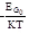Semiconductors - 1 | Electronic Devices - Electronics and Communication Engineering (ECE) PDF Download
Introduction
Electronic Devices: It is the branch of physics that deals with the emission and effects of electrons emission and effects of electrons and the use of electronic devices.
Electronic Circuits: Components are connected together to create an electronic circuit with a particular function. Active components are sometimes called devices.
 Semiconductor Materials
Semiconductor Materials
- The term conductor is applied to any material that will support a generous flow of charge when a voltage source of limited magnitude is applied across its terminals.
- An insulator is a material that offers a very low level of conductivity under pressure from an applied voltage source.
- A semiconductor, therefore, is a material that has a conductivity level somewhere between the extremes of an insulator and conductor.
- Band Theory: A bonding of atoms, strengthened by the sharing of electrons, is called covalent bonding. In the crystal, closely spaced energy levels form a band called as energy band. Each orbit has a separate energy band. A band of energy levels associated with valence shells is called as valence band. Electrons from other bands cannot be removed but electrons from valence band can be removed by supplying a little energy. The conduction band is generally empty. The valence band and conduction band are separated by a gap called forbidden energy gap.
- Compound Semiconductors: Such as Gallium Arsenide (GaAs), Cadmium Sulphide (CdS), Gallium Arsenide Phosphide (GaAsP), Gallium Nitride (GaN) are constructed by two or more semiconductor materials of different atomic structures are called compound semiconductors.
Intrinsic Semiconductors
- At 0oK, no free carriers are available, Si behaves as an insulator.
- At room temperature, a few covalent bonds will be broken by the thermal energy, electron‐hole pair generation as free carriers.
- Both electrons and holes are free to move, can contribute to current conduction

Extrinsic Semiconductor
- Extrinsic (doped) semiconductor = intrinsic semiconductor + impurities
- According to the species of impurities, the extrinsic semiconductor can be either n‐type or p‐type.
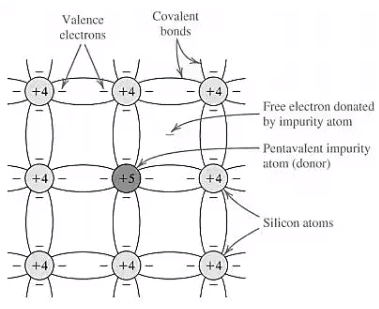
n‐type semiconductor
- The donor impurities have 5 valence electrons are added into silicon.
- P, As Sb, are commonly used as a donor.
- The Si atom is replaced by a donor atom.
- Donor ions are bounded in the lattice structure and thus donate free electrons without contributing holes.
- By adding donor atoms into an intrinsic semiconductor, the number of electrons increases (n > p) → n‐type semiconductor.
- Majority carrier: electron.
- Minority carrier: hole

p‐type semiconductor
- The acceptor impurity has 3 valence electron (Boron).
- Th Si atom is replaced by an acceptor atom.
- The boron lacks one valence electron. It leaves a vacancy in the bond structure.
- This vacancy can accept electron at the expense of creating a new vacancy.
- Acceptor creates a hole without contributing free electron.
- By adding acceptor into an intrinsic semiconductor, the number of holes increase (p > n) → p‐type semiconductor.
- Majority carrier: hole.
- Minority carrier: electron
Properties of Semiconductor Materials
Various materials are classified based on the width of forbidden energy gap. In metal, there is no forbidden gap and valence and conduction band are overlapped. In an insulator, the forbidden gap is very large up to 7eV while in semiconductors it is up to 1eV. The silicon and germanium are widely used semiconductors. Intrinsic materials are those semiconductor that has been carefully refined to reduce the impurities to a very level-essentially' as pure as can be made available through modern technology.
- The conductivity of an intrinsic semiconductor is very less. The properties like conductivity can be changed by adding an impurity to the intrinsic semiconductor. The process of adding impurity is called doping.
- A semiconductor doped with trivalent impurity atoms forms p-type material. It is called acceptor impurity with concentration NA atoms per unit volume.
- A semiconductor doped with pentavalent impurity atoms forms n-type material. It is called donor impurity with concentration ND atoms per unit volume.
- In p-type, holes are majority carriers and in n-type electrons are majority carriers.
- When a material is subjected to an electric field, electrons move in a particular direction with steady speed called drift speed and current drift current.
Negative Temperature Coefficient: Those parameters decreasing with the temperature have a negative temperature coefficient, e.g., an energy gap (Eg).
where, constant β0 = 2.2 × 10–4 (for Ge)
= 3.6 × 10–4 (for Si)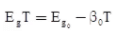
Mobility (μ), μ ∝ T–m
Positive Temperature Coefficient: Those parameters increasing with temperature have a positive temperature coefficient.
Important Terms
- Drift velocity Vd = μE
- Current density J = nq μE
- Conductivity σ = nq μ
- Concentration of free electrons per unit volume n = dU/AM
- Semiconductor conductivity σ = (nμn + pμp)q
- In intrinsic semiconductor, n = p = ni Hence, conductivity σi = ni (μn + μp)q Intrinsic concentration ni = A0T3e

- In extrinsic semiconductor, the conductivity is given by, For n-type, σn = (nnμn + pnμp)q For p-type, σn = (npμn + ppμp)q But in n-type pn < < nn ND = Concentration of donor impurity NA = Concentration of acceptor impurity np = Number of electrons (concentration) in p-type Pp = Number of holes (concentration) in p-type and nn ≅ ND while in p-type np < < pp and pp ≅ NA Hence, conductivity can be calculated as, σn = NDμnq and σp = NAμpq
- Mass-action law np = ni2
In n-type, nnpn = ni2 , hence 
In p-type, ppnp = ni2, hence 
|
21 docs|29 tests
|
FAQs on Semiconductors - 1 - Electronic Devices - Electronics and Communication Engineering (ECE)
| 1. What is a semiconductor? |  |
| 2. How are semiconductors used in electronics? |  |
| 3. What are the properties of semiconductors? |  |
| 4. What is the difference between a conductor and a semiconductor? |  |
| 5. How are semiconductors manufactured? |  |

|
Explore Courses for Electronics and Communication Engineering (ECE) exam
|

|
 Semiconductor Materials
Semiconductor Materials