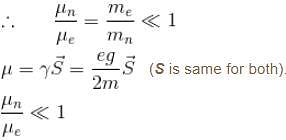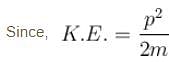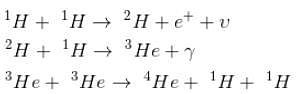Nuclear Physics MCQ Level – 2 - Physics MCQ
10 Questions MCQ Test Topic wise Tests for IIT JAM Physics - Nuclear Physics MCQ Level – 2
Suppose that 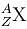 decays by natural radioactivity in two stages to
decays by natural radioactivity in two stages to 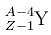 the two stages would most likely be
the two stages would most likely be
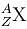 decays by natural radioactivity in two stages to
decays by natural radioactivity in two stages to 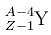 the two stages would most likely be
the two stages would most likely be| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Why do heavier nuclei have a greater ratio of neutrons to protons than lighter nuclei?
Which of the following nuclei has the largest Binding Energy/nucleon?
(Consider the most abundant isotope of each)
The 238U nucleus has a binding energy of about 7.6 MeV/nucleon. If the nucleus were to fission into 2 equal fragments, each would have a K.E. of just over 100 MeV. From this, it can be concluded that
Consider a heavy nucleus with spin 1/2 the magnitude of the ratio of the intrinsic magnetic moment of this nucleus to that of an electron is
A Uranium nucleus decays at rest into a Thorium nucleus, as shown.
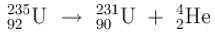
Which of the following is true?
The primary source of sun’s energy is a series of thermo nuclear reactions in which the energy produced is c2 times the mass difference between
Internal conversion is a process where by an excited nucleus transfers its energy directly to one of the most tightly bound atomic electrons, causing the electron to be ejected from the atom and leaving the atom in an excited state. The most probable process after an internal conversion electron is ejected from an atom with a high atomic number is that the


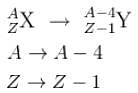
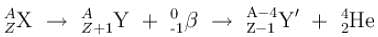
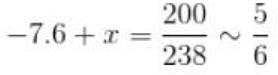 –7.6 + x = 0.83
–7.6 + x = 0.83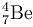 transforms into
transforms into 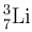 does so by
does so by