Rusting requires moisture and air.
| Card: 2 / 50 |
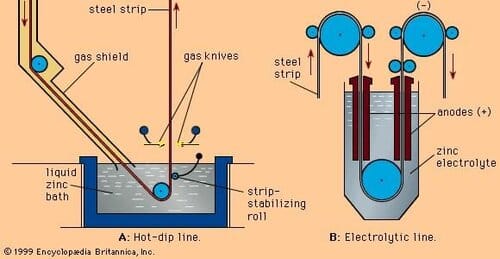
The process of protecting iron by coating it with a more electropositive metal like zinc is called ___ . | Card: 3 / 50 |
True or False: Corrosion of metals can be completely prevented using electroplating alone. | Card: 5 / 50 |
False. While electroplating provides a barrier protection, it does not guarantee complete prevention of corrosion under all conditions. | Card: 6 / 50 |
Fill in the blank: The most easily oxidized and electropositive metals are found at the ___ of the activity series. | Card: 7 / 50 |
Riddle: I protect metals from rust and corrosion, a layer of me keeps the air away. What am I? | Card: 9 / 50 |
What distinguishes alkaline earth metals from alkali metals in terms of reactivity? | Card: 11 / 50 |
Alkaline earth metals are less reactive than alkali metals due to their higher ionization energies and stronger metallic bonds. | Card: 12 / 50 |
Describe the general physical properties that differentiate metals from non-metals. | Card: 13 / 50 |
Metals differ from non-metals in key properties.
| Card: 14 / 50 |
Name the method used to prevent corrosion by ensuring the metal does not come into contact with moisture or air. | Card: 17 / 50 |
Riddle: I can displace metals from their salt solutions, and I'm known for being very reactive. Who am I? | Card: 19 / 50 |
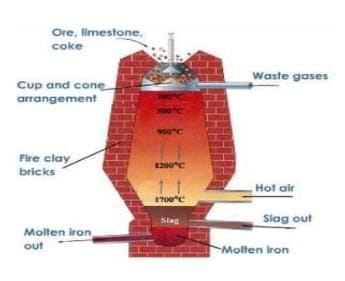
Metallurgy is essential for metal extraction.
| Card: 22 / 50 |
Fill in the blank: Ores are those minerals from which metals are extracted commercially at a comparatively low cost and with minimum ___. | Card: 23 / 50 |
True or False: Gangue refers to the valuable minerals that are extracted during the metallurgy process. | Card: 25 / 50 |
 Unlock all Flashcards with EduRev Infinity Plan Starting from @ ₹99 only |
 False. Gangue refers to the earthy impurities, including silica and mud, associated with the ore. | Card: 26 / 50 |
A flux is a substance added to the charge in a furnace to remove the gangue during the extraction process. | Card: 28 / 50 |
Riddle: I am added to ores to help remove the unwanted parts, making extraction easier and smarter. What am I? | Card: 29 / 50 |
Fill in the blank: The fusible product formed when flux reacts with impurities during the extraction of metals is called ___. | Card: 31 / 50 |
Smelting is the process of reducing the roasted oxide ore and removing the gangue with the help of an appropriate flux added with the ore. | Card: 34 / 50 |
Crushing and grinding enhance extraction.
| Card: 36 / 50 |
The concentration of ores involves separating the dense ore particles from the gangue using a ___ technique. | Card: 37 / 50 |
 False. Magnetic separation is specifically effective for ores that contain magnetic particles. | Card: 40 / 50 |
In froth flotation, the ore is wetted by oil and separates from the gangue in the form of ___ during agitation. | Card: 41 / 50 |
Riddle: I separate metals from their ores, but I'm not a magician. What process am I? | Card: 45 / 50 |
Fill in the blank: The process of using heat to remove impurities from metals is called ___. | Card: 47 / 50 |
Electrolytic refining is used to purify impure metals through electrolysis, which allows for the separation of pure metal from impurities. | Card: 50 / 50 |