NEET Exam > NEET Tests > Chemistry Class 12 > Test: General Study of Carbonyl Compounds - NEET MCQ
Test: General Study of Carbonyl Compounds - NEET MCQ
Test Description
10 Questions MCQ Test Chemistry Class 12 - Test: General Study of Carbonyl Compounds
Test: General Study of Carbonyl Compounds for NEET 2024 is part of Chemistry Class 12 preparation. The Test: General Study of Carbonyl Compounds questions and answers have been
prepared according to the NEET exam syllabus.The Test: General Study of Carbonyl Compounds MCQs are made for NEET 2024 Exam. Find important
definitions, questions, notes, meanings, examples, exercises, MCQs and online tests for Test: General Study of Carbonyl Compounds below.
Solutions of Test: General Study of Carbonyl Compounds questions in English are available as part of our Chemistry Class 12 for NEET & Test: General Study of Carbonyl Compounds solutions in
Hindi for Chemistry Class 12 course. Download more important topics, notes, lectures and mock
test series for NEET Exam by signing up for free. Attempt Test: General Study of Carbonyl Compounds | 10 questions in 15 minutes | Mock test for NEET preparation | Free important questions MCQ to study Chemistry Class 12 for NEET Exam | Download free PDF with solutions
Test: General Study of Carbonyl Compounds - Question 1
Which of the following statements are correct in case of the carbonyl bond between carbon and oxygen?
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 1
Test: General Study of Carbonyl Compounds - Question 2
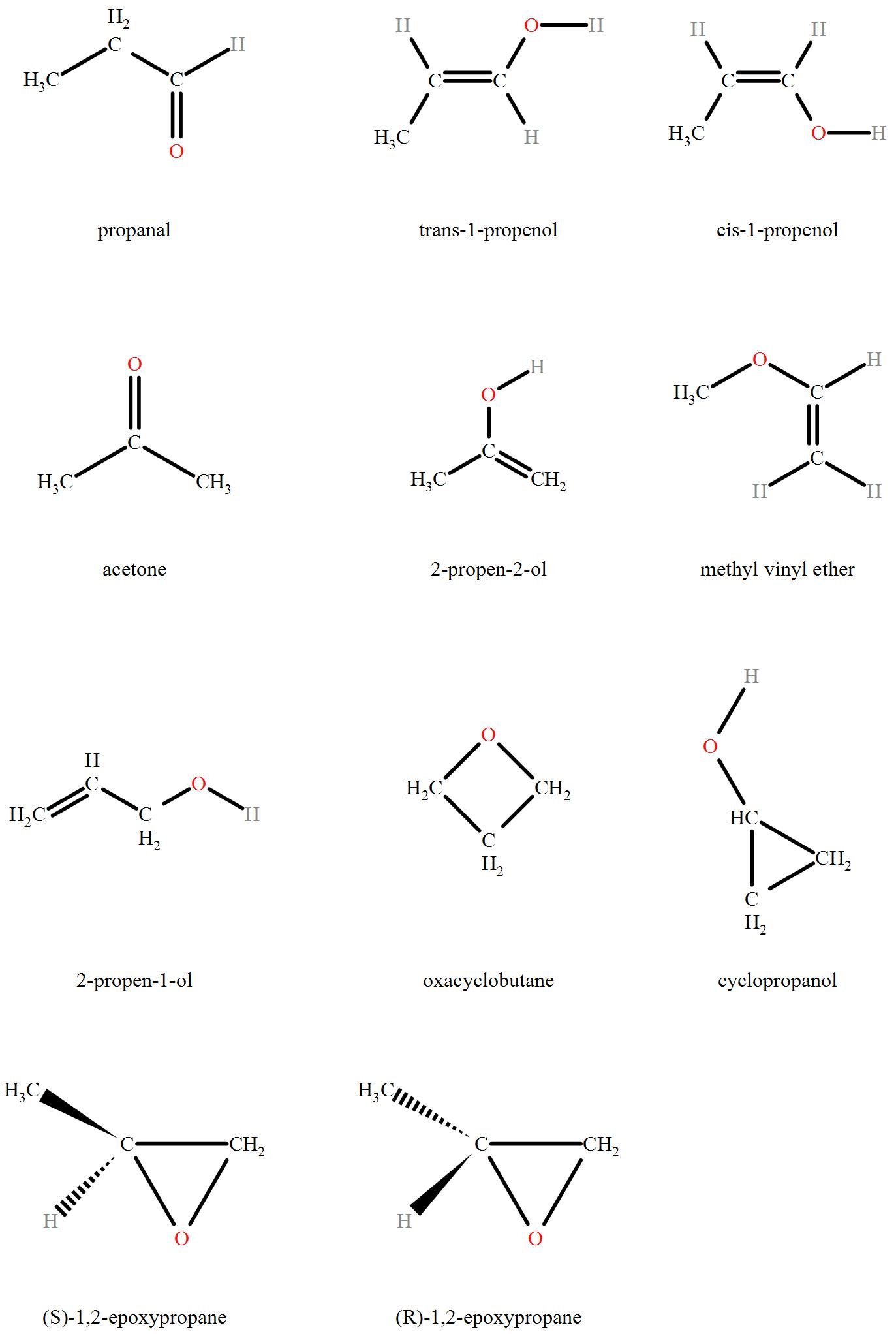
How many structural isomers can compound with molecular formula ‘C3H6O’ have?
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 2
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
Test: General Study of Carbonyl Compounds - Question 3
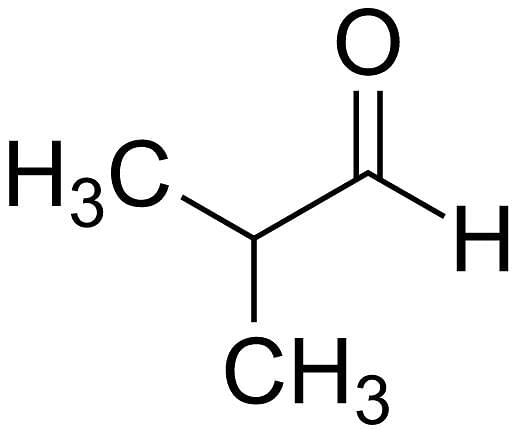
What is the common name of 2-methyl-propanal?
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 3
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 4
Test: General Study of Carbonyl Compounds - Question 5
Carbonyl compounds when oxidized easily to form carboxylic group involves
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 5
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 6
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 7
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 8
Test: General Study of Carbonyl Compounds - Question 9
Which of the following statement about C=O and C=C is correct?
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 9
Test: General Study of Carbonyl Compounds - Question 10
Propanone and prop-2-en-1-ol are examples of which type of isomerism?
Detailed Solution for Test: General Study of Carbonyl Compounds - Question 10
|
108 videos|286 docs|123 tests
|
Information about Test: General Study of Carbonyl Compounds Page
In this test you can find the Exam questions for Test: General Study of Carbonyl Compounds solved & explained in the simplest way possible.
Besides giving Questions and answers for Test: General Study of Carbonyl Compounds, EduRev gives you an ample number of Online tests for practice