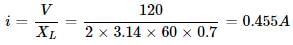SRMJEE Mock Test (Med) - 2 - JEE MCQ
30 Questions MCQ Test SRMJEEE Subject Wise & Full Length Mock Tests 2024 - SRMJEE Mock Test (Med) - 2
A 0.7 Henry inductor is connected across a 120V - 60Hz A.C. source. The current in the inductor will be very nearly
The third line of Balmer series of an ion equivlaent to hydrogen atom has wavelength of 108.5 nm. The ground state energy of an electron of this ion will be
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
In purely inductive circuit, the relation of phase between current and voltage is
A particle is moving on a circular path with constant speed, then its acceleration will be
The drift velocity of free electrons in a conductor is 'υ' when a current 'ι' is flowing in it. If both the radius and current are doubled, then drift velocity will be
A mass m falls freely from rest. The linear momentum after it has fallen through a height h is
When ultraviolet rays are incident on a metal plate, then the photoelectric effect does not occurs. It occurs by the incidence of
Which of the following are essential for maintaining oscillations in an oscillator?
1. Positive feedback
2. Design of load (turned or phase shifting network
3. Non-linear biasing circuit
4. high gain amplifier
Select the correct answer using the codes given below:
Codes:
There is an electric field E in x-direction. If the work done on moving a charge of 0.2C through a distance of 2m, along a line making an angle 60o with x-axis is 4 J, then what is the value of E?
If the coefficient of friction is √3, the angle of friction is
If g is the acceleration due to gravity at the earth's surface and r is the radius of the earth, the escape velocity for the body to escape out of earth's gravitational field is
At a given temperature, the pressure of an ideal gas of density ρ is proportional to
A body is imparted motion from rest to move in a straight line. If it is then obstructed by an opposing force, then
Which of the following relations represent Biot-Savart's law?
The susceptibility of magnetic substance for different purposes is determined by its
If the radius of a soap bubble is four times that of another, then the ratio of their pressures will be
A body falls freely from rest. It covers as much distance in the last second of its motion as covered in the first three seconds. The body has fallen for a time of
A car is moving in a circular horizontal track of radius 10 m with a constant speed of 10 m−s-1. A plumb bob is suspended from the roof of the car by a light rod of length 1m. The angle made by the rod with the track is (Take g=10 m−s-1)
Two forces are such that the sum of their magnitudes is 18 N and their resultant is perpendicular to the smaller force and magnitude of resultant is 12. Then the magnitudes of the forces are
A particle execute simple harmonic motion with a frequency f. The frequency with which its kinetic energy oscillates is
The maximum range of a gun on horizontal terrain is 16 km. If g = 10 m/s2. What must be the muzzle velocity of the shell
According to kinetic theory of gases the absolute temperature of a gas is directly related to the average of
With respect to air, critical angle in a medium for light of red colour [λ₁] is θ. Other facts remaining same, critical angle for light of yellow colour [λ₂] will be
Of two eggs which have identical sizes, shapes and weights, one is raw and the other is half-boiled. The ratio between the moment of inertia of the raw egg and that of the half-boiled egg about a central axis is
The dimensional formula for universal gravitational constant is
When two capillary tubes of different diameters are dipped vertically, the rise of the liquid is
The ratio of diameters of two similar copper wires of qual length is 1:2.A constant current is passed through them when connected in series. The ratio of heat produced in the two will be
Two marks on a glass rod 10cm apart are found to increase their distance by 0.08mm when the rod is heated from 0oC to 100oC. A flask made of the same glass as that of rod measures a volume 1000c.c at 0 oC. The volume it measures at 100 oC in c.c is
|
1 videos|1 docs|68 tests
|
|
1 videos|1 docs|68 tests
|