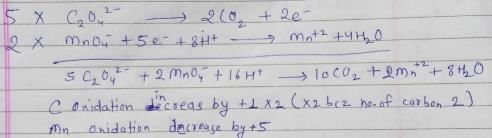NEET Exam > NEET Tests > Chemistry Class 11 > Test: Balancing Redox Reactions - NEET MCQ
Test: Balancing Redox Reactions - NEET MCQ
Test Description
5 Questions MCQ Test Chemistry Class 11 - Test: Balancing Redox Reactions
Test: Balancing Redox Reactions for NEET 2024 is part of Chemistry Class 11 preparation. The Test: Balancing Redox Reactions questions and answers have been
prepared according to the NEET exam syllabus.The Test: Balancing Redox Reactions MCQs are made for NEET 2024 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: Balancing Redox Reactions below.
Solutions of Test: Balancing Redox Reactions questions in English are available as part of our Chemistry Class 11 for NEET & Test: Balancing Redox Reactions solutions in
Hindi for Chemistry Class 11 course. Download more important topics, notes, lectures and mock
test series for NEET Exam by signing up for free. Attempt Test: Balancing Redox Reactions | 5 questions in 10 minutes | Mock test for NEET preparation | Free important questions MCQ to study Chemistry Class 11 for NEET Exam | Download free PDF with solutions
Test: Balancing Redox Reactions - Question 1
To balance the oxygen atom in the given reaction in acidic medium
Cr2O72- (aq) → Cr3+(aq) we
Cr2O72- (aq) → Cr3+(aq) we
Detailed Solution for Test: Balancing Redox Reactions - Question 1
Test: Balancing Redox Reactions - Question 2
The oxidation half reaction for following reaction is
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)
Fe2+(aq) + Cr2O72-(aq) → Fe3+ (aq) + Cr3+(aq)
Detailed Solution for Test: Balancing Redox Reactions - Question 2
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Test: Balancing Redox Reactions - Question 3
In this method, the two half equations are balanced separately and then added together to give balanced equation
Detailed Solution for Test: Balancing Redox Reactions - Question 3
Test: Balancing Redox Reactions - Question 4
The process in which the strength of an unknown solution is calculated using a known standard solution.
Detailed Solution for Test: Balancing Redox Reactions - Question 4
Test: Balancing Redox Reactions - Question 5
For the redox reaction
MnO4– + C2O42- + H+ → Mn2+ + CO2 + H2O
The correct coefficients of the reactants for the balanced reaction are:
Detailed Solution for Test: Balancing Redox Reactions - Question 5
|
129 videos|244 docs|88 tests
|
Information about Test: Balancing Redox Reactions Page
In this test you can find the Exam questions for Test: Balancing Redox Reactions solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: Balancing Redox Reactions, EduRev gives you an ample number of Online tests for practice