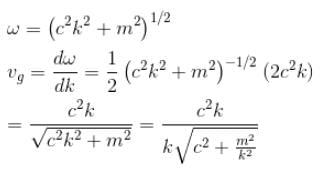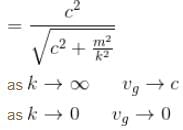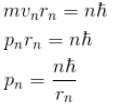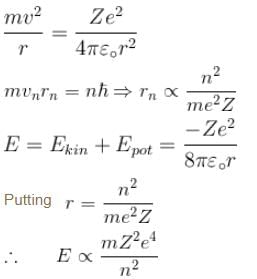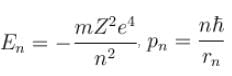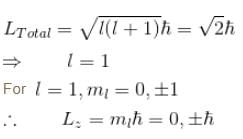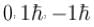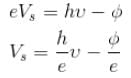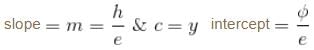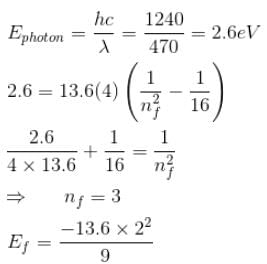Origin Of Quantum Mechanics (MSQ) - Physics MCQ
10 Questions MCQ Test Topic wise Tests for IIT JAM Physics - Origin Of Quantum Mechanics (MSQ)
The dispersion law for a certain type of wave motion is ω = (c2k2 + m2)1/2 where ω is the angular frequency, k is the magnitude of the propagation vector and c, m are constants. The group velocity of these waves approaches :
In the photoelectric effect experiment we use a source of light whose frequency can be varied. If we gradually increase the frequency of the incident light then :
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
In case of an one electron Bohr atom, pn is the linear momentum of the electron in the nth orbit and En is its total energy. Which of the following is true? Here rn is the radius of the nth orbit and m is the mass.
For single electron atom with orbital angular momentum 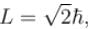 which of the following can be the possible values of a measurement of Lz. (z component of L)?
which of the following can be the possible values of a measurement of Lz. (z component of L)?
For the photoelectric effect, stopping potential v/s incident frequency is plotted. Choose the correct statements.
The kinetic energy of electrons ejected from the surface of a metal ranges from :
Which of the following is true about the absorption and emission of energy by an atom?
A singly ionized Helium atom in n = 4 state emits a photon of wavelength 470nm. If Ef is the final state energy and nf is the principal quantum number of the final state then :