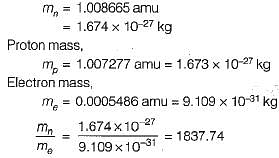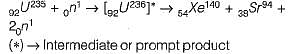Mechanical Engineering Exam > Mechanical Engineering Tests > Topicwise Question Bank for Mechanical Engineering > Test: Nuclear Power Plants - 1 - Mechanical Engineering MCQ
Test: Nuclear Power Plants - 1 - Mechanical Engineering MCQ
Test Description
10 Questions MCQ Test Topicwise Question Bank for Mechanical Engineering - Test: Nuclear Power Plants - 1
Test: Nuclear Power Plants - 1 for Mechanical Engineering 2024 is part of Topicwise Question Bank for Mechanical Engineering preparation. The Test: Nuclear Power Plants - 1 questions and answers have been
prepared according to the Mechanical Engineering exam syllabus.The Test: Nuclear Power Plants - 1 MCQs are made for Mechanical Engineering 2024 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Nuclear Power Plants - 1 below.
Solutions of Test: Nuclear Power Plants - 1 questions in English are available as part of our Topicwise Question Bank for Mechanical Engineering for Mechanical Engineering & Test: Nuclear Power Plants - 1 solutions in
Hindi for Topicwise Question Bank for Mechanical Engineering course. Download more important topics, notes, lectures and mock
test series for Mechanical Engineering Exam by signing up for free. Attempt Test: Nuclear Power Plants - 1 | 10 questions in 30 minutes | Mock test for Mechanical Engineering preparation | Free important questions MCQ to study Topicwise Question Bank for Mechanical Engineering for Mechanical Engineering Exam | Download free PDF with solutions
Test: Nuclear Power Plants - 1 - Question 1
The basic raw material of the nuclear power plant is
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Test: Nuclear Power Plants - 1 - Question 3
Atomic number of an element in the periodic table represents the numbers of
Test: Nuclear Power Plants - 1 - Question 4
The mass number of a substance represents the sum of total number of
Test: Nuclear Power Plants - 1 - Question 5
Which of the following is not identical for an atom and an isotope?
Test: Nuclear Power Plants - 1 - Question 6
The mass of electron as compared to that of neutron is
Detailed Solution for Test: Nuclear Power Plants - 1 - Question 6
Test: Nuclear Power Plants - 1 - Question 7
Which of the following energy acts as a glue which binds the protons and neutrons together in the nucleus?
Detailed Solution for Test: Nuclear Power Plants - 1 - Question 7
Test: Nuclear Power Plants - 1 - Question 8
How many fast neutrons are produced on the average as a product of reaction of each fission of U-235?
Detailed Solution for Test: Nuclear Power Plants - 1 - Question 8
Test: Nuclear Power Plants - 1 - Question 9
The energy released during the fission of one atom of U-235 in MeV is about
Detailed Solution for Test: Nuclear Power Plants - 1 - Question 9
Test: Nuclear Power Plants - 1 - Question 10
Uranium 238 is represented as 92U238. What does it mean?
Detailed Solution for Test: Nuclear Power Plants - 1 - Question 10
|
45 videos|314 tests
|
Information about Test: Nuclear Power Plants - 1 Page
In this test you can find the Exam questions for Test: Nuclear Power Plants - 1 solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Nuclear Power Plants - 1, EduRev gives you an ample number of Online tests for practice
|
45 videos|314 tests
|
Download as PDF