NEET Exam > NEET Notes > Chemistry Class 11 > Mind Map: Organic Chemistry: Some basic Principles and Techniques
Mind Map: Organic Chemistry: Some basic Principles and Techniques
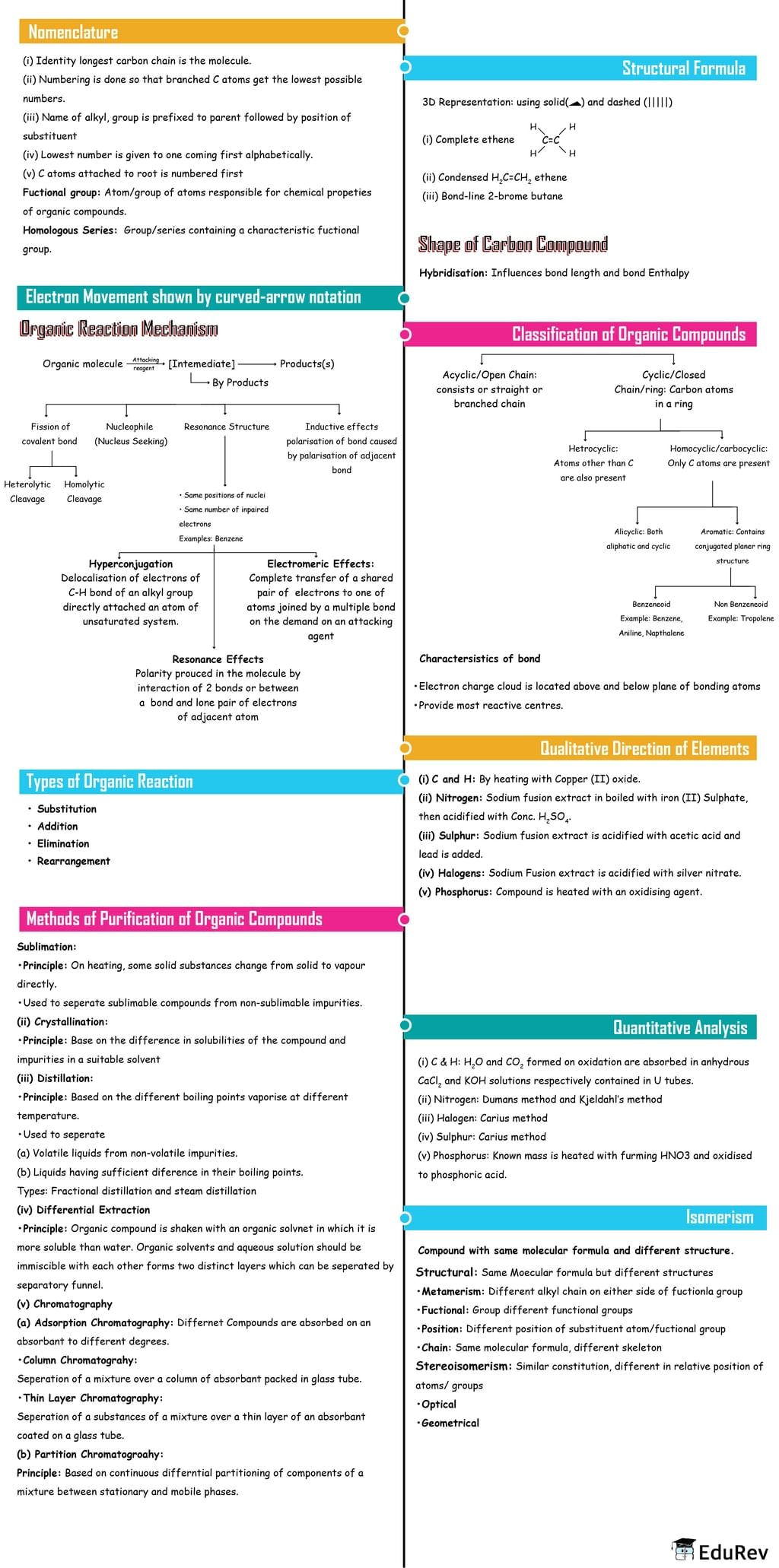
The document Mind Map: Organic Chemistry: Some basic Principles and Techniques is a part of the NEET Course Chemistry Class 11.
All you need of NEET at this link: NEET
FAQs on Mind Map: Organic Chemistry: Some basic Principles and Techniques
| 1. What are the main principles of organic chemistry I need to know for NEET? |  |
Ans. Organic chemistry fundamentals include understanding covalent bonding, hybridisation, molecular structure, and reaction mechanisms. Students must grasp how carbon forms stable bonds and how functional groups determine molecular properties. These basic principles form the foundation for studying nomenclature, isomerism, and organic reactions essential for NEET success.
| 2. How do I identify and name different types of isomers in organic compounds? |  |
Ans. Isomers are compounds with identical molecular formulas but different structural arrangements. Structural isomers differ in carbon skeleton or functional group position, while stereoisomers have the same connectivity but different spatial arrangements. Mastering isomerism classification helps predict compound properties and reactions crucial for NEET problem-solving.
| 3. What's the difference between sigma and pi bonds, and why does it matter? |  |
Ans. Sigma bonds form through direct head-on overlap of orbitals and allow free rotation, while pi bonds form through lateral overlap and restrict rotation. Understanding these bond types explains molecular geometry, reactivity patterns, and electron behaviour in organic compounds-essential for predicting how molecules interact during chemical reactions.
| 4. Why do some organic compounds show resonance, and how does it affect their stability? |  |
Ans. Resonance occurs when a molecule can be represented by multiple Lewis structures with delocalised electrons, increasing stability through electron distribution. Resonance stabilisation explains why certain compounds are more chemically stable and less reactive. Recognising resonance structures helps predict reaction sites and product formation in organic synthesis pathways.
| 5. How should I approach learning the nomenclature system for organic compounds efficiently? |  |
Ans. IUPAC nomenclature follows systematic rules: identify the longest carbon chain, number from the end nearest to functional groups, and list substituents alphabetically. Regular practice with mind maps and flashcards helps students memorise priorities and naming conventions. Mastering nomenclature enables quick compound identification and effective communication in organic chemistry examinations.
Related Searches
practice quizzes, Sample Paper, Objective type Questions, ppt, Exam, Important questions, mock tests for examination, pdf , Summary, Extra Questions, Semester Notes, Mind Map: Organic Chemistry: Some basic Principles and Techniques, MCQs, past year papers, Previous Year Questions with Solutions, Viva Questions, Free, shortcuts and tricks, study material, Mind Map: Organic Chemistry: Some basic Principles and Techniques, Mind Map: Organic Chemistry: Some basic Principles and Techniques, video lectures;






