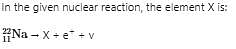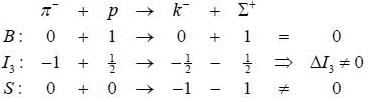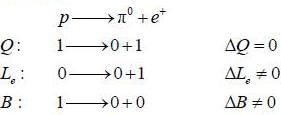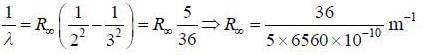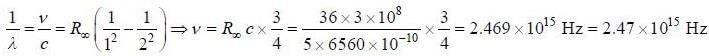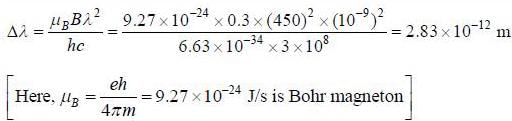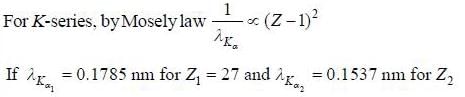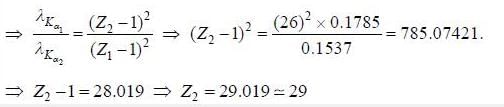Test: Combined (Atomic + Nuclear + Solid) - Physics MCQ
30 Questions MCQ Test GATE Physics Mock Test Series 2025 - Test: Combined (Atomic + Nuclear + Solid)
For a 3-D free electron gas, the electron density n and the Fermi energy EF, are related as
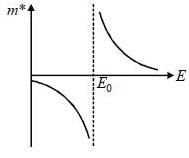
The tight binding energy dispersion relation for electrons in 1-D array of a toms having lattice constant a and total length l is E = E0 + γ cosh (ka) . where E0, γ are constants and k is wave vector. Which graph best represents the variation of effective mass with energy.
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
The magnetization (M) of some hypothetical substance in magnetic field (H) is given by
M (H) = C In2(H + H0), H > l - H0
where H0 and C are positive and negative constants respectively. In what category should tliis substance be put
M (H) = C In2(H + H0), H > l - H0
where H0 and C are positive and negative constants respectively. In what category should tliis substance be put
The temperature at which the probability that an energy state with an energy 0.2 e V above the fenni level will be occupied by an electron is 10 %
The dispersion relation of phonons in a solid is given by
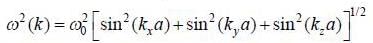
Tlie velocity of phonons at large wavelength is related to wave number k as
Consider a crystal with base-centered-cubic lattice with positions of basis atoms at (0, 0, 0) and 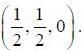
The lattice plane for which there is no diffraction peak is
In nearly free election model, the wavefimctions at the Biillonin zone boimclaiy are 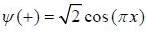 and
and 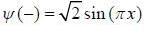 , normalized over unit length of line. If potential energy ofan electron in the crystal at point x is V (x) = V0 [1 - cos (2πx)] . the first order energy gap between two standing waves is
, normalized over unit length of line. If potential energy ofan electron in the crystal at point x is V (x) = V0 [1 - cos (2πx)] . the first order energy gap between two standing waves is
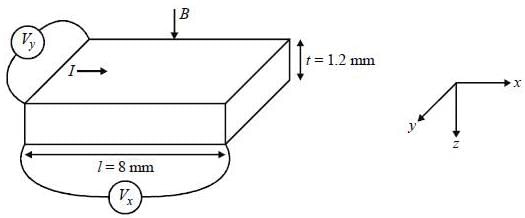
Ap-doped semiconductor slab carries a current I = 150 mA in a magnetic field 0.3 T as shown. Voltage differences Vx and Vy are 2.5 mV and 0.3 mV, respectively. The mobility of majority charge earners is 0.8 m2 V-1 sec-1. The current density in mA/m2 is
Distance of a point inside a superconductor from the surface, at which the magnetic field is two-third of its value at the surface, in terms of London penetration depth (λ) is
Inteiplanar distance of a plane passing through points (0,0,1), (1,0,0) and 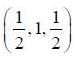 in a crystal with lattice constant 2Å is _________ Å. (Upto two decim al places)
in a crystal with lattice constant 2Å is _________ Å. (Upto two decim al places)
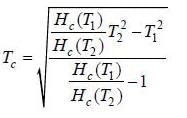
Critical magnetic fields of a superconductor at 2K and 6K is 13.5 mA /m and 4.5 mA /m respectively. The transition temperature of the superconductor is _________ K.
[Upto two decimal places]
Consider a semi-empirical mass formula of the form
M = 939.6(A - Z) + 938.8Z - 14.0A + 13.0 A2/3 + 0.583Z (Z - 1) A-1/3 +19.3(A - 2Z)2 A-1 ±33.5A-3/4
Hence the most stable isobar is
Neutron lias 11011-zero magnetic moment confirm that
According to closed-shell nucleus model parity of 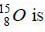
41Ca has a neutron outside closed shell in 1 f7/2. It’s nuclear magnetic momentum is equal to
As one moves along the line of stability from 56Fe to U235 nucleus the binding energy per nucleon decreases from 8 MeV to 7.6 MeV Tliis trend is mainly due to the
Two particles He+ and Li+ are projected in a circular orbit of same radius perpendicular to a given magnetic field
The reaction 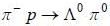 strong interaction is not possible because
strong interaction is not possible because
The life time of a free neutron to decay into a proton, an election and an anti-neutrino is approximately
Consider the strong interaction process

the unknown particle X may be
The binding energy of the neon isotopes 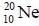 is 160.65 Mev. thus, its atomic mass in atomic unit u, would be
is 160.65 Mev. thus, its atomic mass in atomic unit u, would be
Neutron can be expressed in terms of quark
There is a stream of neutrons with a speed 2.5 x 103 m/s. If half life of neutrons is 700 seconds, the fractions of neutrons that will decay before they travel a distance 10m.
(given mass of neutrons = 1.675 x 10-27kg)
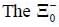 baryon has the following properties Q = -1, S = -2 & B = 1, the quark content is
baryon has the following properties Q = -1, S = -2 & B = 1, the quark content is
The quark content of ∑+, k-, π- and p is indicated
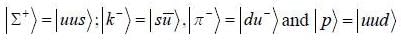
in die process π- + p → k- + ∑+
Considering strong interactions only, which of die following is true?
The conservation law violated by die reaction p → π0 + e+ is conservation of
The wavelength of one of the visible lines in the spectrum of the hyclrogen atom is approximately 6560 Å. It is known that this corresponds to a transition between the states with the principal quantum numbers n = 2 and n = 3. Hie frequency corresponding to the transition from die first excited state to die ground state is_______ x 1015Hz.
(Upto two decimal places)
A sample of certain element is placed in a 0.3 Tesla magnetic field and suitably excited. If the wavelength corresponding to the transition 1P → 1S is 450 nm. then the separation in the Zeeman spectral lines is equal to _______ x 10-12 meter.
(Upto two decim al places)

X-ray spectrum o f a cobalt target (Z = 27) contains strong Kα line of wavelength 0.1785 nm and a weak line having wavelength of 0.1537 nm due to an impurity. The atomic number of impurity element is_______.
|
1 docs|34 tests
|



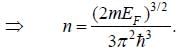
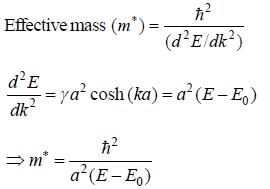
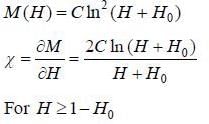
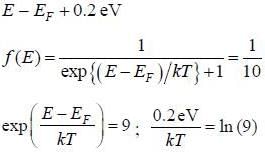
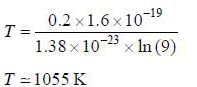
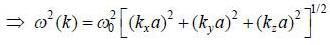
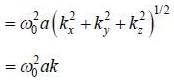
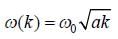
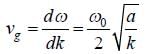
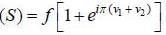
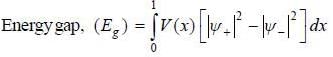
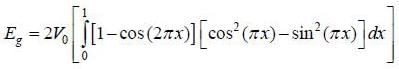
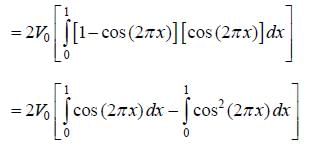

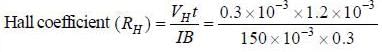
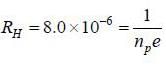
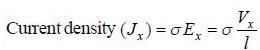
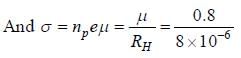
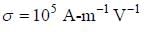
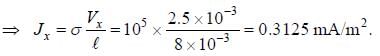
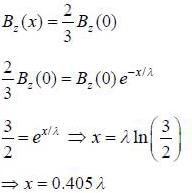
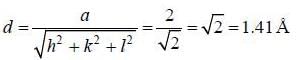
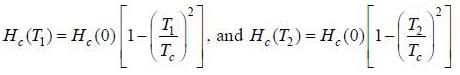
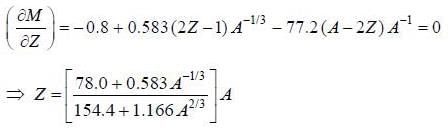


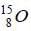 is determined by the angular momentum and parity of the unpaired neutron in the1p1/2 state.
is determined by the angular momentum and parity of the unpaired neutron in the1p1/2 state.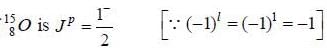
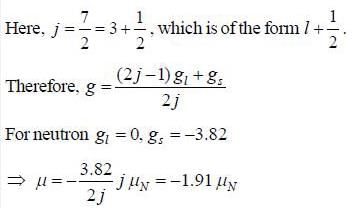
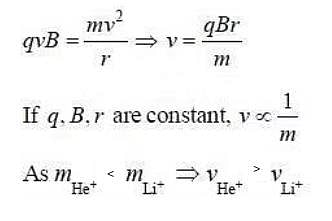
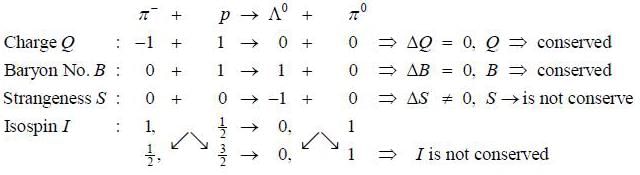
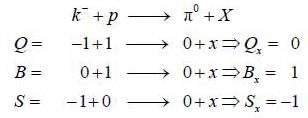
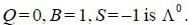
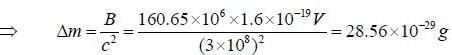
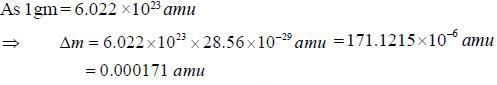
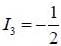
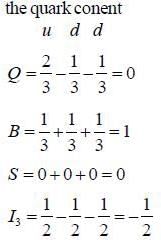
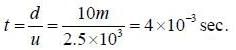

 is dss as it satisfy Q, S & B of
is dss as it satisfy Q, S & B of