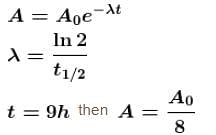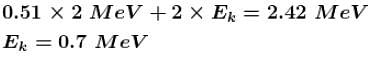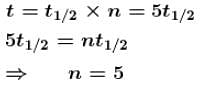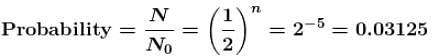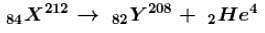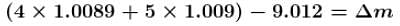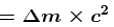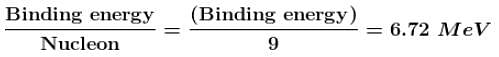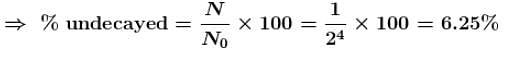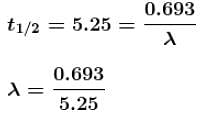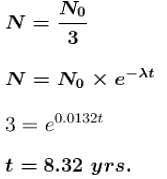Nuclear Physics Nat Level – 1 - Physics MCQ
10 Questions MCQ Test Topic wise Tests for IIT JAM Physics - Nuclear Physics Nat Level – 1
The half life of a radioactive substance is 3h and its activity is 1µCi. Then the activity after 9h will be (in µCi)
An electron positron pair (rest mass of each particle = 0.51 MeV) is produced by a γ -ray photon of energy 2.42 MeV. The K.E. (in MeV) imparted to each of the charged particles shall be :
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
The probability of a radioactive atoms to survive 5 times longer than its half value period is.
A radioactive nuclie decay as follows 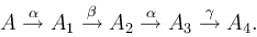 If mass number and atomic number of A are 180 and 72 respectively then atomic number for A4 will be.
If mass number and atomic number of A are 180 and 72 respectively then atomic number for A4 will be.
After emission of an α -particle by a radioactive element 84X212, the resulting mass number would be.
If the mass of proton = 1.008 amu and mass of neutron = 1.009 amu then binding energy (in MeV) per nucleon for 4Be9 (mass = 9.012 amu) would be :
After a time equal to four half lives, the percentage of radioactive material remaining undecayed is :
The half-life of cobalt 60 is 5.25 years. How long after a new sample is delivered, will the activity have decreased to about one third (1/3) of its original value. (Provide the time in years).
The mean life of a radioactive sample is 100 years. Then after 100 years, what percentage of the sample remains active.
1014 fissions per second are taking place in a nuclear reactor having efficiency 40%. The energy released per fission is 250 MeV. The power output (in Watts) of the reactor is :