JEE Exam > JEE Tests > Mock Tests for JEE Main and Advanced 2025 > JEE Main Chemistry Mock Test- 8 - JEE MCQ
JEE Main Chemistry Mock Test- 8 - JEE MCQ
Test Description
25 Questions MCQ Test Mock Tests for JEE Main and Advanced 2025 - JEE Main Chemistry Mock Test- 8
JEE Main Chemistry Mock Test- 8 for JEE 2024 is part of Mock Tests for JEE Main and Advanced 2025 preparation. The JEE Main Chemistry Mock Test- 8 questions and answers have been
prepared according to the JEE exam syllabus.The JEE Main Chemistry Mock Test- 8 MCQs are made for JEE 2024 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for JEE Main Chemistry Mock Test- 8 below.
Solutions of JEE Main Chemistry Mock Test- 8 questions in English are available as part of our Mock Tests for JEE Main and Advanced 2025 for JEE & JEE Main Chemistry Mock Test- 8 solutions in
Hindi for Mock Tests for JEE Main and Advanced 2025 course. Download more important topics, notes, lectures and mock
test series for JEE Exam by signing up for free. Attempt JEE Main Chemistry Mock Test- 8 | 25 questions in 60 minutes | Mock test for JEE preparation | Free important questions MCQ to study Mock Tests for JEE Main and Advanced 2025 for JEE Exam | Download free PDF with solutions
JEE Main Chemistry Mock Test- 8 - Question 1
The heating together of sodium ethoxide and ethyl chloride will give
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
JEE Main Chemistry Mock Test- 8 - Question 4
The rate, at which a substance reacts at constant temperature, depends upon its
JEE Main Chemistry Mock Test- 8 - Question 5
What will be the weight of deposited silver on passing 965 coulombs of electricity in solution of AgNO₃?
JEE Main Chemistry Mock Test- 8 - Question 6
The indicator that is obtained by coupling the diazonium salt of sulphanilic acid with N,N-dimethylaniline is
JEE Main Chemistry Mock Test- 8 - Question 7
For the reaction A→B, the rate law expression is rate = k[A]. Which of the following statements is incorrect?
JEE Main Chemistry Mock Test- 8 - Question 9
AgCl is soluble in NH₄OH solution. The solubility is due to formation of
JEE Main Chemistry Mock Test- 8 - Question 10
The name of the element with atomic number 100 was adopted in honour of
Detailed Solution for JEE Main Chemistry Mock Test- 8 - Question 10
JEE Main Chemistry Mock Test- 8 - Question 11
Which one of the following is the strongest acid?
JEE Main Chemistry Mock Test- 8 - Question 13
Which one of the following can be considered as weak electrolyte ?
JEE Main Chemistry Mock Test- 8 - Question 14
A mixture of a weak acid (say acetic acid )and its salt with a strong base (say sodium acetate )is a buffer solution.Which other pair of substances from the following may have a similar property ?
JEE Main Chemistry Mock Test- 8 - Question 15
Which of the following is not true in case of reaction with heated copper at 300oC?
Detailed Solution for JEE Main Chemistry Mock Test- 8 - Question 16
JEE Main Chemistry Mock Test- 8 - Question 18
Ethyl bromide treatements with alcoholic KOH gives
Detailed Solution for JEE Main Chemistry Mock Test- 8 - Question 19
JEE Main Chemistry Mock Test- 8 - Question 20
Which one of the following solutions of sulphuric acid will exactly neutralise 25 ml of 0.2 M sodium hydroxide solution ?
*Answer can only contain numeric values
JEE Main Chemistry Mock Test- 8 - Question 21
Sum of number of ions in aqueous solution of CrCl3.5NH3 and CrCl3 . 4NH3.
*Answer can only contain numeric values
JEE Main Chemistry Mock Test- 8 - Question 22
Find the planner species out of the following species
SF2, SF4, SF6 , SO2, SO3
*Answer can only contain numeric values
JEE Main Chemistry Mock Test- 8 - Question 23
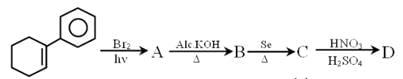
if ‘x’ is total number of benzene ring in compound(C) and ‘y’ is the total number of degree of unsaturation in compound (D) then the value of (x + y) is
*Answer can only contain numeric values
JEE Main Chemistry Mock Test- 8 - Question 24
Identify total number of compound (s) which are unstable at room temperature ?
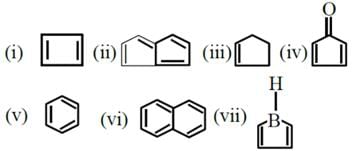
*Answer can only contain numeric values
JEE Main Chemistry Mock Test- 8 - Question 25
27 gm of Al react with excess of oxygen to give 4.95 gm of Al2O3, calculate % yield of reaction.
Detailed Solution for JEE Main Chemistry Mock Test- 8 - Question 25
|
357 docs|148 tests
|
Information about JEE Main Chemistry Mock Test- 8 Page
In this test you can find the Exam questions for JEE Main Chemistry Mock Test- 8 solved & explained in the simplest way possible.
Besides giving Questions and answers for JEE Main Chemistry Mock Test- 8, EduRev gives you an ample number of Online tests for practice
|
357 docs|148 tests
|
Download as PDF

















