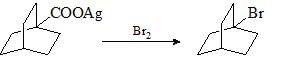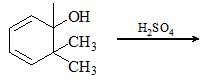GOC - Stereochemistry - Reaction Mechanism - Chemistry MCQ
30 Questions MCQ Test Mock Test Series for IIT JAM Chemistry - GOC - Stereochemistry - Reaction Mechanism
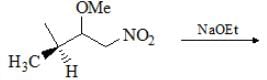
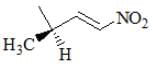
The reaction given below is an example of:


| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
The hyperconjugative stabilities of tert-butyl cation and 2-butene, respectively, are due to?
Amongst the following, the compound which has the lowest energy barrier for the cis-trans isomerization is?
The order of basicity among the following compounds is?
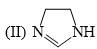
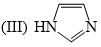
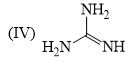
Which of the following is the most stable conformer of the following molecule?
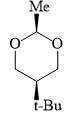
In the given chemical reaction, major product will be?
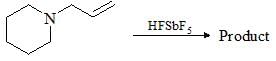
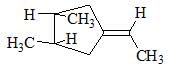
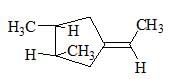
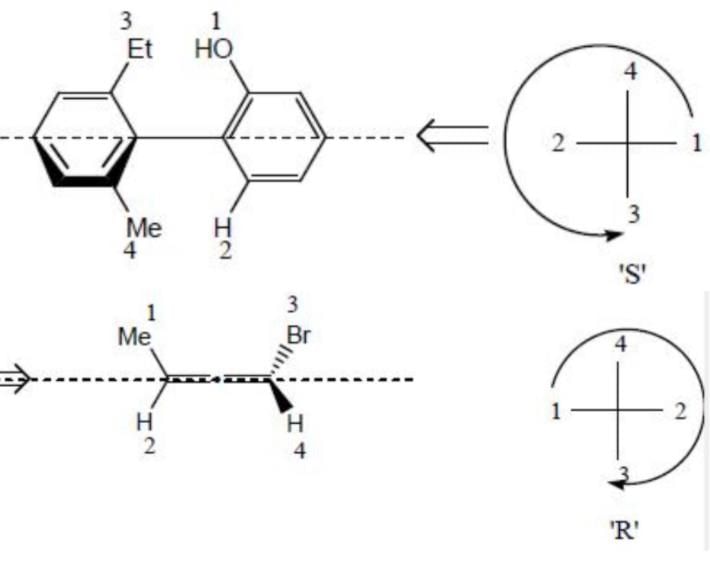
The correct relation between the following compounds is:
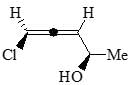
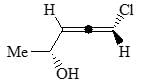
The major product formed in the following reaction is:
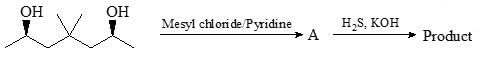
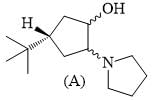
Among the choices, the correct statements for A formed in the following reaction.

The gauche interaction values for Me/Me, Me/Br, and Br/Br are 3.3, 0.8 and 3.0 kJ/mol, respectively. Among the following, the most stable conformation of 2, 3-dibromobutane is:
For each of the following pairs of compounds, which is having second compound more stable than first compound:
Choose the correct product of the following reaction:
Which of the following groups has the highest priority according to the Cahn-Ingold-Prelog sequence rules?
Lindane (hexachlorocyclohexane) is an agricultural insecticide that can also be used in the treatment of head lice.
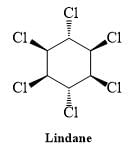
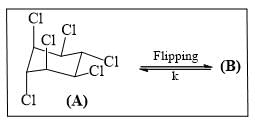
Draw the other chair conformation (B) of Lindane and inspect which of the following statements are correct.
The increasing order of pKa values of the circled hydrogen in the following compounds is?

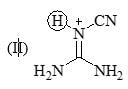
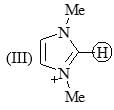

1, 2, 3, 4, 5, 6-hexamethylcyclohexane has 8 cis/trans stereoisomer. Which stereoisomer would be least likely to undergo conformation flipping?
The major product formed in the following reaction is:
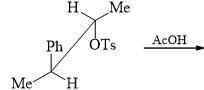
The major product of the following reaction is:
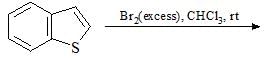
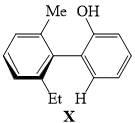
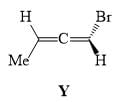
The absolute configuration for compounds X and Y respectively are:
The major product X and Y of the following reaction sequence are:

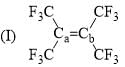
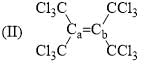
Arrange the following compounds in order of Ca—Cb bond length:
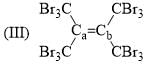
Among the following, the Newman projections of meso-2, 3-butanediol are:
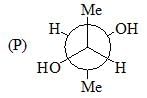
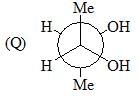
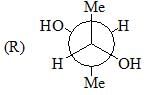
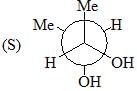
Among A-C, the compounds which can exhibit optical activity are:
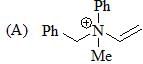
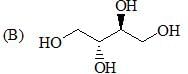
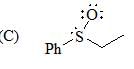
The correct order of acidity values of circled protons (in the following compound) will be?
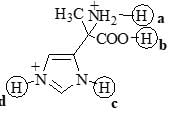
Choose the correct product of the following reaction:
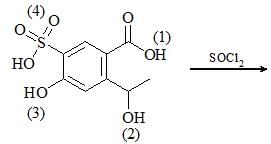
In the given chemical reaction, SOCl2 will react at:
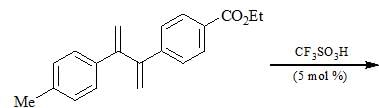
For the given reaction,
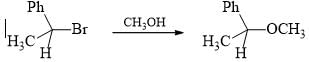
What should be the percentage of backside attack assuming complete reaction?
|
2 docs|25 tests
|