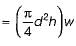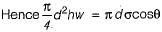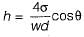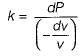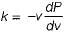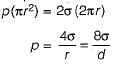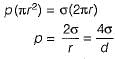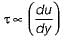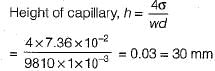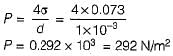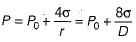Test: Properties of Fluids - 3 - Mechanical Engineering MCQ
20 Questions MCQ Test Fluid Mechanics for Mechanical Engineering - Test: Properties of Fluids - 3
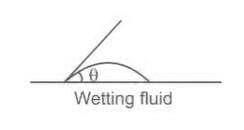
If angle of contact of a drop of liquid is acute than
What is unit of dynamic viscosity of a fluid termed as ’Poise’ equivalent to
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
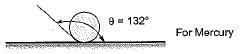
The mercury is known as non-wetting liquid because it does not wet the surface, The mercury posses this property due to
Which one of the following is an example of Bingham plastic?
Which one of the following is a typical example of non-newtonian fluid of Pseudo plastic variety?
Match List-I (Physical properties of fluid) with List-II (Dimensions/definitions) and select the correct answer using the codes given below the lists:
List-I
A. Absolute viscosity
B. Kinematic viscosity
C. Newtonian fluid
D. Surface tension
List-II
1. du/dy is constant
2. Newton per meter
3. Poise
4. constant stress/strain
5. Stroke
Codes:
A B C D
(a) 5 3 1 2
(b) 3 5 2 4
(c) 5 3 4 2
(d) 3 5 1 2
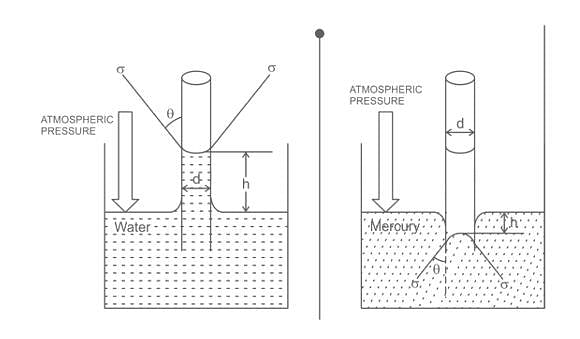
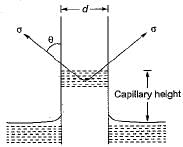
For a glass tube of diameter d, height of capillarity is given by
where w = specific weight of liquid
σ = surface tension
θ = angle of contact between liquid and surface
The approximate value of θ (angle of contact) for mercury is
What is the dimensions of kinematic viscosity of a fluid?
The property by virtue of which a liquid opposes relative motion between its different layers is called
The Bulk modulus of elasticity of a fluid is defined a
In the phenomenon of cavitation, the characteristic fluid property involved is
The difference of pressure between the inside and outside the soap bubble is
The difference of pressure between the inside and outside the liquid jet
The difference of pressure between the inside and outside the liquid drop is
Match List-I (Properties of fluids) with List-ll (Definition) and select the correct answer using the codes given below the lists:
List-I
A. Ideal fluid
B. Newton ion fluid
C. μ/ρ
D. Mercury in glass
List-ll
1. Viscosity does not vary with rate of deformation
2. Fluid of zero viscosity
3. Dynamic viscosity
4. Capillary depression
5. Kinematic viscosity
6. Capillary rise
Codes:
A B C D
(a) 1 2 4 6
(b) 1 2 3 4
(c) 2 1 3 6
(d) 2 1 5 4
The capillary rise at 20°C in clean glass tube of 1 mm diameter containing water is
If the surface tension of water air interface is 0.073 N/m, the gauge pressure inside a rain drop of 1 mm diameter will be
Consider a soap film bubble of diameter D. If the external pressure is P0 and the surface tension of the soap film is σ, the expression for the pressure inside the bubble is
|
56 videos|104 docs|75 tests
|
|
56 videos|104 docs|75 tests
|