Aromaticity - General Organic Chemistry | Organic Chemistry PDF Download
The factor which explains the extra stability of cyclic molecule is called ‘aromaticity’. In aromatic compound, resonance increases the stability of the molecule, therefore, energy is called resonance energy while in anti-aromatic compound, resonance decreases the stability of the compound, therefore, energy is called destabilization energy.
The following rule’s explain the concept of aromaticity:
1. Huckel theory 2.Crag’s rule
1. Huckel rule’s: For a compound to be aromatic, following conditions must be followed
(i) Compound must be cyclic & planar or nearly planar.
(ii) It must show resonance & delocalization.
(iii) It obeys [4n + 2] π electron rule: n is called Huckel’s number or aromaticity index
Aromaticity index [n] = 0, 1, 2, 3….

Homoaromatic molecules are molecules which are form
by passing one or more than one sp3 carbon atom in the ring
through delocalization of π-electron and it become aromatic in nature.
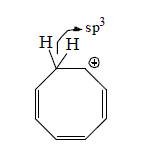
Stability order: Aromatic > Homoaromatic > Nonaromatic > Antiaromatic.
Number of π -electron in cyclic and a cyclic conjugated system
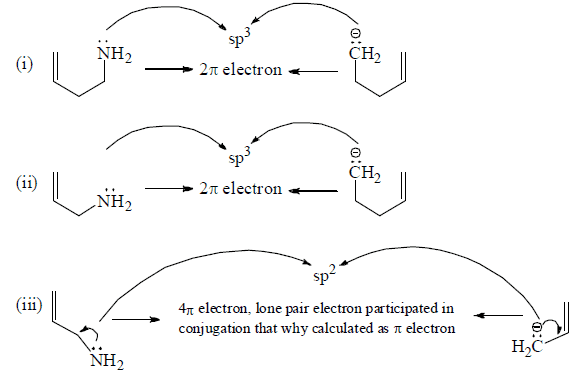
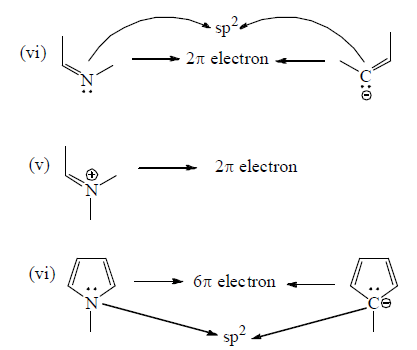
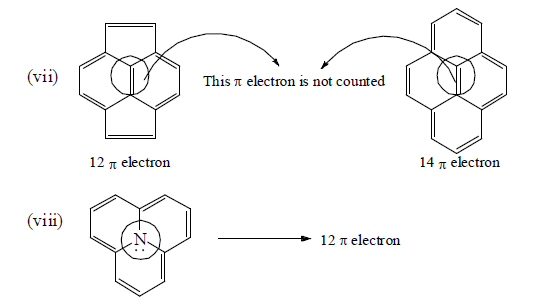
How To Apply Aromaticity Rule:
1. Aromaticity in three member ring.

2. Aromaticity in four membered ring.

3. Aromaticity in five membered ring:
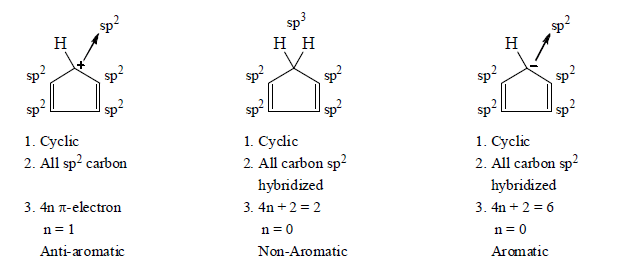
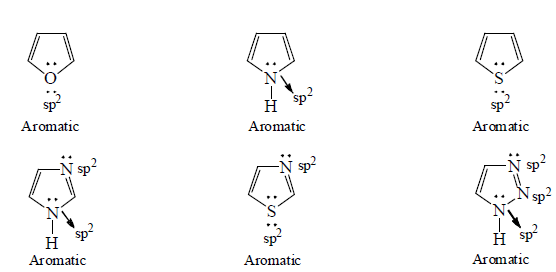
4. Aromaticity in six membered ring:

5. Aromaticity in seven membered ring:
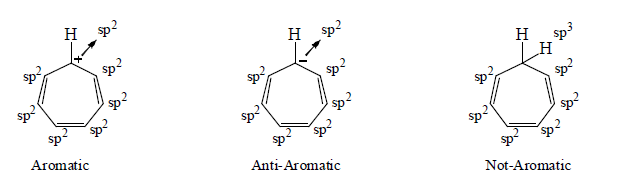
6. Aromaticity in eight membered ring:
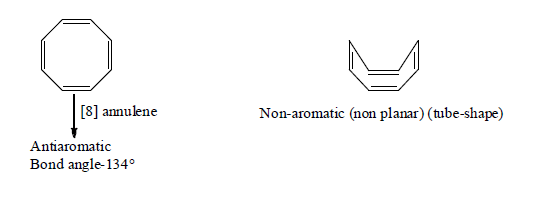
7. Non-Benzenoid aromatics Compounds: Annulenes
The monocyclic conjugated system of the general formula (CH)2n have been called annulenes, the number of carbon atoms inthe ring being denoted by a prefixed of breackts. In this system of nomenclature, benzene is [6] annulene and COT is [8] annulene. Since the carbon atoms occur as donded pair, an annulene must have an even of carbons. The annulenes can be grouped into two series.
(a) Those in which m is odd having (4n + 2) π -electrons.
(b) Those in which m is even having (4n) π -electrons.
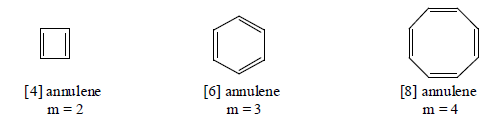
Here we will discuss only those anulenes which have (4n + 2) π-electrons and aromatic in character.
ANNULENE
Annulene can exist in three isomeric forms: di-trans--[10] annulene (1), all-cis-[10] annulene (2) and mono-trans-[10] annulesn (3).
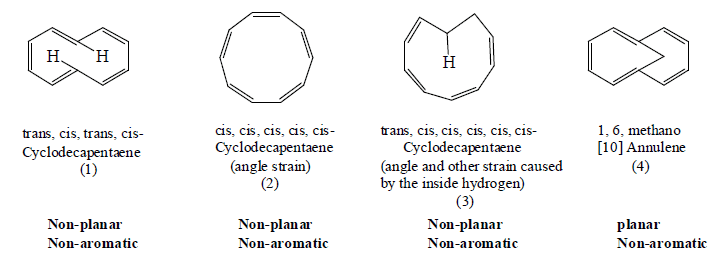
Althrough these three isomers have (4n + 2) π -electrons, they are not aromatic in character. All the three isomers are non-planar. In di-trans-[10] annulene two internal hydrogends prevent the molecule from assuming a planar configuration. Replacing two interacting hydrogens in di-trans-[10] annulene gives 1, 6-methano [10] annulene (4)
in which the transannular interaction are removed. The NMR spectrum of (4) show an AB’BB’ system for the ring protons at low field [δ - 0.52] . This indicates that compound (4) is diatropic molecule and hence is aromatic.
[14] ANNULENE
[14] Annulene (cyclotetradecaheptaene) (5) follows the Huckel (4n + 2) π rule; but models and scale drawing indicate that the four inner hydrogens overlap so that a planar molecule is unlike. X-ray studies show that althrough all carbon-carbon bonds are of equal length but molecule is not planar. [14] Annulene is a diatropic molecule. Its inner protons appears at δ0.00 and outer protons appear at δ7.6 . Thus [14] annulene is aromatic althrough it is not planar.
Althrough [14] annulene is aromatic, the overlapping of the van der Waals radii of the four inner hydrogens reduces it aromaticity. thus, [14] annulene is not very stable. It is completely destroyed by light and air within 24 hrs. It does not give substitution reactions.
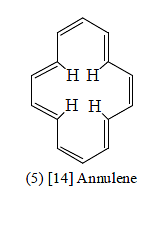
A variety of 1.6 : 8, 13 bridged [14] annulenes have been prepared with both carbon and heteroatom bridges. syn- 1, 6:8, 13-Bisoxio [14] annulene (6) is a red crystalline, thermally stable aromatic comopund. Syn-1, 6-methano-8, 13-oxiodo (7) is also diatropic in nature.

[18] Annulene (Cyclooctadecanonaene)
[18] Annulene (8) confirms the Huckel rule and, provided the molecule can assume nearly a planar conformation, should exhibit aromaticity. Molecular models and scale drawing indicate that the six inner hydrogens overlap slightly, but that this overlap is not so extreme as to exclude a reasonably planar structure.
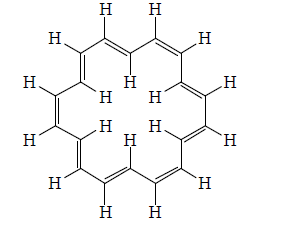
A three dimensional X-ray crystallographic analysis of [18] annulene showered that it was a near planar molecule with non-alternate bonds of two type six cisolid (141.9 pm) and twelve transoid (138.2 pm). The resonance energy of the comopund is 37 kal/mole. The NMR Spectrum of the compuond showred that it is a diatroic molecule: the twelve outer protons appear at about δ9.0 and the six inter protons at δ-3.0.
It is stable in crystalline state but decomposes in solution at room temperature. [20]
[20]Annulene:
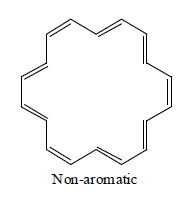
[22] Annulene
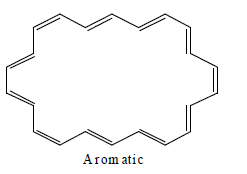
Annulene are found to be non-aromatic as per Huckel rule because their planarity are not justified therefore they are considered to be non-aromatic.
Bicyclic & Polycyclic Aromatic System
Bicyclic Aromatic System
Tricyclic
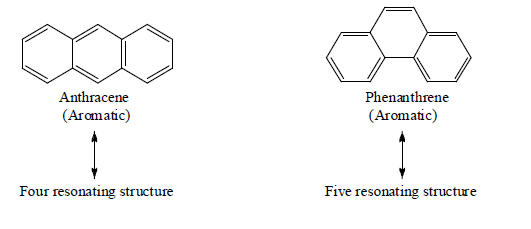
Tetracyclic
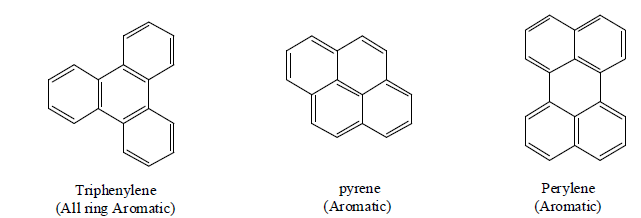
2. CRAIG’S RULE:
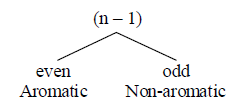
According to Huckel’s rule [4n+2]π , the molecule C and D is aromatic however A and B should be antiaromatic[4nπ,all atom is sp2 hybridized and cyclic] but in actual practise, the compound A and B is non aromatic, the question arises how A and B become nonaromatic?

Crag’s Rule:
Condition:
(1) Cyclic and planar (2) Non benzenoid and polynuclear benzenoid (3) Either aromatic or non aromatic. Where n is total number of π electron
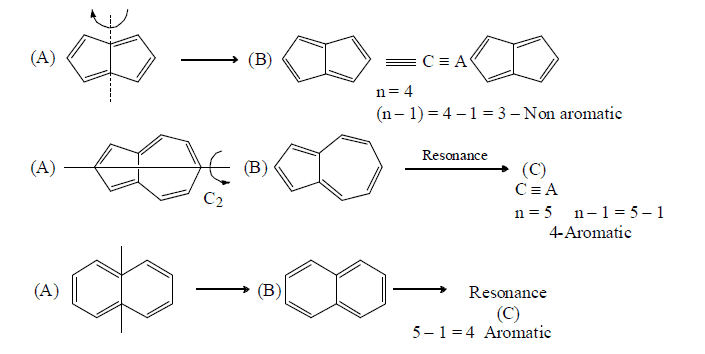
Annulation effect: In aromaticity the delocalisation should be restricted to single ring in polycyclic compounds. As the delocalisation passes from one ring to another it loses its extent of aromaticity. This is called annulation effect.
|
35 videos|92 docs|46 tests
|
FAQs on Aromaticity - General Organic Chemistry - Organic Chemistry
| 1. What is aromaticity in organic chemistry? |  |
| 2. How can we determine if a molecule is aromatic or not? |  |
| 3. What is the difference between aromatic and antiaromatic compounds? |  |
| 4. What are some examples of aromatic compounds? |  |
| 5. Can non-aromatic compounds exhibit aromaticity? |  |

|
Explore Courses for Chemistry exam
|

|


















