Reagents are substances that react with other substances to help identify or analyze them. | Card: 2 / 28 |
Fill in the blank: Sodium hydroxide and ammonium hydroxide are commonly used alkalis in analytical chemistry that react with ___ to help identify metal cations. | Card: 3 / 28 |
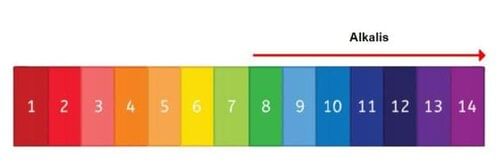
Riddle: I am a solution that can identify metal ions, both strong and weak. What am I? | Card: 5 / 28 |
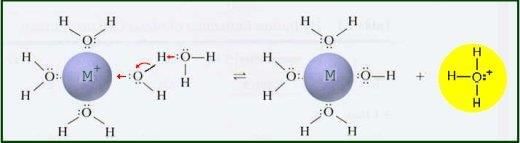
Alkalis like sodium hydroxide react with metallic salt solutions to form precipitates, which can indicate the presence of specific metal cations. | Card: 8 / 28 |
Fill in the blank: Sodium hydroxide reacts with ___ to produce a soluble salt and water. | Card: 9 / 28 |
 Alkalis can react with certain metals to produce hydrogen gas and a soluble metal hydroxide. | Card: 12 / 28 |
Fill in the blank: The reaction between an alkali and a metal oxide often leads to the formation of ___ and water. | Card: 13 / 28 |
 Unlock all Flashcards with EduRev Infinity Plan Starting from @ ₹99 only |
Multiple Choice: What do alkalis typically produce when they react with amphoteric oxides? A) Hydrogen gas B) Soluble salts C) Insoluble compounds D) None of the above | Card: 15 / 28 |
What is the primary difference between qualitative and quantitative analysis in analytical chemistry? | Card: 17 / 28 |
Qualitative vs Quantitative Analysis
| Card: 18 / 28 |
Fill in the blank: Sodium hydroxide and ammonium hydroxide are the most commonly used ___ in analytical chemistry. | Card: 19 / 28 |
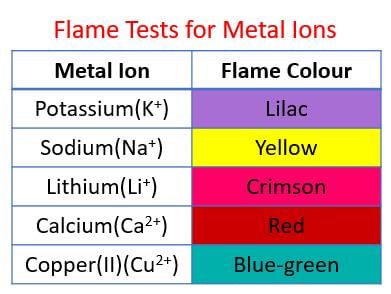
True or False: Ammonium hydroxide can be used to identify metal cations through characteristic tests. | Card: 21 / 28 |
Riddle: I am used in labs, I can help you find, whether a substance is present or left behind. What am I? | Card: 23 / 28 |
Fill in the blank: The characteristic tests of sodium hydroxide and ammonium hydroxide help in the identification of ___ . | Card: 25 / 28 |
Alkalis are crucial laboratory reagents.
| Card: 28 / 28 |