The general formula for carbonyl compounds is ___ where n represents the number of carbon atoms. | Card: 3 / 44 |
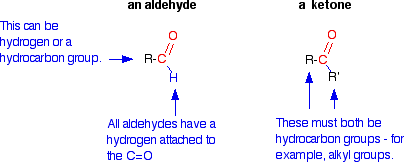
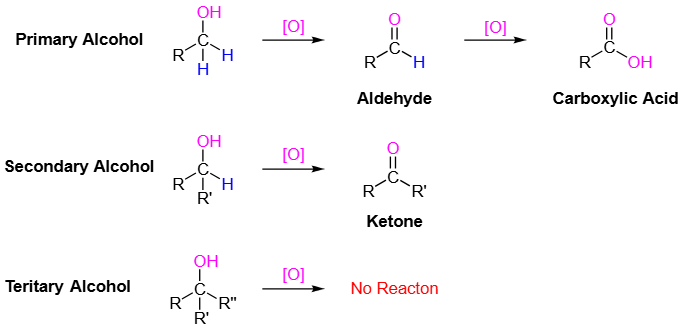
Aldehydes have the carbonyl group bonded to at least one hydrogen atom, while ketones have the carbonyl group bonded between two carbon atoms. | Card: 6 / 44 |
The IUPAC naming convention for aldehydes ends with the suffix ___ and for ketones with ___ . | Card: 9 / 44 |
True or False: Cyclic ketones are numbered starting from the carbon atom furthest from the carbonyl group. | Card: 11 / 44 |
Fill in the blank: The carbonyl group exhibits ___ hybridization, leading to a trigonal coplanar structure. | Card: 13 / 44 |
The carbonyl oxygen acts like a Lewis base due to its two unshared pairs of electrons. | Card: 16 / 44 |
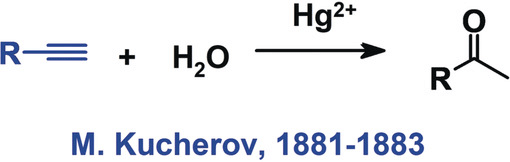
It adds water to the carbon-carbon triple bond, forming a carbonyl compound (ketone or aldehyde). | Card: 22 / 44 |
 Unlock all Flashcards with EduRev Infinity Plan Starting from @ ₹99 only |
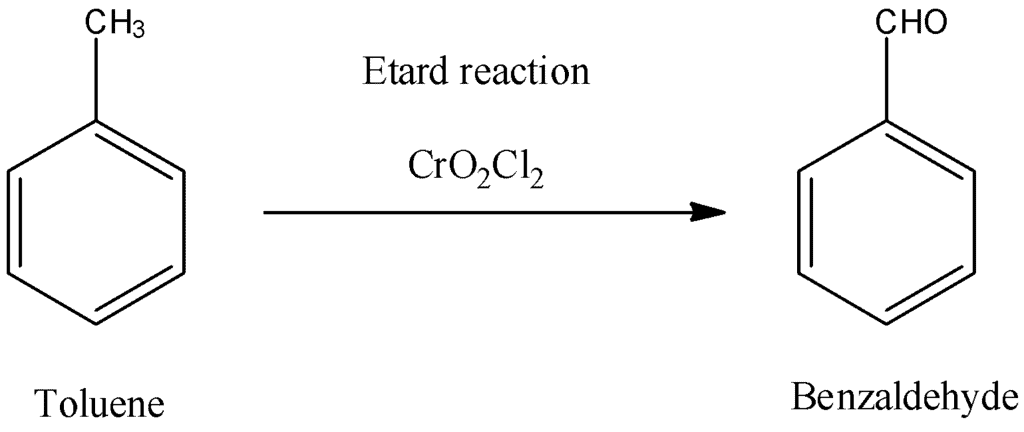
The Etard reaction uses ___ as an oxidizing agent to convert toluene into benzaldehyde. | Card: 23 / 44 |
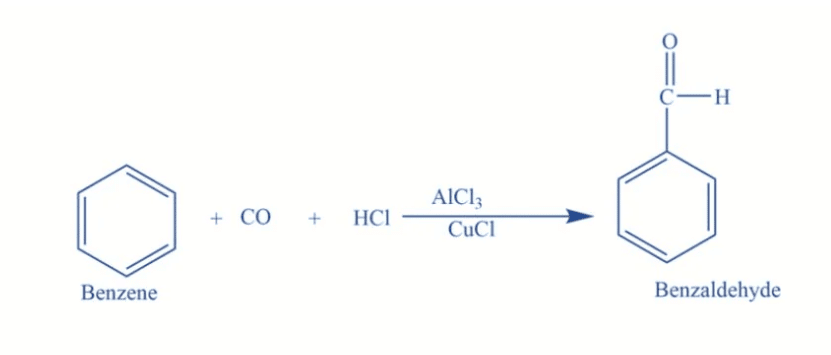
The Gattermann-Koch reaction introduces a formyl group (-CHO) to an aromatic compound using ___ and ___. | Card: 25 / 44 |
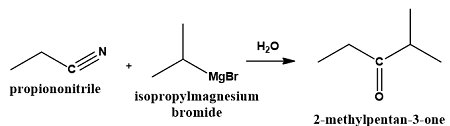
What is produced when a nitrile is treated with a Grignard reagent followed by hydrolysis? | Card: 27 / 44 |
Fill in the blank: The carbonyl carbon in a carbonyl group has ___ sigma bonds and one ___ bond. | Card: 29 / 44 |
The hydroboration-oxidation reaction can be used to convert alkenes into ___ if the starting material is an alkene. | Card: 33 / 44 |
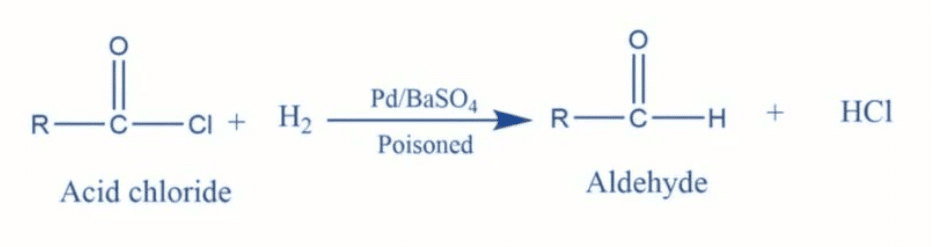
What are common reducing agents used in the reduction of acyl halides, esters, and nitriles to form aldehydes? | Card: 37 / 44 |
Fill in the blank: The polarization in the carbon-oxygen bond of carbonyl compounds is due to the higher electronegativity of ___ compared to ___. | Card: 39 / 44 |