Test: Acids, Bases & Salts - CTET & State TET MCQ
20 Questions MCQ Test NCERT Textbooks (Class 6 to Class 12) - Test: Acids, Bases & Salts
'Litmus', a natural dye, is an extract of which of the following?
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
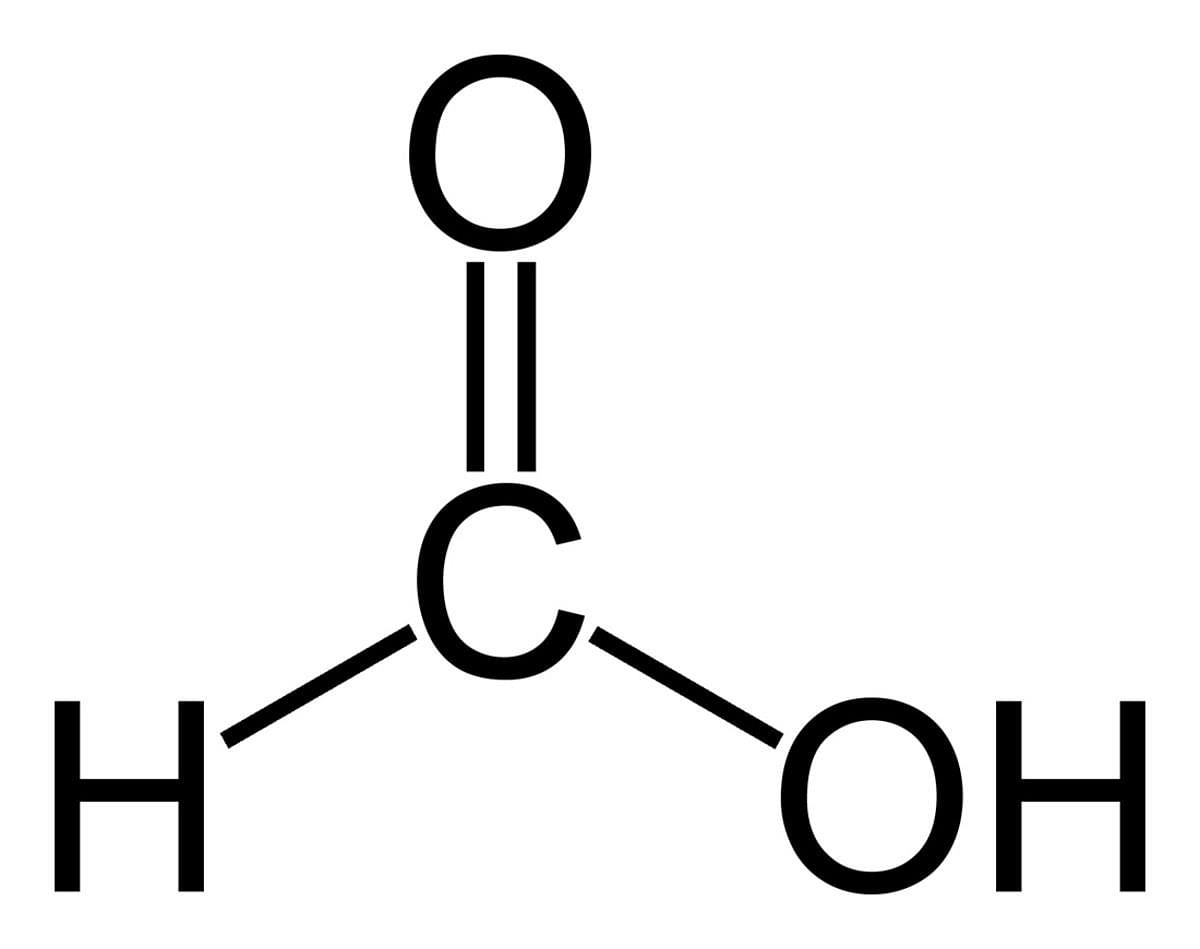
Carbon dioxide reacts with calcium hydroxide to form calcium carbonate and water. Ca(OH)2 + CO2 → CaCO3 + H2O. This reaction is known as
Assertion: Metal Oxides react with acids to form salt and water.
Reason: Most of the metal oxides are acidic in nature.
Some alkali metal salts are coloured Na2CrO4 —yellow, KMnO4- pink, K2MnO4 -green. It is due to:
Molten sodium chloride conducts electric current due to the presence of:
The pH of a compound is found to be 9. The compound can be:
To protect tooth decay, we are advised to brush our teeth regularly. The nature of the toothpaste commonly used is:
Sodium chloride imparts a yellow colour to the Bunsen flame. This can be interpreted due to the
A milkman added a small pinch of baking soda to fresh milk which had pH close to 6. As a result, pH of the medium
|
3 videos|687 docs|659 tests
|
|
3 videos|687 docs|659 tests
|