Case Based Questions Test: The p-Block Elements - NEET MCQ
12 Questions MCQ Test Topic-wise MCQ Tests for NEET - Case Based Questions Test: The p-Block Elements
Direction: Read the passage given below and answer the following questions:
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone gives carbonyl compounds with
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone gives carbonyl compounds with
Direction: Read the passage given below and answer the following questions:
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone reacts with moist iodine gives
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone reacts with moist iodine gives
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Direction: Read the passage given below and answer the following questions:
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone acts as an oxidising agent due to
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Ozone acts as an oxidising agent due to
Direction: Read the passage given below and answer the following questions:
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. The colour of ozone molecule is
Direction: Read the passage given below and answer the following questions:
In spite of the predictions of stable noble gas compounds since at least 1902, unsuccessful attempts at their synthesis gave rise to the widely held opinion that noble gases are not only noble but also inert. It was not until 1962 that this dogma was shattered when Bartlett in Canada published the first stable noble gas compound XePtF6. This discovery triggered a worldwide frenzy in this area, and within a short time span many new xenon, radon, and krypton compounds were prepared and characterized. The recent discoveries show the ability of xenon to act as a ligand . The discovery by Seppelt’s group that more than one xenon atom can attach itself to a metal center which in the case of gold leads to surprisingly stable Au- Xe bonds. The bonding in [AuXe4]2+ involves 4 Xe ligands attached by relatively strong bonds to a single Au(II) center in a square planar arrangement with a Xe-Au bond length of about 274 pm This discovery provides not only the first example of multiple xenon ligands but also represents the first strong metal - xenon bond.
Q. In the complex ion [AuXe4]2+, Xe acts as:
Direction: Read the passage given below and answer the following questions:
In spite of the predictions of stable noble gas compounds since at least 1902, unsuccessful attempts at their synthesis gave rise to the widely held opinion that noble gases are not only noble but also inert. It was not until 1962 that this dogma was shattered when Bartlett in Canada published the first stable noble gas compound XePtF6. This discovery triggered a worldwide frenzy in this area, and within a short time span many new xenon, radon, and krypton compounds were prepared and characterized. The recent discoveries show the ability of xenon to act as a ligand . The discovery by Seppelt’s group that more than one xenon atom can attach itself to a metal center which in the case of gold leads to surprisingly stable Au- Xe bonds. The bonding in [AuXe4]2+ involves 4 Xe ligands attached by relatively strong bonds to a single Au(II) center in a square planar arrangement with a Xe-Au bond length of about 274 pm This discovery provides not only the first example of multiple xenon ligands but also represents the first strong metal - xenon bond.
Q. Hybridisation shown by Au in [AuXe4]2+ is:
Direction: Read the passage given below and answer the following questions:
In spite of the predictions of stable noble gas compounds since at least 1902, unsuccessful attempts at their synthesis gave rise to the widely held opinion that noble gases are not only noble but also inert. It was not until 1962 that this dogma was shattered when Bartlett in Canada published the first stable noble gas compound XePtF6. This discovery triggered a worldwide frenzy in this area, and within a short time span many new xenon, radon, and krypton compounds were prepared and characterized. The recent discoveries show the ability of xenon to act as a ligand . The discovery by Seppelt’s group that more than one xenon atom can attach itself to a metal center which in the case of gold leads to surprisingly stable Au- Xe bonds. The bonding in [AuXe4]2+ involves 4 Xe ligands attached by relatively strong bonds to a single Au(II) center in a square planar arrangement with a Xe-Au bond length of about 274 pm This discovery provides not only the first example of multiple xenon ligands but also represents the first strong metal - xenon bond.
Q. Compounds of noble gases except _______ are known.
Direction: Read the passage given below and answer the following questions:
Ozone is an unstable, dark blue diamagnetic gas. It absorbs the UV radiation strongly, thus protecting the people on earth from the harmful UV-radiation from the sun. The use of chlorofluorocarbon (CFC) in aerosol and refrigerator and their subsequent escape into the atmosphere, is blamed for making holes in the ozone layer over the Antarctica. Ozone acts as a strong oxidising agent in acidic and alkaline medium. For this property, ozone is used as a germicide and disinfectant for sterilizing water. It is also used in laboratory for the ozonolysis of organic compounds and in industry for the manufacture of potassium permanganate, artificial silk, etc.
Q. Xe is a ___________ ligand
Direction: Read the passage given below and answer the following questions:
In the last 10 years much has been learned about the molecular structure of elemental sulfur. lt is now known that many different types of rings are sufficiently metastable to exist at room temperature for several days. It is known that at high temperature, the equilibrium composition allows for a variety of rings and chains to exist in comparable concentration, and it is known that at the boiling point and above, the vapour as well as the liquid contains small species with three, four, and five atoms.
The sulfur atom has the same number of valence electrons as oxygen. Thus, sulfur atoms S2 and S3 have physical and chemical properties analogous to those of oxygen and ozone. S2 has a ground state of 38 σ3s2σ*3s2σ3pz2π3px 2 = π3py2π*3px1 = π*3py1. S3 , thiozone has a well known uv spectrum, and has a bent structure, analogous to its isovalent molecules O3, SO2, and S2O. The chemistry of the two elements, sulphur and oxygen, differs because sulfur has a pronounced tendency for catenation. The most frequently quoted explanation is based on the electron structure of the atom. Sulfur has low-lying unoccupied 3d orbitals, and it is widely believed that the 4s and 3d orbitals of sulfur participate in bonding in a manner similar to the participation of 2s and 2p orbitals in carbon.
Q. In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion (A): Sulphur belongs to same group in the periodic table as oxygen.
Reason (R): S2 has properties analogous to O2.
Direction: Read the passage given below and answer the following questions:
In the last 10 years much has been learned about the molecular structure of elemental sulfur. lt is now known that many different types of rings are sufficiently metastable to exist at room temperature for several days. It is known that at high temperature, the equilibrium composition allows for a variety of rings and chains to exist in comparable concentration, and it is known that at the boiling point and above, the vapour as well as the liquid contains small species with three, four, and five atoms.
The sulfur atom has the same number of valence electrons as oxygen. Thus, sulfur atoms S2 and S3 have physical and chemical properties analogous to those of oxygen and ozone. S2 has a ground state of 38 σ3s2σ*3s2σ3pz2π3px 2 = π3py2π*3px1 = π*3py1. S3 , thiozone has a well known uv spectrum, and has a bent structure, analogous to its isovalent molecules O3, SO2, and S2O. The chemistry of the two elements, sulphur and oxygen, differs because sulfur has a pronounced tendency for catenation. The most frequently quoted explanation is based on the electron structure of the atom. Sulfur has low-lying unoccupied 3d orbitals, and it is widely believed that the 4s and 3d orbitals of sulfur participate in bonding in a manner similar to the participation of 2s and 2p orbitals in carbon.
Q. In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion (A): S2 is paramagnetic in nature
Reason (R): The electrons in π*3px and π*3py orbitals in S2 are unpaired.
Direction: Read the passage given below and answer the following questions:
In the last 10 years much has been learned about the molecular structure of elemental sulfur. lt is now known that many different types of rings are sufficiently metastable to exist at room temperature for several days. It is known that at high temperature, the equilibrium composition allows for a variety of rings and chains to exist in comparable concentration, and it is known that at the boiling point and above, the vapour as well as the liquid contains small species with three, four, and five atoms.
The sulfur atom has the same number of valence electrons as oxygen. Thus, sulfur atoms S2 and S3 have physical and chemical properties analogous to those of oxygen and ozone. S2 has a ground state of 38 σ3s2σ*3s2σ3pz2π3px 2 = π3py2π*3px1 = π*3py1. S3 , thiozone has a well known uv spectrum, and has a bent structure, analogous to its isovalent molecules O3, SO2, and S2O. The chemistry of the two elements, sulphur and oxygen, differs because sulfur has a pronounced tendency for catenation. The most frequently quoted explanation is based on the electron structure of the atom. Sulfur has low-lying unoccupied 3d orbitals, and it is widely believed that the 4s and 3d orbitals of sulfur participate in bonding in a manner similar to the participation of 2s and 2p orbitals in carbon.
Q. In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion (A): Sulphur has a greater tendency for catenation than oxygen.
Reason (R): 3d and 4s orbitals of Sulphur have same energy.
Direction: Read the passage given below and answer the following questions:
In the last 10 years much has been learned about the molecular structure of elemental sulfur. lt is now known that many different types of rings are sufficiently metastable to exist at room temperature for several days. It is known that at high temperature, the equilibrium composition allows for a variety of rings and chains to exist in comparable concentration, and it is known that at the boiling point and above, the vapour as well as the liquid contains small species with three, four, and five atoms.
The sulfur atom has the same number of valence electrons as oxygen. Thus, sulfur atoms S2 and S3 have physical and chemical properties analogous to those of oxygen and ozone. S2 has a ground state of 38 σ3s2σ*3s2σ3pz2π3px 2 = π3py2π*3px1 = π*3py1. S3 , thiozone has a well known uv spectrum, and has a bent structure, analogous to its isovalent molecules O3, SO2, and S2O. The chemistry of the two elements, sulphur and oxygen, differs because sulfur has a pronounced tendency for catenation. The most frequently quoted explanation is based on the electron structure of the atom. Sulfur has low-lying unoccupied 3d orbitals, and it is widely believed that the 4s and 3d orbitals of sulfur participate in bonding in a manner similar to the participation of 2s and 2p orbitals in carbon.
Q. In the following questions, a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices on the basis of the above passage.
Assertion (A): Thiozone has bent structure like ozone.
Reason (R): Ozone has a lone pair which makes the molecule bent.
|
9 docs|1272 tests
|



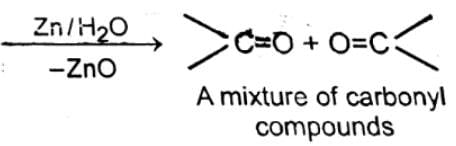
 O2 + O (nascent oxygen) Since nascent oxygen is very reactive, therefore, O3 acts as a powerful oxidising agent.
O2 + O (nascent oxygen) Since nascent oxygen is very reactive, therefore, O3 acts as a powerful oxidising agent.














