Chapter Notes: Structure of the Atom
Atomic Structure
The structure of an atom consists of protons, neutrons, and electrons. Protons and neutrons each have a mass of one unit, while the mass of an electron is so small that it is often ignored. These fundamental components determine the mass and charge of the atom.
Atomic structure is about how these subatomic particles-protons, neutrons, and electrons-are arranged within an atom, which affects its composition and behaviour.
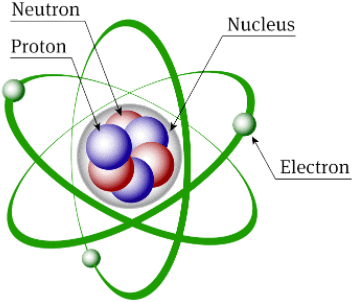 Structure of Atom
Structure of Atom- John Dalton believed that the atom cannot be divided.
- In 1886, E. Goldstein found new radiations in a gas discharge tube, naming them canal rays. These rays carry a positive charge.
- In 1897, J.J. Thomson discovered the electron, a subatomic particle with a negative charge.
- The neutron was discovered by Chadwick and has no charge.
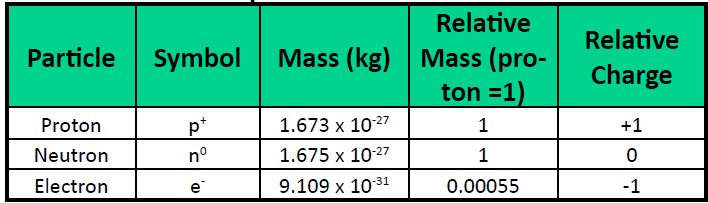
Let's Revise: Why is the mass of the electron usually ignored?
Thomson's Model of an Atom
Thomson's Model of the Atom, referred to as the plum pudding model, suggested that the atom is made up of a positively charged sphere with negatively charged electrons scattered throughout it, akin to currants in a Christmas pudding. Another way to picture it is like a watermelon, where the positive charge is spread out like the red fruit, and the electrons are like seeds embedded within.
- Electrons are embedded in a positively charged sphere; overall atom is neutral.
- The negative and positive charges are balanced, leading to an atom that is overall electrically neutral.
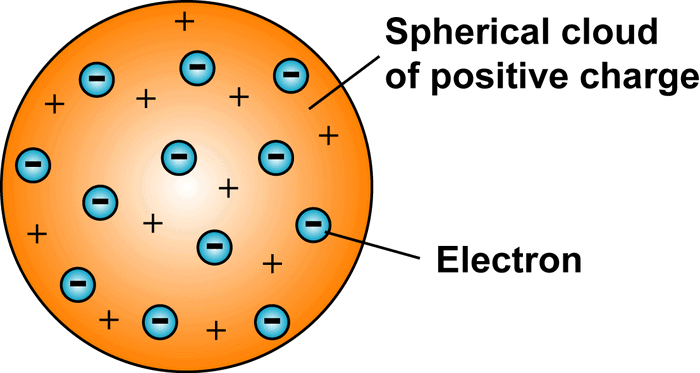 Plum Pudding Model
Plum Pudding Model
Rutherford's Model of an Atom
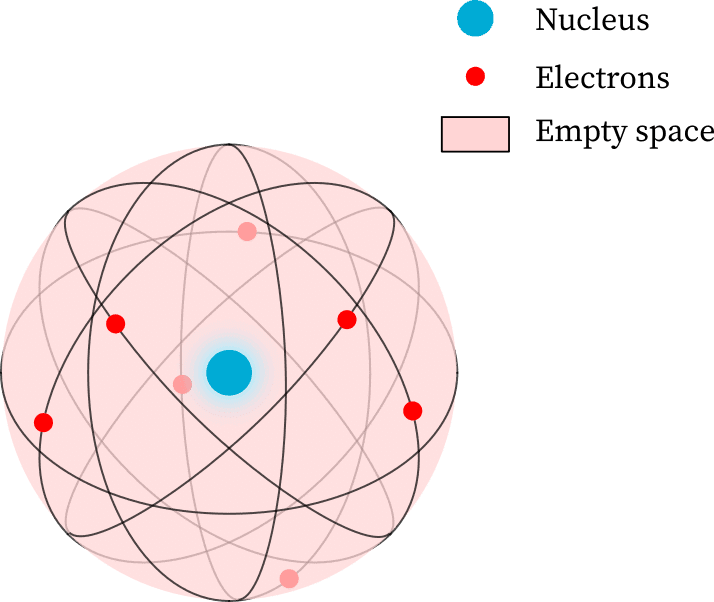
Rutherford's Model of the Atom brought forth the concept of a small, dense nucleus at the centre of the atom, with electrons moving around it, which greatly changed our understanding of atomic structure.
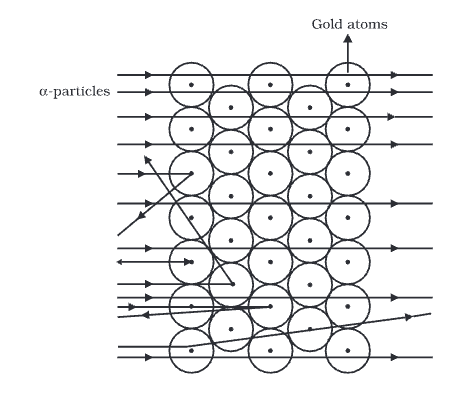 Rutherford's Experiment
Rutherford's Experiment
- α-particles are He nuclei (mass ≈ 4 u, charge +2e) emitted at high speeds, hence have high kinetic energy.
- Most of the atom's interior is empty, as many α-particles went through the gold foil without deflecting.
- A few particles were deflected, suggesting that the positive charge of the atom takes up very little space.
- A tiny number of α-particles were deflected back by 180°, showing that the positive charge and mass of the gold atom are concentrated in a very small area.
Conclusions made by Rutherford
- He calculated that the nucleus's radius is about 100,000 times smaller than that of the atom.
- The Nuclear Model of an Atom proposed by Rutherford includes:
- A positively charged centre called the nucleus, where nearly all the mass of the atom is found.
- The electrons orbit the nucleus in circular paths.
- The nucleus is very small compared to the atom's overall size.
 Rutherford's Nuclear Model of Atom
Rutherford's Nuclear Model of Atom
Drawbacks of Rutherford's Model of the Atom
- The orbiting electron in a circular path should not be stable. Any particle in such an orbit would experience acceleration. During this acceleration, charged particles would lose energy by radiating it. Therefore, the electron would lose energy and eventually spiral into the nucleus. If this were true, atoms would be highly unstable, which contradicts the fact that matter exists in a stable form.
Try yourself: Rutherford’s ‘alpha (α) particles scattering experiment’ resulted in the discovery of
Bohr's Model of Atom
Bohr's Model of the Atom changed how we understand atomic structure by introducing the idea that electrons move around the nucleus in specific energy levels. This model helps explain why atoms are stable and how they produce spectral lines.
Historical Context of Niels Bohr
Niels Bohr (1885-1962) was born in Copenhagen on 7 October 1885. He became a professor of physics at Copenhagen University in 1916 and won the Nobel Prize for his contributions to atomic structure in 1922. Some of his important writings include:
- The Theory of Spectra and Atomic Constitution
- Atomic Theory
- The Description of Nature
Postulates of Niels Bohr
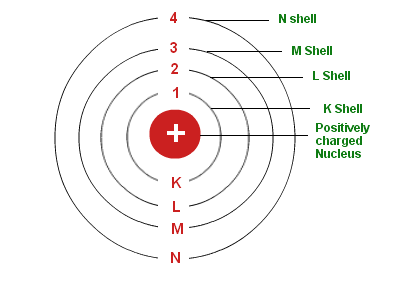
- Only certain specific orbits, called discrete orbits, are allowed for electrons inside the atom.
- Electrons do not emit energy while they are moving in these discrete orbits.
- These orbits or shells are referred to as energy levels. Energy levels in an atom are illustrated in Fig. 4.3.
Drawbacks of Bohr's Model of Atom
- Works for hydrogen but fails for multi-electron atoms.
- Cannot explain the splitting of spectral lines (fine structure; effects in magnetic/electric fields).
- Does not account for line intensities in spectra.
- Assumes fixed circular orbits; later quantum model uses orbitals (no fixed paths).
Neutrons
In 1932, J. Chadwick discovered a subatomic particle with no charge, which has a mass almost equal to that of a proton. This particle is called a neutron. Neutrons are found in the nucleus of all atoms, except hydrogen-1 (protium). Deuterium and tritium contain neutrons. Generally, a neutron is denoted as 'n'. The mass of an atom is the total of the masses of the protons and neutrons in the nucleus.
 Bohr's Model
Bohr's Model
Let's Revise: How is a hydrogen atom different from atoms of all other elements?
Distribution of Electrons in Different Orbits
The way electrons are arranged in various orbits, or energy levels, defines an atom's electron configuration.
Rules
- The maximum number of electrons that can fit in a shell is determined by the formula 2n², where 'n' represents the orbit number or energy level index (1, 2, 3, ...).
- The outermost orbit can hold a maximum of 8 electrons.
- Fill shells step-wise (K → L → M ...); outermost shell holds at most 8 electrons even if 2n2 allows more.
Thus, the maximum number of electrons in various shells is as follows:
- First orbit or K-shell can hold = 2 electrons
- Second orbit or L-shell can hold = 8 electrons
- Third orbit or M-shell can hold = 18 electrons
- Fourth orbit or N-shell can hold = 32 electrons
The atomic structure of the first eighteen elements is illustrated in a diagram.
The electrons in the outermost shell of an atom are called valence electrons. The number of valence electrons is essential in defining the chemical properties of the element.
Valency
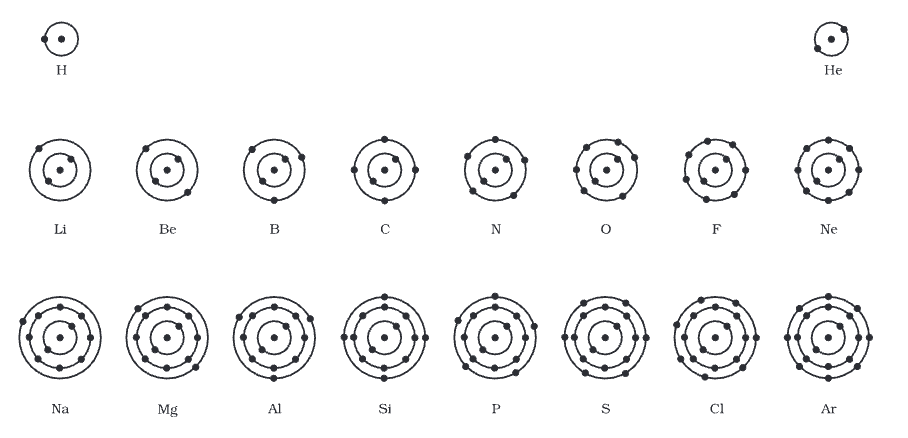 Atomic structure of the first eighteen elements
Atomic structure of the first eighteen elements
- An atom of each element has a definite combining capacity, called its valency.
- The number of bonds that an atom can form in a compound is shown by its valency.
- Valence electrons are the electrons in the outermost orbit of the atom.
Let's Revise
Q: How is the maximum number of electrons in a shell calculated?
Q: Why are valence electrons important?
Atomic Number & Mass Number
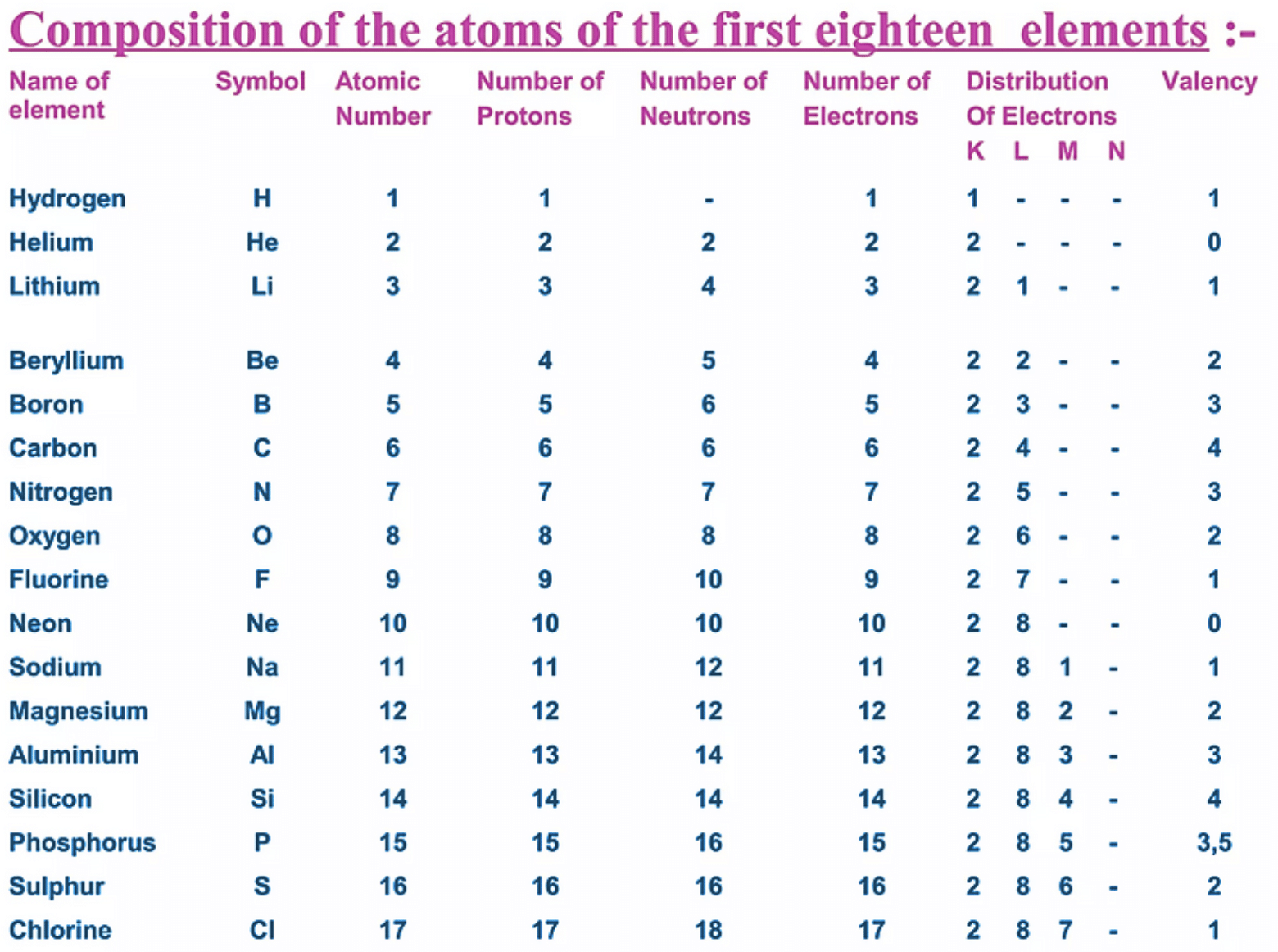
- The atomic number indicates the number of protons in an atom's nucleus, represented by 'Z'.
- The mass number is the total count of protons and neutrons, giving information about the atom's identity and mass.
- The total number of protons in an atom's nucleus is its atomic number, symbolised as 'Z'.
- The mass number of an atom is the sum of its protons and neutrons, represented by the letter 'A'.
An element is represented as AXZ, where Z is the atomic number (equal to the number of protons), A is the mass number, and X is the element's symbol. The mass number (A) can be calculated as: Mass number (A) = Number of protons (Z) + Number of neutrons.
Let's Revise: What is the mass number?
Isotopes
- Atoms of the same element with the same atomic number but different mass numbers are called isotopes. For example, hydrogen has three isotopes: protium (H), deuterium (²H or D), and tritium (³H or T).
- Chemical properties → same
- Physical properties → different
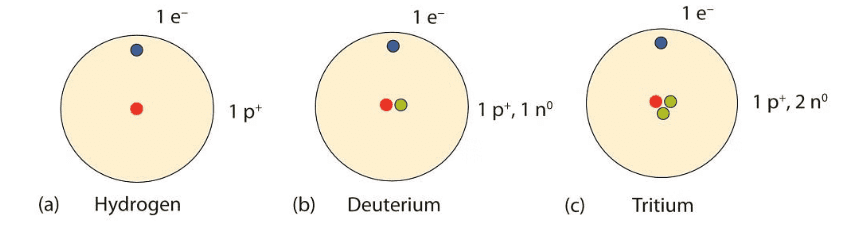 Isotopes of HydrogenApplications of Isotopes:
Isotopes of HydrogenApplications of Isotopes:
(a) An isotope of Uranium is used as fuel in nuclear reactors.
(b) An isotope of Cobalt is used in the treatment of cancer.
(c) An isotope of Iodine is used in the treatment of goitre.
Try yourself: The number of electrons in a neutral atom of an element X is 15, and the number of neutrons is 16. Which of the following is the correct representation of the element?
Isobars
- Atoms of different elements that have the same mass number but different atomic numbers are called isobars. For example, Argon-40 (₁₈Ar⁴⁰) and calcium-40 (₂₀Ca⁴⁰) are isobars.
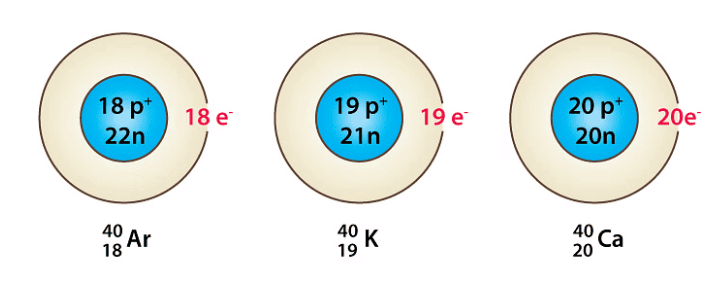 Examples of Isobars
Examples of Isobars
Let's Revise
Q: What are General Features of Isotopes?
You can practice questions from this chapter here: Case Based Questions: Structure of the Atom
FAQs on Chapter Notes: Structure of the Atom
| 1. What are protons, neutrons, and electrons, and where are they located in an atom? |  |
| 2. How do I figure out the number of protons, neutrons, and electrons in an element? |  |
| 3. What's the difference between Bohr's model and Rutherford's model of the atom? |  |
| 4. Why can't electrons just fall into the nucleus if they're attracted to the positive charge? |  |
| 5. What are valence electrons and why do they matter for CBSE Class 9 chemistry? |  |







