Integrated Rate Equations: Zero, First & Second Order Reactions
What are Integrated Rate Equations?
Integrated rate equations are mathematical expressions that relate the concentrations of reactants and/or products to time for a chemical reaction.
These equations are obtained by integrating the rate law, which describes how the rate of a reaction depends on the concentrations of reactants.
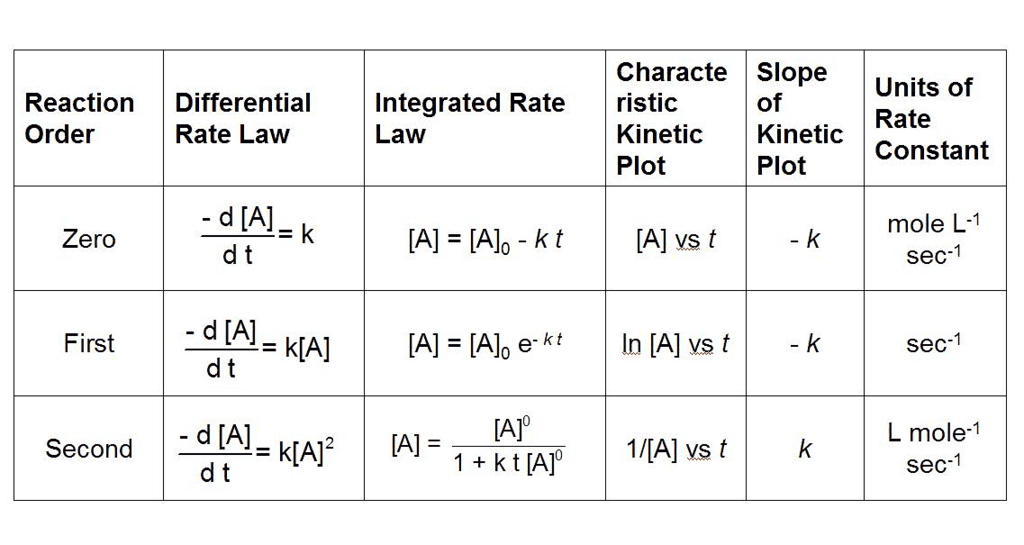
- The integrated rate equations provide a direct relation between concentration and time, allowing determination of reaction kinetics and calculation of the rate constant.
- The form of the integrated rate equation depends on the order of the reaction, which must be determined experimentally.
- The three most common reaction orders treated in Class 12 are zero order, first order and second order. The integrated forms for each are given below.
Zero Order Reactions
Rate law and integrated form
The rate law for a zero order reaction is
rate = -d[A]/dt = k
Integration of the rate law gives the concentration as a function of time.
d[A]/dt = -k
Integrate both sides with limits [A] = [A]0 at t = 0 to [A] at time t:
∫[A]0[A]d[A] = -k ∫0tdt
Which yields
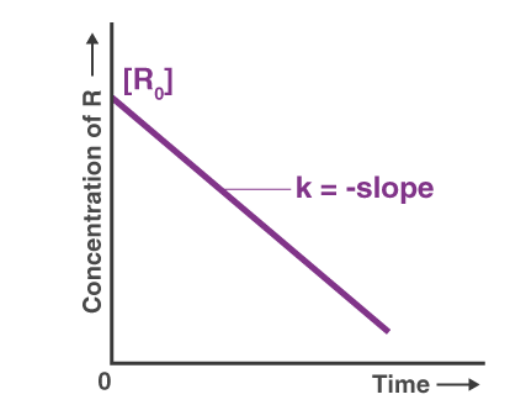
[A] = [A]0 - kt
The slope of the plot [A] against t is -k and is therefore linear.
Half-life (t1/2) of a zero order reaction
Half-life is the time in which the concentration of the reactant falls to half its initial value.
Set [A] = [A]0/2 at t = t1/2 in the integrated equation:
[A]0/2 = [A]0 - k t1/2
Rearrange to obtain
t1/2 = [A]0 / (2k)
Thus, for a zero order reaction, half-life is directly proportional to the initial concentration of the reactant.
Remarks and examples
Zero-order reactions have rates independent of the concentration of reactant; their rate depends only on external factors such as light or catalyst surface. Many photochemical reactions are approximately zero order under steady illumination. Additional examples include decomposition of HI on a gold surface and decomposition of NH3 on tungsten.
Examples
- Photochemical reactions and photosynthesis (under certain conditions).
- Reactions initiated on a catalyst surface, e.g., CH4 + Cl2 under strong photochemical conditions (chain initiation under light) - see reaction scheme below.
Cl-Cl (photolysis) → 2 Cl•
CH4 + Cl• → CH3Cl + H•
First Order Reactions
Rate law and integrated form
The rate law for a first order reaction is
rate = -d[A]/dt = k[A]
Separate variables and integrate with the limits [A] = [A]0 at t = 0 to [A] at time t:
d[A]/[A] = -k dt
Integrate both sides:
∫[A]0[A]d[A]/[A] = -k ∫0tdt
Which gives
ln[A] - ln[A]0 = -kt
Rearranged:
ln([A]0/[A]) = kt
Half-life (t1/2) of a first order reaction
Set [A] = [A]0/2 at t = t1/2 in the integrated equation:
ln([A]0/[A]0/2) = k t1/2
Therefore
ln 2 = k t1/2
t1/2 = ln 2 / k
Hence the half-life of a first order reaction is independent of the initial concentration.
Concentration after n half-lives
After n half-lives, the concentration remaining is
[A] = [A]0 / 2n
Average life (mean lifetime)
For a first order process the average life or mean lifetime τ is the reciprocal of the rate constant:
τ = 1 / k
Features of a first order reaction
- A first order reaction must follow the form rate = k[A] at all times. If concentration-time data are truly first order, calculations of k from different time intervals give approximately the same value.
- The half-life is independent of the initial concentration of reactant.
- The concentration of reactant in a first order reaction decreases exponentially with time; a plot of ln[A] (or log10[A]) versus time is linear with slope -k.
Examples of first order reactions
- Radioactive disintegration.
- Decomposition of PCl5(g) → PCl3(g) + Cl2(g).
- Decomposition of H2O2 → H2O + 1/2 O2 (in some conditions).
- Decomposition of NH4NO2 → N2 + 2 H2O.
Determination of k from two concentration measurements
If [A]1 and [A]2 are the concentrations at times t1 and t2 respectively, then from ln([A]0/[A]) = kt we can write:
ln([A]1) = ln([A]0) - k t1
ln([A]2) = ln([A]0) - k t2
Subtracting, we obtain
k = ln([A]1/[A]2) / (t2 - t1)
Worked example
| [NO] (mol L-1) | [H2] (mol L-1) | Rate (mol L-1 s-1) | |
|---|---|---|---|
| 1. | 5 × 10-3 | 2.5 × 10-3 | 3 × 10-5 |
| 2. | 15 × 10-3 | 2.5 × 10-3 | 9 × 10-5 |
| 3. | 15 × 10-3 | 10 × 10-3 | 3.6 × 10-4 |
Problem:
(a) Calculate the order of reaction.
(b) Find the rate constant.
(c) Find the initial rate if [NO] = [H2] = 8.0 × 10-3 M.
Solution:
Assume the rate law is rate = k [NO]x [H2]y.
Compare experiments 1 and 2: [H2] is constant and [NO] is tripled from 5 × 10-3 to 15 × 10-3 while rate increases from 3 × 10-5 to 9 × 10-5, which is also a factor of 3.
Therefore rate ∝ [NO] ⇒ x = 1.
Compare experiments 2 and 3: [NO] is constant and [H2] increases fourfold from 2.5 × 10-3 to 10 × 10-3 while rate increases from 9 × 10-5 to 3.6 × 10-4, also fourfold.
Therefore rate ∝ [H2] ⇒ y = 1.
Hence the rate law is
rate = k [NO] [H2]
Use any experimental set to find k. From experiment 1:
k = rate / ([NO][H2])
k = (3 × 10-5) / ((5 × 10-3) (2.5 × 10-3))
k = (3 × 10-5) / (12.5 × 10-6)
k = 2.4 (units: L mol-1 s-1)
Initial rate when [NO] = [H2] = 8.0 × 10-3 M is
rate = k [NO] [H2]
rate = 2.4 × (8.0 × 10-3) × (8.0 × 10-3)
rate = 2.4 × 64 × 10-6
rate = 153.6 × 10-6 = 1.536 × 10-4 mol L-1 s-1.
Second Order Reactions
Case I: rate = k[A]2
When the rate law is
rate = -d[A]/dt = k [A]2
Separate variables and integrate with limits [A] = [A]0 at t = 0 to [A] at time t:
d[A] / [A]2 = -k dt
Integrate both sides:
∫[A]0[A]d[A] / [A]2 = -k ∫0tdt
Which gives
-1/[A] + 1/[A]0 = -kt
Rearrange to obtain the familiar linear form:
1/[A] = kt + 1/[A]0
Half-life (t1/2) of a second order reaction (Case I)
Set [A] = [A]0/2 at t = t1/2 in 1/[A] = kt + 1/[A]0:
1/([A]0/2) = k t1/2 + 1/[A]0
Which is
2/[A]0 = k t1/2 + 1/[A]0
Therefore
t1/2 = 1 / (k [A]0)
Thus for a second order reaction (rate ∝ [A]2) the half-life is inversely proportional to the initial concentration.
Examples (Case I)
- Alkaline hydrolysis of esters (under suitable conditions).
- Some self-condensation and Cannizzaro-type reactions where rate ∝ [reactant]2.
Case II: second order with two different reactants A + B → products
Consider the reaction A + B → products with rate = k [A][B]. Let initial concentrations be [A]0 = a and [B]0 = b and let x be the amount reacted at time t. Then [A] = a - x and [B] = b - x and
dx/dt = k (a - x)(b - x)
Integrate by separating variables and using partial fractions; with a ≠ b the integrated form is
ln( (a [B]) / (b [A]) ) = (b - a) k t
where [A] and [B] are concentrations at time t. This expression can be rearranged to solve for [A] or k as required.
If a = b (equal initial concentrations), this reduces to the Case I result 1/[A] = kt + 1/[A]0.
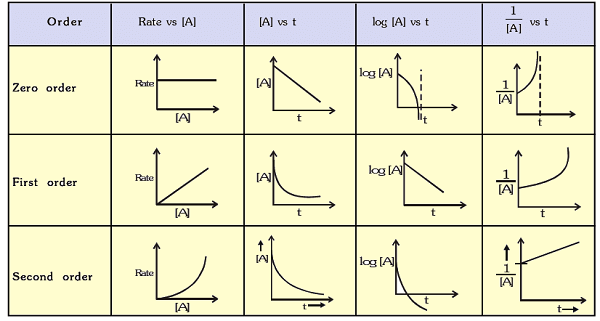
Graphs of Zero, First and Second Order Reactions
Each order has a characteristic linear plot that is used to identify the order and determine k.
- Zero order: plot of [A] vs t is linear with slope -k.
- First order: plot of ln[A] (or log10[A]) vs t is linear with slope -k.
- Second order (for rate = k[A]2): plot of 1/[A] vs t is linear with slope k.
Summary
This chapter has presented the integrated rate equations for zero, first and second order reactions, derived half-life expressions wherever applicable, described characteristic plots for identifying reaction order and illustrated how to determine the rate constant from experimental data. Worked examples and typical reaction examples have been included to make the methods clear and exam-relevant.
FAQs on Integrated Rate Equations: Zero, First & Second Order Reactions
| 1. What is the difference between zero-order, first-order, and second-order reactions? |  |
| 2. How is the half-life of a zero-order reaction related to the initial concentration of the reactant? |  |
| 3. How can you determine the rate constant of a first-order reaction from experimental data? |  |
| 4. What factors can influence the rate of a second-order reaction? |  |
| 5. How do integrated rate equations help in understanding the kinetics of chemical reactions? |  |







