Class 10 Exam > Class 10 Tests > Extra Documents, Videos & Tests for Class 10 > Test: Chemical Equations - Class 10 MCQ
Test: Chemical Equations - Class 10 MCQ
Test Description
10 Questions MCQ Test Extra Documents, Videos & Tests for Class 10 - Test: Chemical Equations
Test: Chemical Equations for Class 10 2024 is part of Extra Documents, Videos & Tests for Class 10 preparation. The Test: Chemical Equations questions and answers have been
prepared according to the Class 10 exam syllabus.The Test: Chemical Equations MCQs are made for Class 10 2024 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Chemical Equations below.
Solutions of Test: Chemical Equations questions in English are available as part of our Extra Documents, Videos & Tests for Class 10 for Class 10 & Test: Chemical Equations solutions in
Hindi for Extra Documents, Videos & Tests for Class 10 course. Download more important topics, notes, lectures and mock
test series for Class 10 Exam by signing up for free. Attempt Test: Chemical Equations | 10 questions in 10 minutes | Mock test for Class 10 preparation | Free important questions MCQ to study Extra Documents, Videos & Tests for Class 10 for Class 10 Exam | Download free PDF with solutions
Detailed Solution for Test: Chemical Equations - Question 1
Detailed Solution for Test: Chemical Equations - Question 2
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Test: Chemical Equations - Question 3
The substances that are formed after completion of the reaction are called:
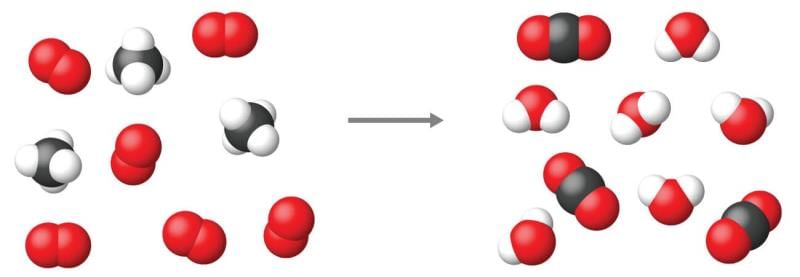

Detailed Solution for Test: Chemical Equations - Question 3
Test: Chemical Equations - Question 4
The reaction in which two compound exchange their ions to form two new compounds is called
Detailed Solution for Test: Chemical Equations - Question 4
Test: Chemical Equations - Question 5
The reaction of H2 gas with oxygen gas to form water is an example of
Detailed Solution for Test: Chemical Equations - Question 5
Test: Chemical Equations - Question 6
The balanced chemical equation for the reaction of zinc metal with hydrochloric acid is:
Detailed Solution for Test: Chemical Equations - Question 6
Test: Chemical Equations - Question 7
The electrolytic decomposition of water gives H2 and O2 in the ratio of
Detailed Solution for Test: Chemical Equations - Question 7
Test: Chemical Equations - Question 8
We store silver chloride in a dark coloured bottle because it is
Detailed Solution for Test: Chemical Equations - Question 8
Test: Chemical Equations - Question 9
When copper sulphate solution reacts with iron metal, copper metal is formed. This reaction comes under which of the following category?
Detailed Solution for Test: Chemical Equations - Question 9
Test: Chemical Equations - Question 10
A chemical reaction has taken place can be represented by which of the following condition?
Detailed Solution for Test: Chemical Equations - Question 10
|
5 videos|292 docs|59 tests
|
Information about Test: Chemical Equations Page
In this test you can find the Exam questions for Test: Chemical Equations solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Chemical Equations, EduRev gives you an ample number of Online tests for practice
|
5 videos|292 docs|59 tests
|
Download as PDF

















