Chemistry: Topic-wise Test- 6 - NEET MCQ
30 Questions MCQ Test NEET Mock Test Series 2025 - Chemistry: Topic-wise Test- 6
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
When a zinc rod is kept in a copper nitrate solution what happens?
In which of the following complexes, the nickel metal is in the highest oxidation state?
Which of the following can be termed as mixed complex?
Which of the following ligand gives chelate complexes?
Arrange the following alcohols, hydrocarbon and ether in order of their increasing boiling points Pentan – 1 – ol, n – butane, pentanal, ethoxyethane.
Propanamide on treatment with bromine in an aqueous solution of sodium hydroxide gives:
We can obtain ethylamine by Hoffmann bromamide reaction. The amide used in this reaction is:
Nitro compounds are reduced to amines. The catalyst that is preferred is:
The following reaction takes place in the presence of
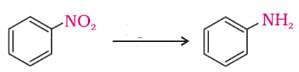
Nitro compounds are reduced by iron scrap and hydrochloric acid to yield one of the following compounds.
Which one of the following is the weakest base in gaseous phase?
Benzene diazonium chloride when reacts with hypophosphorus acid produces
p-amino azo benzene is obtained by treating diazoniumchloride with:
When hypophosphorous acid is treated with diazonium salts, it is reduced to:
Which of the following amine will form stable diazonium salt at 273-283 K ?
Replacement of diazo group by other groups is helpful in preparing those substituted aromatic compounds which cannot be prepared by
To balance the oxygen atom in the given reaction in acidic medium
Cr2O72- (aq) → Cr3+(aq) we
The oxidation half reaction for following reaction is
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)
A standard hydrogen electrode has zero electrode potential because
|
1 videos|26 docs|111 tests
|
|
1 videos|26 docs|111 tests
|


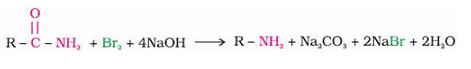
 is a tertiary amine having IUPAC name as:
is a tertiary amine having IUPAC name as: