Isomerism refers to compounds that have the same molecular formula but ___ properties. | Card: 1 / 50 |
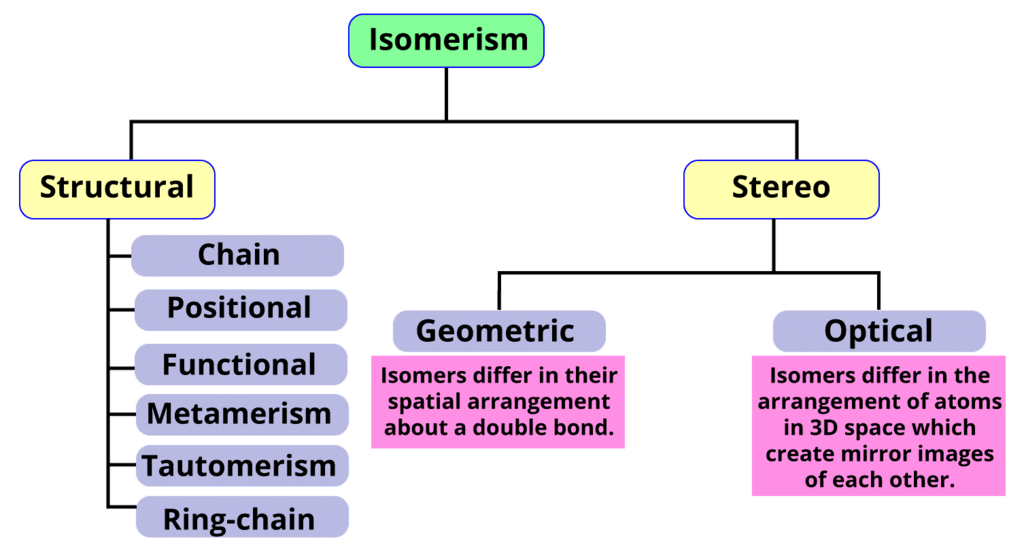
True or False: Structural isomerism involves molecules with the same connectivity of atoms. | Card: 3 / 50 |
Structural isomers differ in atom connectivity.
| Card: 8 / 50 |
Fill in the blank: Tautomeric isomers exist in equilibrium due to the migration of a ___ or a ___ between different forms. | Card: 9 / 50 |
Chain isomerism involves compounds with the same molecular formula but differing in ___. | Card: 11 / 50 |
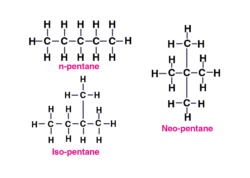
C5H12 has three isomers: n-pentane, isopentane, and neopentane, formed by varying the arrangement of carbon chains. | Card: 14 / 50 |
Position isomers differ from chain isomers in that they have the same molecular formula but differ in the position of ___ and ___ within the molecule. | Card: 15 / 50 |
True or False: The compounds 1-chloropropane and 2-chloropropane are examples of chain isomers. | Card: 17 / 50 |
What is the significance of total unsaturation in determining the structural isomers of a compound such as C₄H₆? | Card: 19 / 50 |
Total unsaturation affects structural isomers.
| Card: 20 / 50 |
Fill in the blank: The total number of structural isomers, both cyclic and acyclic, for C₄H₆ is ___. | Card: 21 / 50 |
Functional isomers differ in functional groups.
| Card: 24 / 50 |
True or False: Primary, secondary, and tertiary amines can have the same molecular formula but different structural formulas. | Card: 25 / 50 |
 Unlock all Flashcards with EduRev Infinity Plan Starting from @ ₹99 only |
True. Primary, secondary, and tertiary amines can indeed have the same molecular formula but differ in their structural arrangements. | Card: 26 / 50 |
What types of amides can be formed from the molecular formula C3H7NO, and how many structural isomers are possible for each type? | Card: 27 / 50 |
From C3H7NO, one primary amide, two secondary amides (total 4 structural isomers), and one tertiary amide can be formed. | Card: 28 / 50 |
False. Tertiary amides cannot form hydrogen bonds due to the lack of a hydrogen atom attached to the nitrogen. | Card: 30 / 50 |
What is the total number of structural isomers for the compound with the formula C6H12? | Card: 31 / 50 |
There are a total of 13 structural isomers for the compound with the formula C6H12. | Card: 32 / 50 |
True or False: A cumulated diene can be formed from a compound with the formula C5H8. | Card: 33 / 50 |
Metamerism is a type of isomerism characterized by compounds that have the same molecular formula but differ in the nature of the ___ groups attached to a polyvalent atom. | Card: 35 / 50 |
True or False: Tautomerism involves compounds that have the same molecular formula and arise from the oscillation of an atom, usually ___ . | Card: 37 / 50 |
What role does intramolecular hydrogen bonding play in the stability of certain compounds? | Card: 39 / 50 |
Intramolecular hydrogen bonding leads to the formation of a stable six-membered ring, which significantly enhances the stability of the compound. | Card: 40 / 50 |
Enol content in a compound is influenced by the stability of which intermediate? | Card: 41 / 50 |
Enol content depends on carbanion stability.
| Card: 42 / 50 |
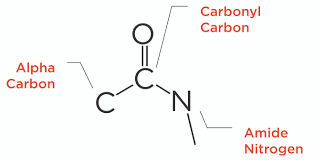
Amides and amines differ primarily in their functional groups, where amides contain a ___ group attached to a nitrogen atom. | Card: 43 / 50 |
True or False: Azo compounds are typically more stable than hydrazones due to the presence of extended conjugation within their structure. | Card: 45 / 50 |
What distinguishes alkenes from cycloalkanes in terms of their structural representation? | Card: 49 / 50 |
Alkenes have a double bond between carbon atoms, while cycloalkanes consist of carbon atoms arranged in a ring structure without any double bonds. | Card: 50 / 50 |