Revision Notes: Chemical Bonding & Molecular Structure
1. Lewis dot structures are shorthand to represent the valence electrons of an atom. The structures are written as the element symbol surrounded by dots that represent the valence electrons. 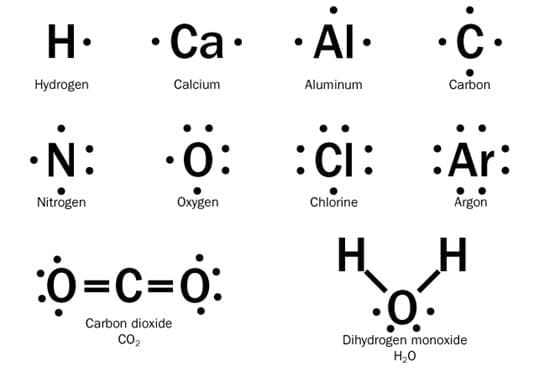
2. Covalent Bonds: The bond formed between two atoms by mutual sharing of electrons between them so as to complete their octets or duplets. When two atoms share one electron pair they are said to be joined by a single covalent bond. e.g H2 If two atoms share two electron pairs of electrons, the covalent bond between them is called a double bond, e.g O2 If two atoms share three electron pairs of electrons, the covalent bond between them is called a triple bond, e.g N2 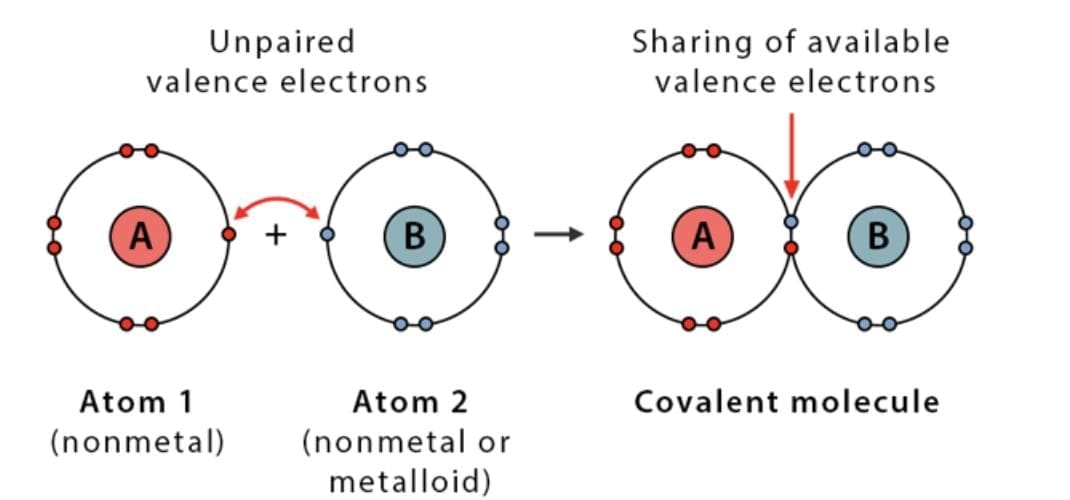
3. Octet Rule: Kossel and Lewis in 1916 developed an important theory of chemical combination between atoms known as the electronic theory of chemical bonding. According to this, atoms can combine either by the transfer of valence electrons from one atom to another or by sharing valence electrons to attain their octet. This is known as the octet rule.
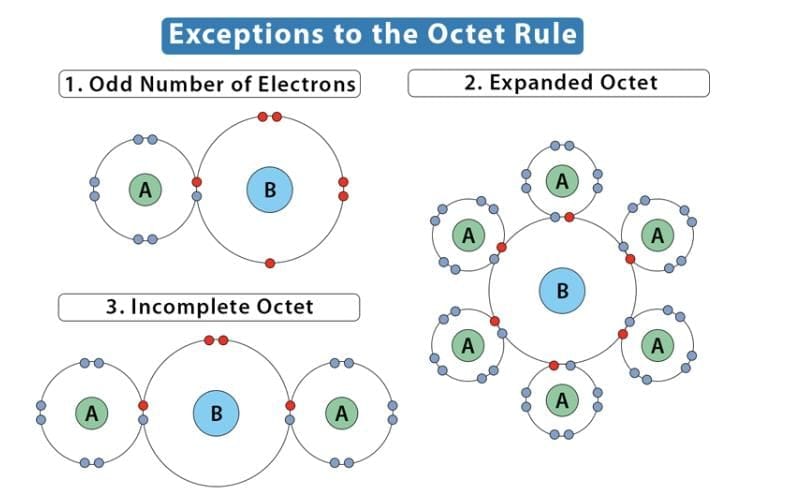
4. Limitations of octet rule:
- Incomplete octet of the central atom: In some compounds, the number of electrons surrounding the central atom is less than eight. This is especially common with elements having less than four valence electrons.
Examples: LiCl, BeCl2, BCl3 - Odd-electron molecules: Molecules with an odd number of electrons, such as nitric oxide (NO) and nitrogen dioxide (NO2), do not satisfy the octet rule for all atoms.
- Expanded octet: Elements in and beyond the third period of the periodic table have 3d orbitals available for bonding, in addition to 3s and 3p orbitals. Hence, these elements can accommodate more than eight electrons around the central atom. This phenomenon is called the expanded octet. Examples include PF5 and SF6.
- The octet rule does not explain the shape of molecules.
5. Electrovalent bond or Ionic Bond: An ionic bond is formed by the transfer of electrons from one atom (electropositive) to another atom (electronegative).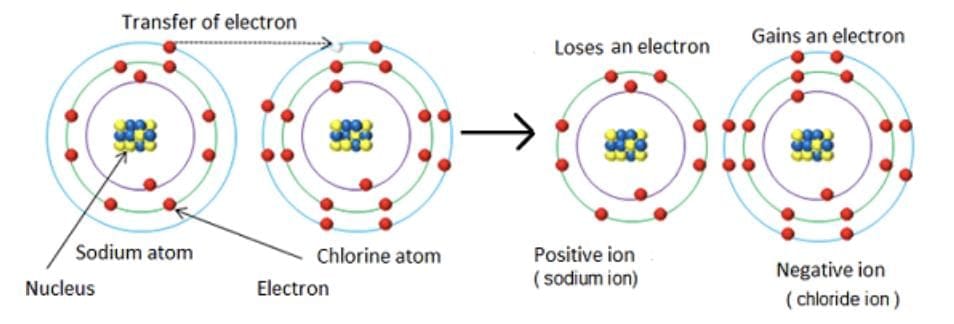
Ionic bonds form more easily between elements with low ionisation enthalpy and elements with high (negative) electron gain enthalpy. Most ionic compounds consist of cations derived from metallic elements and anions from non-metallic elements.
M(g) → M+(g) + e⁻; Ionization enthalpy
X(g) + e⁻ → X⁻(g) ; Electron gain enthalpy
M+(g) + X⁻(g) → MX(s)
7. Bond length is defined as the equilibrium distance between the nuclei of two bonded atoms in a molecule. 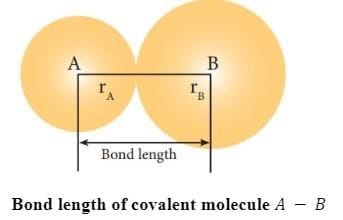
8. Bond Angle: It is the angle between the orbitals containing bonding electron pairs around the central atom in a molecule or complex ion. It provides information about the spatial distribution of orbitals around the central atom, helping to determine the shape of the molecule or ion.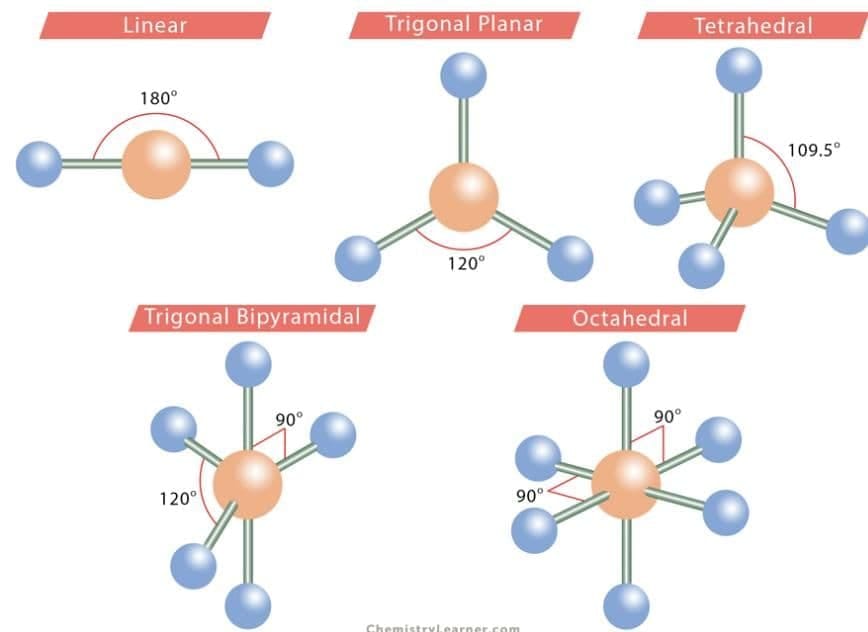
9. Bond enthalpy: It is defined as the amount of energy required to break one mole of bonds of a particular type between two atoms in a gaseous state. The unit of bond enthalpy is kJ mol-1.
10. Bond Order: Bond order is the number of chemical bonds between two atoms in a molecule. For example, the bond order of O2 is 2. As the bond order increases, the bond enthalpy increases, and the bond length decreases.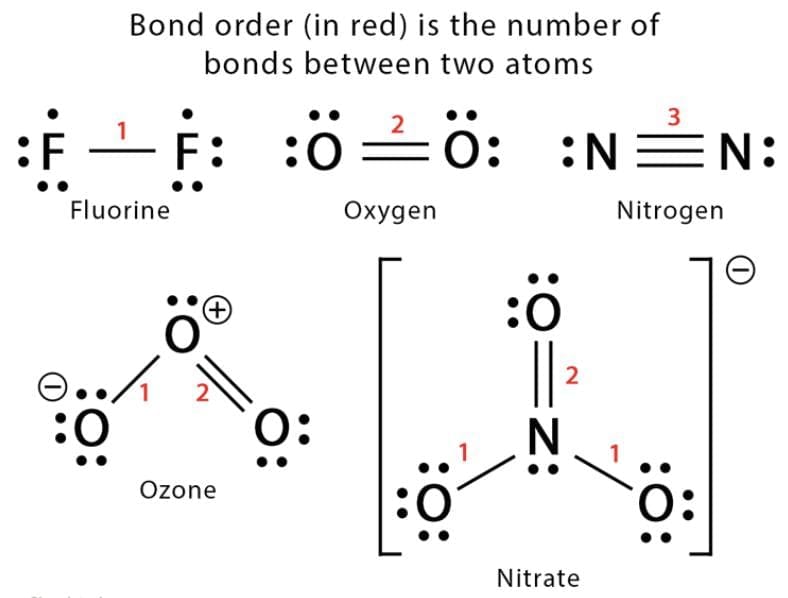
11. Resonance: According to the concept of resonance, when a single Lewis structure cannot accurately describe a molecule, multiple structures with similar energy, identical positions of nuclei, bonding, and non-bonding electron pairs are considered canonical forms. The actual molecule is a hybrid of these resonance structures.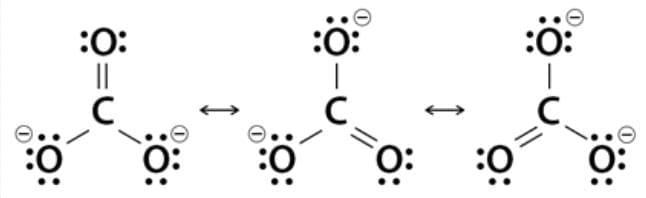
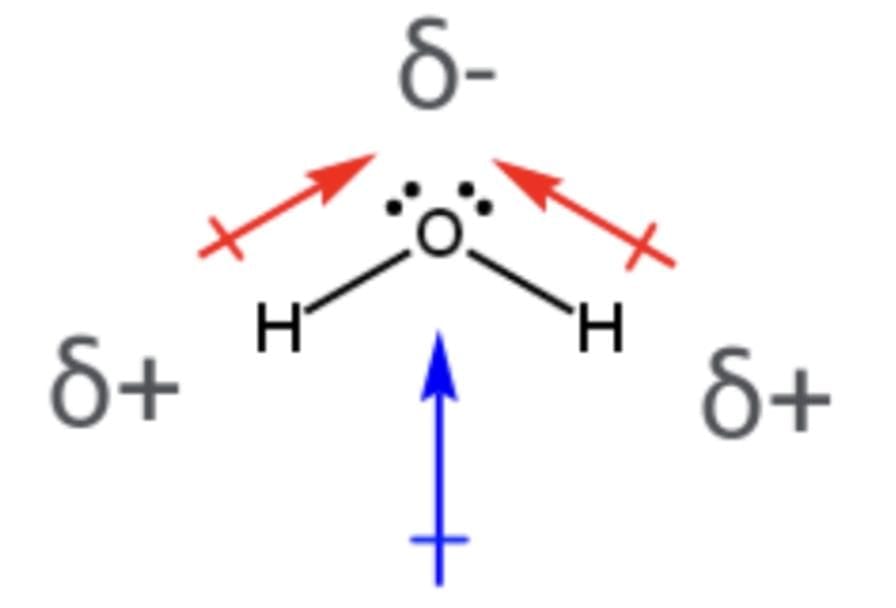
12. Polarity of bonds: In heteronuclear molecules such as HCl, the shared electron pair is displaced towards the more electronegative atom (chlorine in this case). The resulting covalent bond is called a polar covalent bond.
13. Dipole moment:Due to bond polarization, a molecule may possess a dipole moment, which is the product of the charge and the distance between the centres of positive and negative charges. It is usually denoted by the Greek letter μ. Mathematically,
Dipole moment (μ) = charge (Q) × distance of separation (r)
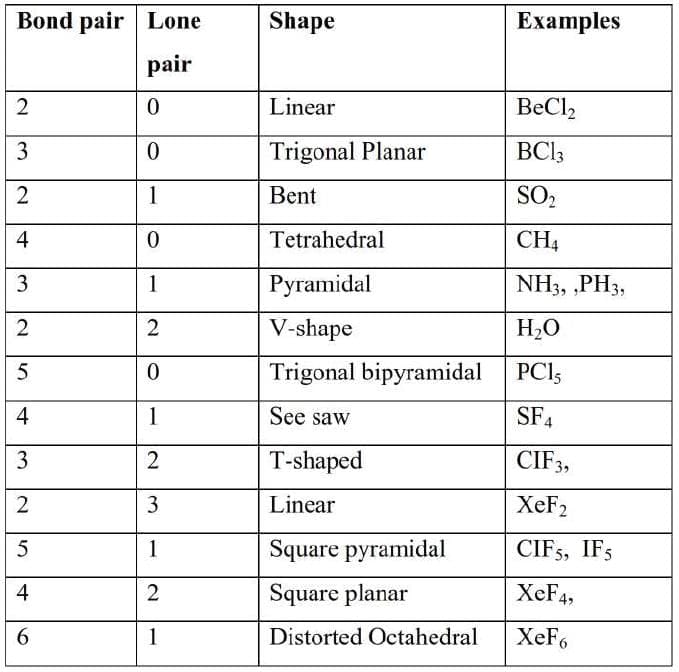
14. VSEPR Theory
- The shape of a molecule depends upon the number of valence shell electron pairs (bonded or nonbonded) around the central atom.
- Pairs of electrons in the valence shell repel one another since their electron clouds are negatively charged.
- These pairs of electrons tend to occupy such positions in space that minimize repulsion and thus maximise distance between them.
- The valence shell is taken as a sphere with the electron pairs localising on the spherical surface at maximum distance from one another.
- A multiple bond is treated as if it is a single electron pair and the two or three electron pairs of a multiple bond are treated as a single super pair.
- Where two or more resonance structures can represent a molecule, the VSEPR model applies to any such structure.
- Order of repulsive interaction strength: Lone pair (lp) - Lone pair (lp) > Lone pair (lp) - Bond pair (bp) > Bond pair (bp) - Bond pair (bp)
- Geometry of Molecules based on VSEPR Theory
15. Hybridization: It can be defined as the process of intermixing of the orbitals of slightly different energies so as to redistribute their energies, resulting in the formations of new set of orbitals of equivalent energies and shape. 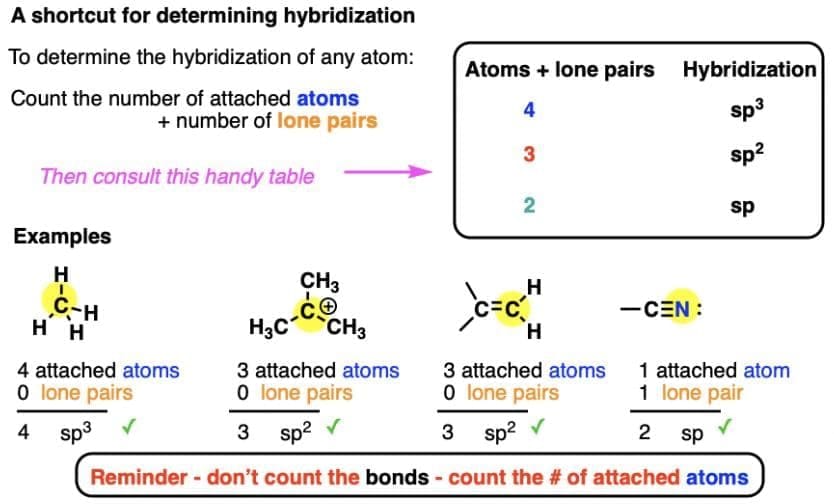 Salient Features of hybridization:
Salient Features of hybridization:
- The number of hybrid orbitals equals the number of atomic orbitals combined.
- Hybrid orbitals are equivalent in energy and shape.
- Hybrid orbitals form more stable bonds than pure atomic orbitals.
- They are oriented in space to minimise electron pair repulsions, leading to a stable arrangement.
16. Types of Hybridisation
- sp hybridisation: Involves mixing one s and one p orbital to form two equivalent sp hybrid orbitals. Example: BeCl2.
- sp2 hybridisation: Involves one s and two p orbitals forming three equivalent sp2 hybrid orbitals. Example: BCl3.
- sp3 hybridisation: Involves one s and three p orbitals forming four equivalent sp3 hybrid orbitals. Example: CH4.
17. Molecular orbital: It gives the electron probability distribution around a group of nuclei in a molecule. They are filled in the same way as atomic orbitals. Molecular orbitals are formed by a linear combination of atomic orbitals.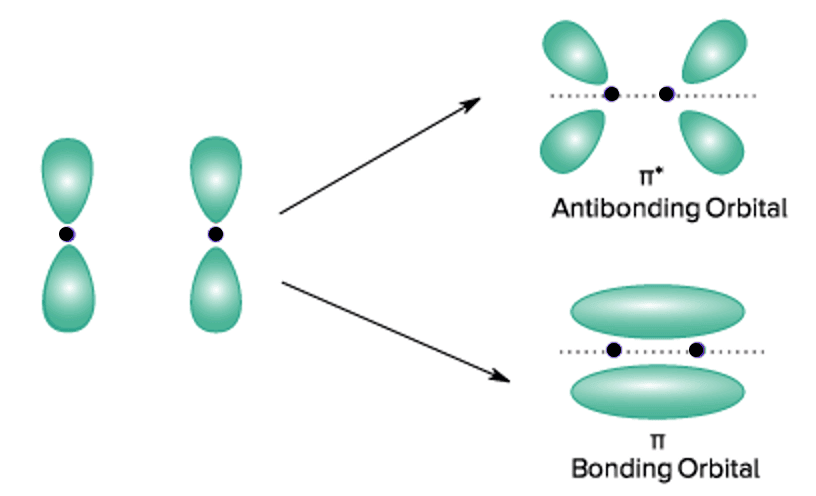
(A) Bonding molecular orbital.
A molecular orbital that is formed by addition overlap (i.e., when the lobes of atomic orbitals overlap with the same sign) of two atomic orbitals is known as a bonding molecular orbital.
It is represented as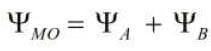 Its energy is lower than the atomic orbitals from which it is formed. It favours bonding.
Its energy is lower than the atomic orbitals from which it is formed. It favours bonding.
(b) Anti-bonding molecular orbital.
A molecular orbital that is obtained by the subtraction overlap (i.e., when the lobes of atomic orbitals overlap with the opposite sign) of two atomic orbitals is known as an anti-bonding molecular orbital.
It is represented as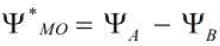 Its energy is higher than the atomic orbitals from which it is formed. It does not favour bonding.
Its energy is higher than the atomic orbitals from which it is formed. It does not favour bonding.
20. Bond order: Bond order is defined as half the difference between the number of electrons in bonding and antibonding orbitals:
B.O. = 1/2 (Nb - Na)
where Nb is the number of electrons in bonding orbitals and Na is the number of electrons in anti-bonding orbitals. Bond order helps estimate the stability of a molecule.
21. Relationship between electronic configuration and molecular behaviour:
- If Nb > Na, the molecule is stable.
- If Na > Nb, the molecule is unstable.
- If Na = Nb, the molecule is also unstable because the anti-bonding effect cancels the bonding effect.
22. Sigma (σ) molecular orbitals: A sigma molecular orbital is formed by the overlap of two s atomic orbitals, head-to-head overlap of an s and a p atomic orbital, or head-to-head overlap of two p atomic orbitals.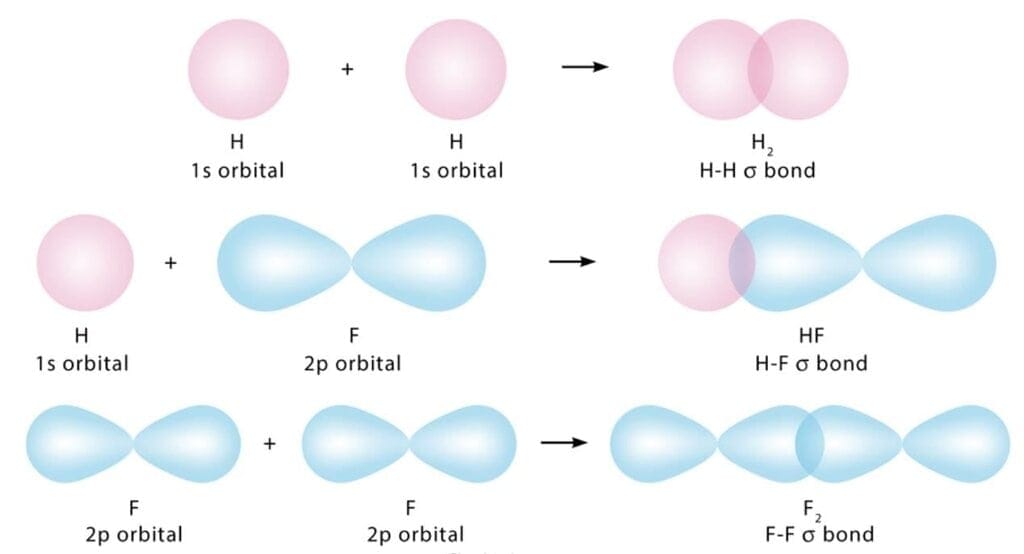
23. pi (π) molecular orbitals: A pi (π) molecular orbital is formed by lateral overlap of two parallel p-orbitals.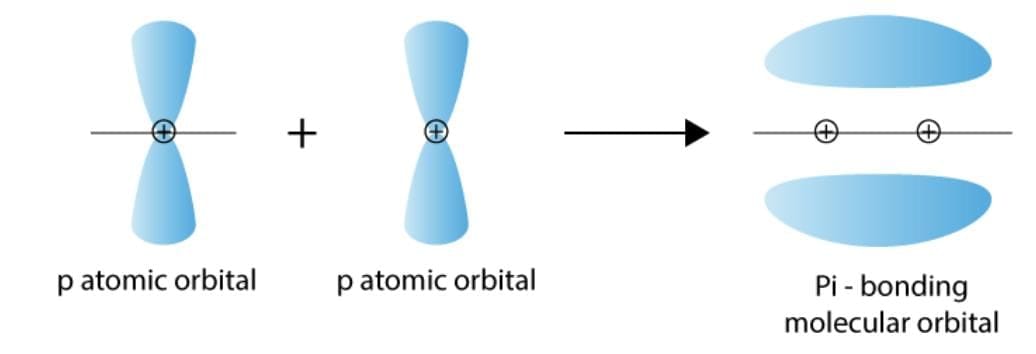
24. Conditions for the Combination of Atomic Orbitals. The linear combination of atomic orbitals takes place only if the following conditions are satisfied:
- The atomic orbitals must have the same or nearly the same energy.
- The atomic orbitals must have the same symmetry about the molecular axis.
- By convention, the z-axis is taken as the molecular axis.
- The atomic orbitals must overlap to the maximum extent.
- The greater the extent of overlap, the greater the electron density between the nuclei of the molecular orbital.
25. Energy level Diagrams for Molecular Orbitals. The increasing order of energies of various molecular orbitals for O2 and F2 is given below.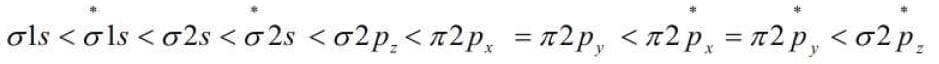
However, this sequence of energy levels of molecular orbitals is not correct for remaining molecules Li2, Be2, B2, C2, N2. For instance, it has been observed experimentally that for molecules such as B2, C2, N2 etc., the increasing order of energies of various molecular orbitals is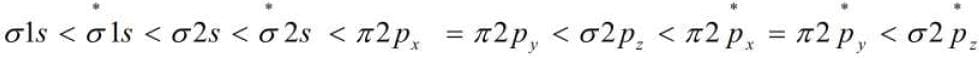
The important characteristic feature of this order is that the energy of σ2Pz molecular orbital is higher than that of π2px, and π2py, molecular orbitals in these molecules.
FAQs on Revision Notes: Chemical Bonding & Molecular Structure
| 1. What is the difference between ionic and covalent bonding? |  |
| 2. How can molecular geometry be determined using VSEPR theory? |  |
| 3. What are resonance structures, and why are they important in chemical bonding? |  |
| 4. What role do hybrid orbitals play in molecular bonding? |  |
| 5. How does electronegativity affect bond polarity? |  |





