Mechanical Engineering Exam > Mechanical Engineering Notes > Mechanical Engineering SSC JE (Technical) > First Law of Thermodynamics
First Law of Thermodynamics | Mechanical Engineering SSC JE (Technical) PDF Download
FIRST LAW OF THERMODYNAMICS
- The First Law of Thermodynamics states that heat is a form of energy, and thermodynamic processes are therefore subject to the principle of conservation of energy. This means that heat energy cannot be created or destroyed. It can, however, be transferred from one location to another and converted to and from other forms of energy.
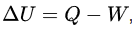
where ΔU denotes the change in the internal energy of a closed system,
Q denotes the quantity of energy supplied to the system as heat, and
W denotes the amount of thermodynamic work done by the system on its surroundings.
System undergoing change of states
Qnet = ΔE +W
(Q2 + Q3 – Q1) = ( ΔE +W2 +W3 - W 1- W4 )

- Energy is a point function and a property of the system. Energy is an extensive property while specific energy is an intensive property.
- The internal energy depends only on temperature, The enthalpy also depends only on temperature for an ideal gas.
Heat transfer at constant volume
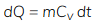
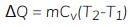
Cv = speecific heat at constant volume
- Heat transfer at constant volume increases the internal energy of the system.
Heat transfer at constant pressure
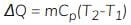
Cp = Specific heat at constant pressure
- Heat transfer at constant pressure increases the enthalpy of the system.
Heat transfer for an isolated system
dQ = 0, dw = 0 & dE = 0
- Energy of an isolated system is always constant.
- PMM-1 There can be no machine which would continuously supply mechanical work without absorbing some other form of energy simultaneously. Such a fictitions machine is called perpetual motion machine of first kind (PMM–1)
- The first law states the principle of conservation of energy.
- A PMM–1 is the impossible.
The document First Law of Thermodynamics | Mechanical Engineering SSC JE (Technical) is a part of the Mechanical Engineering Course Mechanical Engineering SSC JE (Technical).
All you need of Mechanical Engineering at this link: Mechanical Engineering
|
5 videos|103 docs|59 tests
|
FAQs on First Law of Thermodynamics - Mechanical Engineering SSC JE (Technical)
| 1. What is the First Law of Thermodynamics in mechanical engineering? |  |
Ans. The First Law of Thermodynamics, also known as the law of energy conservation, states that energy cannot be created or destroyed in an isolated system. It can only change its form from one type to another or be transferred from one location to another.
| 2. How does the First Law of Thermodynamics apply to mechanical engineering? |  |
Ans. In mechanical engineering, the First Law of Thermodynamics is crucial for understanding and analyzing energy conversions and transfers in various systems such as engines, turbines, and heat exchangers. It helps engineers determine the efficiency and performance of these systems by balancing the energy inputs and outputs.
| 3. What are the key components of the First Law of Thermodynamics? |  |
Ans. The key components of the First Law of Thermodynamics are the internal energy of a system, the work done on or by the system, and the heat transferred to or from the system. These components are interconnected and can be expressed through the equation: ΔU = Q - W, where ΔU is the change in internal energy, Q is the heat added to the system, and W is the work done by the system.
| 4. How can the First Law of Thermodynamics be applied to an engine? |  |
Ans. The First Law of Thermodynamics can be applied to an engine by considering the energy input (fuel combustion) and energy output (mechanical work). By analyzing the heat transfer, work done, and changes in internal energy, engineers can calculate the efficiency of the engine and make improvements to enhance its performance.
| 5. What are some practical applications of the First Law of Thermodynamics in mechanical engineering? |  |
Ans. The First Law of Thermodynamics finds various practical applications in mechanical engineering, such as analyzing the performance of power plants, designing efficient HVAC systems, optimizing combustion processes in engines, and assessing the energy utilization in industrial processes. It provides engineers with a fundamental understanding of energy conservation and helps them make informed decisions to improve energy efficiency.
|
5 videos|103 docs|59 tests
|
Download as PDF

|
Explore Courses for Mechanical Engineering exam
|

|
Signup for Free!
Signup to see your scores go up within 7 days! Learn & Practice with 1000+ FREE Notes, Videos & Tests.
Related Searches


















