils of a transformer have 50 and 1500 turns respectively. If the magnetic flux φ linked with the primary coil is given by φ = φ0 + 4t, where φ is in weber, t is time in second and φ0 is a constant, the output voltage across the secondary coil is
In a pure inductive circuit with a.c. source, the current lags behind e.m.f. by
In a car spark coil, an e.m.f. of 40000 V is induced in a secondary when the primary current changes from 4 A to zero in 10μs. Find the mutual inductance between the primary and secondary windings of the spark coil
In an oscillating LC circuit the maximum charge on the capacitor is Q. The charge on the capacitor when the energy is stored equally between the electric and magnetic fields is
An electromagnetic wave of frequency 3 MHz passes from vacuum into a medium with dielectric constant k = 4. Then
If 1000 droplets each of charge q and radius r are mixed to form a big drop, then the potential of big drop, as compared to small droplet will be
A capacitor of capacitance 1 μF is charged to 1KV. The energy stored in the capacitor in joules is
If the unit of force and length be each increased by four times, then the unit of energy is increased by
Two bodies of different masses m₁ and m₂ have equal momenta. Their kinetic energies E₁ and E₂ are in the ratio
A solid copper cube of edges 1 cm is suspended in an evacuated enclosure. Its temperature is found to fall from 100ºC to 99ºC in 100 s. Another solid copper cube of edges 2 cm, with similar surface nature, is suspended in a similar manner. The time required for this cube to cool from 100ºC to 99ºC will be approximately
An electric iron which when hot has a resistance of 80Ω, is used on a 200 V source. Calculate the electric energy spent in kilowatt-hour if it is used for 2 hours.
When 2 ampere current is passed through a tangent galvanometer, it gives a deflection of 30º. For 60º deflection, the current must be
An elastic material with Young's modulus 'Y' is subjected to a tensile stress 'σ'. The elastic potential energy stored per unit volume of the material is
Three identical bar magnets are placed in the form of an equilateral triangle with north pole of one touching the south pole of the other as shown in fig. The net magnetic moment of the system is
A force of 100N acts on a body of mass 2kg for 10s. The change in momentum of the body is
A radioactive isotope decays at such a rate that after 192 minutes, only 1/16 of the original amount remains. The half-life of the radioactive isotope is
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): A lens of water formed by very thin convex glass layer has lesser focal length than a double convex lens of glass.
Reason(R): Refractive index of glass is more than that of water.
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): The path of γ-rays in Wilson's chamber are very thin continuous lines.
Reason(R): γ-rays produce very little ionisation.
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): Neutrons can be accelerated by cyclotron.
Reason(R): Neutrons are too heavy.
In the following question, a Statement of Assertion (A) is given followed by a corresponding Reason (R) just below it. Read the Statements carefully and mark the correct answer-
Assertion(A): When temperature of gas remains constant, speed of sound changes.
Reason(R) : 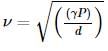 (Symbols have their usual meaning)
(Symbols have their usual meaning)
|
3 videos|44 docs|66 tests
|
|
3 videos|44 docs|66 tests
|