Revision Notes: Thermodynamics
Introduction to Thermodynamics
- System: The portion of the universe under investigation.
- Open system: A system that can exchange both energy and matter with its surroundings.
- Closed system: A system that can exchange energy but not mass with its surroundings.
- Isolated system: A system that exchanges neither energy nor matter with its surroundings.
- Surroundings: The part of the universe external to the system and interacting with it.
- Boundary: The real or imaginary surface that separates the system from the surroundings.
State Variables and State Functions
- State variables: Parameters needed to specify the state of a system, for example pressure (P), volume (V), temperature (T), mass, composition.
- State functions (state properties): Properties that depend only on the current state of the system, not on how the system reached that state; examples: P, V, T, internal energy (E or U), enthalpy (H), entropy (S).
- Intensive properties: Properties independent of the size or amount of matter in the system, e.g. temperature, pressure, density, concentration.
- Extensive properties: Properties proportional to the amount of substance, e.g. volume, mass, internal energy, enthalpy, entropy.
Processes in Thermodynamics
- Process: Any change that takes a system from one state to another along a path in state space.
- Isothermal process: Process at constant temperature (T = constant).
- Isobaric process: Process at constant pressure (P = constant).
- Isochoric (isometric) process: Process at constant volume (V = constant).
- Adiabatic process: Process with no heat exchange between system and surroundings (q = 0).
- Cyclic process: Process in which the system returns to its initial state after a series of changes.
- Reversible process: An ideal process that proceeds infinitely slowly, remaining infinitesimally close to equilibrium at every stage and whose direction can be exactly reversed by infinitesimal changes.
- Irreversible process: Any real process that proceeds at a finite rate, involves dissipative effects (friction, rapid expansion, mixing), and cannot be exactly reversed.
Heat, Energy and Work
Heat (Q)
- Heat is energy transferred between system and surroundings due to a temperature difference.
- Heat added to the system is taken as positive; heat removed from the system is taken as negative (sign convention used here).
- Heat is an extensive quantity and it is not a state function (it depends on the path).
Energy
- Energy is the capacity to do work.
- Energy is an extensive property.
- SI unit: joule (J).
Work (W)
- Mechanical work (one common form) = force × displacement; infinitesimal: dW = F dx.
- Sign convention used here: work done on the system is positive; work done by the system is negative.
- Pressure-volume (PV) work: Work associated with volume change against an external pressure.
- For a reversible expansion/compression of an ideal gas, the mechanical work done on the system is
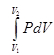
- For an isothermal reversible change of an ideal gas:
- W = -nRT ln(V2/V1)
- Using base-10 logarithm, W = -2.303 nRT log(V2/V1)
- Since pV = constant for isothermal change, ln(V2/V1) = ln(p1/p2). Thus W = -nRT ln(V2/V1) = -nRT ln(p1/p2).
- Unit: joule (J).
Internal Energy (E or U)
- Internal energy is the sum of all microscopic energy contributions of a system (translational, rotational, vibrational, electronic, chemical bond energies, etc.).
- Change in internal energy at constant volume equals the heat exchanged at constant volume: ΔE = qv.
- ΔE is a state function and is extensive.
- For an exothermic process ΔE is negative; for an endothermic process ΔE is positive (using ΔE = Efinal - Einitial).
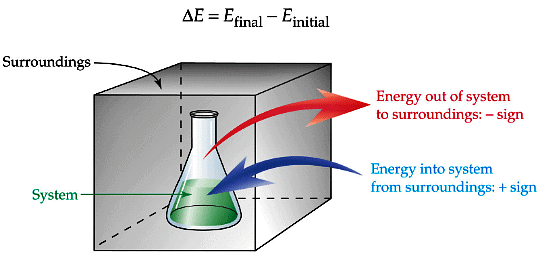
First Law of Thermodynamics
Energy cannot be created or destroyed but can be converted from one form to another. For an isolated system the total energy is constant.
Mathematical statement:
- Heat supplied to the system = increase in internal energy + work done by the system.
- q = ΔE + w
- For an infinitesimal change: dq = dE + dw
- Here q is heat supplied to the system and w is work done on the system (sign convention used).
- For an ideal gas undergoing an isothermal change, ΔE = 0, hence q = -w.
Special cases
- For an isolated system, dq = 0, so dE = -dw.
- For a system involving mechanical work only, ΔE = q - pΔV (when p is external pressure and sign conventions are observed).
- At constant volume (isochoric), ΔE = qv.
- For an isothermal process of an ideal gas, ΔE = 0 and q = -w = -∫pext dV.
- For an adiabatic process, q = 0, so ΔE = w (work done on the system changes internal energy).
Enthalpy (H)
- Definition: H = E + PV.
- For differential changes, dH = dE + d(PV) = dE + PdV + VdP.
- At constant pressure and when only PV work is present, the heat exchanged equals the change in enthalpy: qp = ΔH.
- For exothermic reactions ΔH is negative; for endothermic reactions ΔH is positive (ΔH = Hproducts - Hreactants).
Relation between ΔH and ΔE
- At constant temperature, ΔH = ΔE + Δ(ng)RT
- Here Δ(ng) = (moles of gaseous products - moles of gaseous reactants) and R is the gas constant.
- This relation is useful when a reaction involves change in number of moles of gases.
Heat Capacity
- Heat capacity (C): Amount of heat required to raise the temperature of the system by 1 K (or 1 °C). C = q / ΔT.
- Specific heat capacity (Cs): Heat required to raise the temperature of 1 g of a substance by 1 K. Cs = C / mass (in g).
- Molar heat capacity (Cm): Heat required to raise the temperature of 1 mole of a substance by 1 K. Cm = C / n (n = number of moles).
- Heat capacity at constant volume: Cv = (∂E/∂T)v.
- Heat capacity at constant pressure: Cp = (∂H/∂T)p.
- For an ideal gas (per mole), Cp - Cv = R.
Variation of Heat of Reaction with Temperature
- Average molar heat capacity between T1 and T2 may be estimated from changes in enthalpy or internal energy:
- Cp,avg = (H2 - H1) / (T2 - T1).
- Cv,avg = (E2 - E1) / (T2 - T1).
Bomb Calorimeter
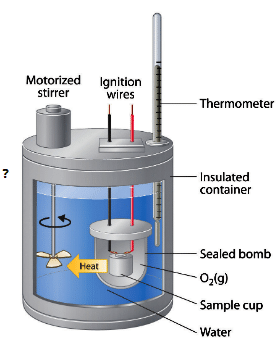
- In a bomb calorimeter experiments are conducted at constant volume.
- Heat exchanged in the calorimeter system = Z × ΔT, where Z is the heat capacity of the calorimeter (calorimeter constant) and ΔT is the temperature rise.
- Heat changes at constant volume are expressed as ΔE; heat changes at constant pressure are expressed as ΔH.
Enthalpies of Reactions
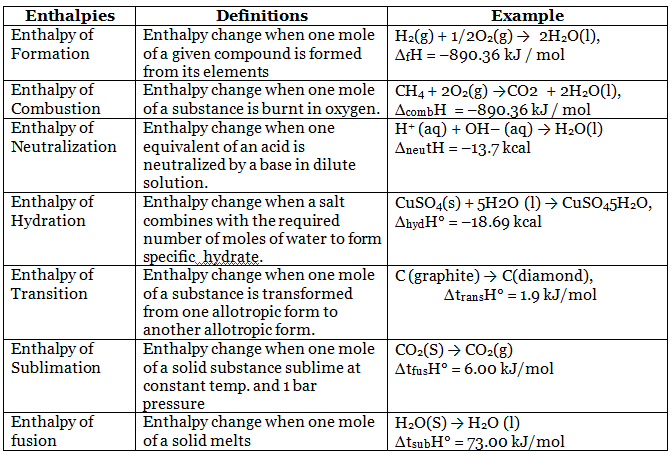
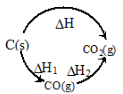
Hess's Law (Law of constant heat summation)
The total enthalpy change for a chemical reaction is the same whether the reaction occurs in one step or via several steps; enthalpy is a state function.
Therefore, if a reaction can be expressed as a sum of steps, the overall enthalpy change ΔH is the algebraic sum of the enthalpy changes of the individual steps:
- ΔH = ΔH1 + ΔH2 + ...
Born-Haber Cycle
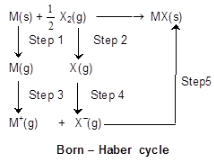
- Used to calculate lattice energy of an ionic solid by applying Hess's law to a sequence of hypothetical steps which form the overall formation reaction.
- For the formation of MX(s) from the elements, the algebraic sum of ionisation energy, bond dissociation energies, electron affinity, sublimation energy, atomisation energies, and lattice energy equals the enthalpy of formation ΔHf.
- Lattice energy: The enthalpy change when 1 mole of an ionic crystalline solid is formed from its gaseous ions (conventionally taken as negative for formation).
Applying Hess's law we may write a typical algebraic sum of steps leading to ΔHf (MX) which includes lattice energy as one term.
Second Law of Thermodynamics
Kelvin-Planck statement: It is impossible to construct a cyclic device whose sole effect is to take heat from a hot reservoir and convert it completely into work without transferring part of the heat to a cold reservoir.
Entropy (S)
- Definition: For a reversible process, the change in entropy is ΔS = qrev / T (for a process at temperature T where qrev is the heat exchanged reversibly).
- Entropy is a state function that measures the degree of randomness or disorder of a system; greater randomness corresponds to larger entropy.
- For an isolated system, the entropy of the universe increases for irreversible (spontaneous) processes: ΔSuniverse > 0. At equilibrium ΔSuniverse = 0.
- For melting of ice or other processes that increase disorder, ΔS for the system is positive.
- Entropy change for an isothermal reversible expansion of an ideal gas:
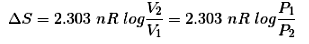
- ΔS = nR ln(V2/V1)
Spontaneous Processes
- Spontaneous processes occur on their own under given conditions (they do not require continuous input of external work to proceed). Examples: spontaneous flow of heat from hot to cold, liquid flowing from higher to lower level under gravity, spontaneous diffusion.
- Spontaneity criteria must consider both system and surroundings; entropy change of the universe and Gibbs free energy provide standard criteria.
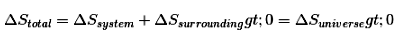
Gibbs Free Energy (ΔG)
- Definition: ΔG = ΔH - TΔS (for processes at constant temperature and pressure).
- At constant T and P, a process is spontaneous if ΔG < 0, non-spontaneous if δg> 0, and at equilibrium ΔG = 0.
- Relationship with equilibrium constant: ΔG° = -RT ln Keq, where ΔG° is the standard free energy change and Keq the equilibrium constant.
- Relation with cell potential (electrochemistry): ΔG° = -nFE°cell, where n is the number of moles of electrons transferred, F is the Faraday constant and E°cell is the standard cell potential. More generally ΔG = -nFEcell.
Bond Energies
- Bond energy (average bond enthalpy) is the average enthalpy change required to break one mole of bonds of a particular type in gaseous molecules.
- Example data as given:
- H-O-H (O-H bond) → fragments: H(g) + ½ O(g) has bond dissociation contributions quoted as 498 kJ (first example in input).
- O-H(g) → H2(g) + ½ O2(g) value quoted as 430 kJ (as per input).
- Average O-H bond energy calculated from these examples: ΔHO-H = (498 + 430) / 2 = 464 kJ mol-1.
- Bond energies are useful for approximate estimation of reaction enthalpies by summing bond energies of bonds broken and formed.
Efficiency of a Heat Engine (Carnot Cycle)
- The Carnot cycle is an ideal reversible heat engine operating between a hot reservoir at temperature TH and a cold reservoir at temperature TC.
- Maximum possible efficiency for any heat engine operating between these two reservoirs is the Carnot efficiency:
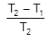
- η = 1 - TC / TH
- Work and heat relations for reversible ideal gas processes:
- For an isothermal reversible process of an ideal gas: W = -nRT ln(V2/V1).
- Heat absorbed at the high temperature (isothermal expansion) qH = nRTH ln(V2/V1).
- Efficiency η = Wnet / qH = 1 - qC / qH = 1 - TC / TH for Carnot cycle.
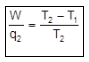
Important Formulae and Conventions (Quick Reference)
- First law: q = ΔE + w (w is work done on the system).
- PV work (reversible): w = -∫Pint dV; for ideal gas isothermal: w = -nRT ln(V2/V1).
- Enthalpy: H = E + PV; at constant pressure qp = ΔH.
- Relation ΔH and ΔE (constant T): ΔH = ΔE + Δ(ng)RT.
- Heat capacities: C = q/ΔT; Cv = (∂E/∂T)v; Cp = (∂H/∂T)p; for ideal gas (per mole) Cp - Cv = R.
- Entropy (reversible): ΔS = qrev / T; for ideal gas isothermal ΔS = nR ln(V2/V1).
- Gibbs free energy: ΔG = ΔH - TΔS; ΔG° = -RT ln Keq; ΔG = -nFE_cell.
- Carnot efficiency: ηmax = 1 - TC / TH.
- Hess's law: overall ΔH = sum of ΔH of individual steps.






