Test: Thermal Expansion - NEET MCQ
10 Questions MCQ Test - Test: Thermal Expansion
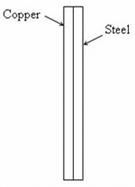
Two identical rectangular strips, one of copper and the other of steel, are riveted as shown to form a bi-metal strip. On heating, the bi-metal strip will

Four cylindrical rods of different radii and lengths are used to connect two heat reservoirs at fixed temperatures t1 and t2 respectively. From the following pick out the rod which will conduct the maximum quantity of heat:
The temperature for which the reading on Celsius and Fahrenheit scales are identical is
A steel tape is calibrated at 20° C. A piece of wood is being measured by steel tape at 10°C and reading is 30 cm on the tape. The real length of the wood is:
Which phase of matter has maximum value of temperature coefficient of cubical expansion?
The coefficient of liner expansion of a cubical crystal along three mutually perpendicular direction is  and
and  . What is the coefficient of cubical expansion of crystal?
. What is the coefficient of cubical expansion of crystal?
Relationship for converting the Fahrenheit and Celsius scales is
The relation between coefficient of volume expansion 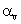 and coefficient of linear expansion
and coefficient of linear expansion 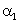 is
is



















