Test: Heat Exchanger Level - 1 - Mechanical Engineering MCQ
10 Questions MCQ Test Heat Transfer - Test: Heat Exchanger Level - 1
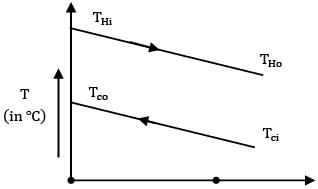
For a heat exchanger the inlet and outlet temperatures of hot and cold fluid are
The type of heat exchanger is
Th1 = 150oC Tc1 = 20oC
Th2 = 70oC Tc2 = 80oC
It is given that for a heat exchangerTc1 and Tc2 are the temperatures of cold fluid at entry and exit respectively and are the temperatures of hot fluid at entry and exit point. If cold fluid has lower heat capacity rate as compared to hot fluid then effectiveness of the heat exchanger is given by
| 1 Crore+ students have signed up on EduRev. Have you? Download the App |
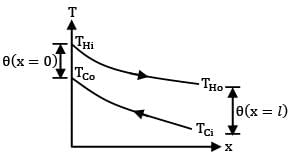
Which one of the following heat exchanger gives parallel straight line pattern of temperature distribution for both cold and hot fluid
In case of boiler in a steam power plant which of the following configurations gives maximum logarithmic mean temperature difference for given values of heat capacity ratios, inlet and outlet temperatures, over all heat transfer coefficient and heat exchange area
Consider the following statements The flow configuration in a heat exchanger whether counter flow or otherwise, will not matter if 1. a liquid is evaporating 2. a vapour is condensing 3. mass flow rate of one of the fluids is far greater Which of these statements correct?
Hot gases enter a heat exchanger at 200°C and leave at 150°C. The cold air enters at 40°C and leaves at 140°C . The capacity ratio of the heat exchanger will be
For a balanced counter flow heat exchanger (i.e. mh ch = mc cc), NTU is equal to 1.0. What is the effectiveness of the heat exchanger?
Which of the following temperature profiles represents a counter flow exchanger with heat capacity rate of hot fluid (CH) greater than heat capacity rate of cold fluid (CC)
(A) 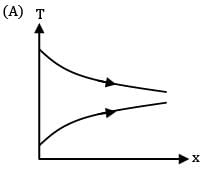
(B) 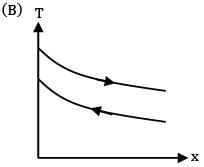
(C) 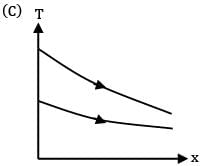
(D) 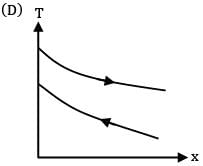
Match List-I with List-II and select the correct answer using the code given below the lists
List-I
a. Number of transfer units
b. Periodic flow heat exchangers
c. Phase change
d. Deposition on heat exchanger surface
List-II
1. Regenerators
2. Fouling factor
3. A measure of heat exchanger size
4. Condensers
Codes:

|
57 videos|77 docs|86 tests
|
|
57 videos|77 docs|86 tests
|