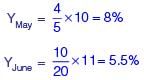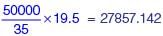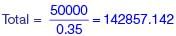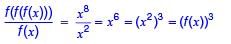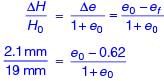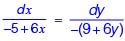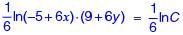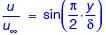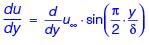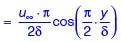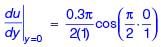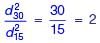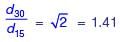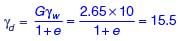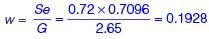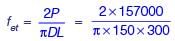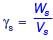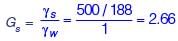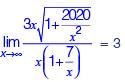Practice Test: Gate Civil Engineering(CE) 2020 Paper: (Session II) - GATE MCQ
30 Questions MCQ Test - Practice Test: Gate Civil Engineering(CE) 2020 Paper: (Session II)
Select the word that fits the analogy:
Partial : Impartial : : Popular : ________
Partial : Impartial : : Popular : ________
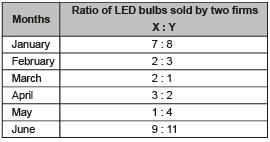
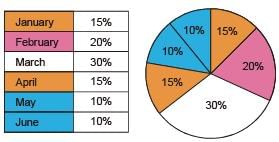
The monthly distribution of 9 Watt LED bulbs sold by two firms X and Y from January to June 2018 is shown in the pie-chart and the corresponding table. If the total number of LED bulbs sold by two firms during April-June 2018 is 50000, then the number of LED bulbs sold by the firm Y during April-June 2018 is _________.


| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
After the inauguration of the new building, the head of department (HOD) collated faculty preferences for office space. P wanted a room adjacent to the lab. Q wanted to be close to the lift. R wanted a view of the playground and S wanted a corner office.
Assuming that everyone was satisfied, which among the following shows a possible allocation?
Assuming that everyone was satisfied, which among the following shows a possible allocation?
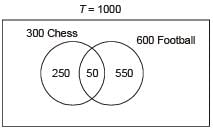
In a school of 1000 students, 300 students play chess and 600 students play football. If 50 students play both chess and football, the number of students who play neither is ________.
Select the most appropriate word that can replace the underlined word without changing the meaning of the sentence:
Now-a-days, most children have a tendency to belittle the legitimate concerns of their parents.
For the year 2019, which of the previous year’s calendar can be used?
If f (x) = x2 for each 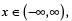 then
then 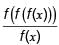 is equal to _________.
is equal to _________.
Rescue teams deployed _________ disaster hit areas combat ________ a lot of difficulties to save the people.
Nominal interest rate is defined as the amount paid by the borrower to the lender for using the borrowed amount for a specific period of time. Real interest rate calculated on the basis of actual value (inflation-adjusted), is approximately equal to the difference between nominal rate and expected rate of inflation in the economy.
Which of the following assertions is best supported by the above information?
The ratio of ‘the sum of the odd positive integers from 1 to 100’ to ‘the sum of the even positive integers from 150 to 200’ is _________.
For an axle load of 15 tonne on a road, the Vehicle Damage Factor (round off to two decimal places), in terms of the standard axle load of 8 tonne, is _________.
Muskingum method is used in
The traffic starts discharging from an approach at an intersection with the signal turning green. The constant headway considered from the fourth or fifth headway position is referred to as
Soil deposit formed due to transportation by wind is termed as
The relationship between oxygen consumption and equivalent biodegradable organic removal (i.e. BOD) in a closed container with respect to time is shown in the figure.
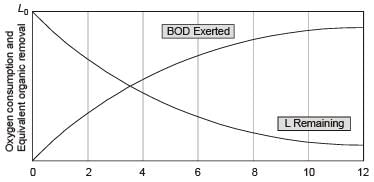
Assume that the rate of oxygen consumption is directly proportional to the amount of degradable organic matter and is expressed as 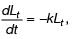 where, L1 (in mg/litre) is the oxygen equivalent of the organics remaining at time t and k (in d–1) is the degradation rate constant. L0 is the oxygen of organic matter at time, t = 0
where, L1 (in mg/litre) is the oxygen equivalent of the organics remaining at time t and k (in d–1) is the degradation rate constant. L0 is the oxygen of organic matter at time, t = 0
In the above context, the correct expression is
A one-dimensional consolidation test is carried out on a standard 19 mm thick clay sample. The oedometer’s deflection gauge indicates a reading of 2.1 mm, just before removal of the load, without allowing any swelling. The void ratio is 0.62 at this stage. The initial void ratio (round off to two decimal places) of the standard specimen is ________.
The velocity components in the x and y directions for an incompressible flow are given as u = (–5 + 6x) and v = –(9 + 6y), respectively. The equation of the streamline is
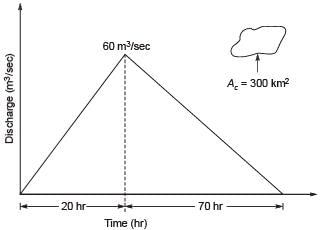
A triangular direct runoff hydrograph due to a storm has a time base of 90 hours. The peak flow of 60 m3/s occurs at 20 hours from the start of the storm. The area of catchment is 300 km2. The rainfall excess of the storm (in cm), is
24-h traffic count at a road section was observed to be 1000 vehicles on a Tuesday in the month of July. If daily adjustment factor for Tuesday is 1.121 and monthly adjustment factor for July is 0.913, the Annual Average Daily Traffic (in veh/day, round off to the nearest integer) is _______.
Velocity distribution in a boundary layer is given by 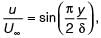 where u is the velocity at vertical coordinate y, U∞ is the free stream velocity and δ is the boundary layer thickness. The values of U∞ and δ are 0.3 m/s and 1.0 m, respectively. The velocity gradient
where u is the velocity at vertical coordinate y, U∞ is the free stream velocity and δ is the boundary layer thickness. The values of U∞ and δ are 0.3 m/s and 1.0 m, respectively. The velocity gradient 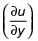 (in s–1, round off to two decimal places) at y = 0, is ________.
(in s–1, round off to two decimal places) at y = 0, is ________.
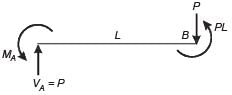
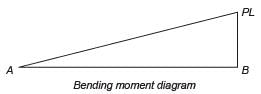
A weightless cantilever beam of span L is loaded as shown in the figure. For the entire span of the beam, the material properties are identical and the cross-section is rectangular with constant width.
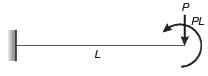
From the flexure-critical perspective, the most economical longitudinal profile of the beam to carry the given loads amongst the options given below, is
Two identically sized primary settling tanks receive water for Type-I settling (discrete particles in dilute suspension) under laminar flow conditions. The surface overflow rate (SOR) maintained in the two tanks are 30 m3/m2.d and 15 m3/m2.d. The lowest diameters of the particles, which shall be settled out completely under SORs of 30 m3/m2.d are designated as d30 and d15 respectively. The ratio d30/d15 (round off to two decimal places), is ___.
As per IS 456:2000, the pH value of water for concrete mix shall NOT be less than
Superpassage is a canal cross-drainage structure in which
A soil has dry weight of 15.5 kN/m3, specific gravity of 2.65 and degree of saturation of 72%. Considering the unit weight of water as 10 kN/m3, the water content of the soil (in %, round off to two decimal places) is ________.
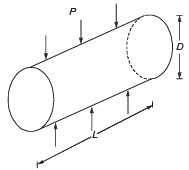
The maximum applied load on a cylindrical concrete specimen of diameter 150 mm and length 300 mm tested as per the split tensile strength test guidelines of IS 5816 : 1999 is 157 kN. The split tensile strength (in MPa, round off to one decimal place) of the specimen is _______.
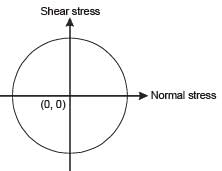
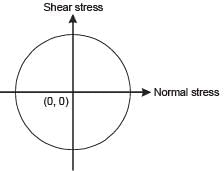
The state of stress represented by Mohr’s circle shown in the figure is
The ratio of the plastic moment capacity of a beam section to its yield moment capacity is termed as
A sample of 500 g dry sand, when poured into a 2 litre capacity cylinder which is partially filled with water, displaces 188 cm3 of water. The density of water is 1 g/cm3. The specific gravity of the sand is
The value of 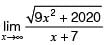 is
is


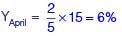 ...(A)
...(A)