HOTS Questions: Atoms and Molecules
Q1: (a) An element shows variable valencies 4 and 6. Write the formulae of its two oxides.
(b) An element forms an oxide A2O5.
(i) What is the valency of the element A ?
(ii) What will be the formula of the chloride of the element ?
Ans: (a) Let the element be represented by the symbol E.
Formula of the oxide in which the valency of E is 4 = E2O4 or EO2. This is because each E atom uses four valence electrons to combine with oxygen (each O has valency 2).
Formula of the oxide in which the valency of E is 6 = E2O6 or EO3. Here each E atom shows valency 6 and combines accordingly with oxygen.
(b) Formula of the oxide of the element = A2O5.
(i) The valency of the element A in the oxide = +5. Explanation: Five oxygen atoms (each of valency 2) give a total negative charge of 10; this is balanced by two A atoms, so each A must have valency +5.
(ii) The formula of the chloride of the element A = ACl5, since A has valency 5 it will combine with five chloride ions (each of valency 1).
Q2: (a) Why does not the atomic mass of an element represent the actual mass of its atom ?
(b) The atomic mass of an element is in fraction. What does it mean ?
(c) Why is the value of Avogadro's number 6.022 x 1023 and not any other value ?
(d) Does one gram mole of a gas occupy 24.4 L under all conditions of temperature and pressure ?
Ans: (a) The actual mass of an atom is extremely small (for example the mass of a hydrogen atom is about 1.67 × 10-27 kg). For convenience we use relative atomic mass (atomic mass in atomic mass units, u) which compares the mass of an atom with 1/12 of the mass of a carbon-12 atom. The relative atomic mass is a dimensionless number (given in u) and the corresponding mass of one mole of the element is expressed in grams (gram-atomic mass). Thus atomic mass given in u is a relative quantity, not the direct actual mass in kilograms.
(b) If the atomic mass of an element is fractional, it means the element exists as a mixture of isotopes with different masses. The reported atomic mass is the weighted average of the masses of these isotopes, weighted by their natural abundances; this average can be a fraction.
(c) Avogadro's number (6.022 × 1023) is the experimentally determined number of atoms (or molecules) in one mole. Historically it is defined so that 12 g of carbon-12 contains exactly 6.022 × 1023 atoms. This value is determined by precise measurements and is not an arbitrary choice; it links the microscopic scale (atoms) to the macroscopic scale (grams and moles).
(d) No. One gram mole of an ideal gas occupies 22.4 L only under standard conditions of temperature and pressure (N.T.P. = 273 K and 1 atm or 760 mm Hg). The volume occupied by one mole of a gas depends on temperature and pressure and can be found from the ideal gas equation PV = nRT for other conditions.
Q3: What weight of calcium contains the same number of atoms as are present in 3.2 g of sulphur ?
Ans: Step I. Number of atoms in 3.2 g of sulphur:
Gram atomic mass of S = 32 g
Number of moles of S in 3.2 g = 3.2 ÷ 32 = 0.10 mole
Number of atoms = 0.10 × 6.022 × 1023 = 6.022 × 1022 atoms.
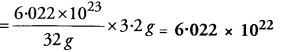
Step II. Weight of calcium containing 6.022 × 1022 atoms:
Number of moles of Ca required = 6.022 × 1022 ÷ 6.022 × 1023 = 0.10 mole
Gram atomic mass of Ca = 40 g
Mass of 0.10 mole of Ca = 0.10 × 40 = 4.0 g
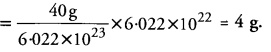
Therefore, 4.0 g of calcium contains the same number of atoms as are present in 3.2 g of sulphur.
Q4: In magnesium sulphide, the ratio by mass of Mg and S is 3 : 4. What is the ratio of the number of Mg and S atoms ?
Ans: Formula of magnesium sulphide = MgS
Given ratio by mass of Mg : S = 3 : 4
Number of moles ratio = (mass/Mr) for each element:
Mg: 3 ÷ 24 = 0.125; S: 4 ÷ 32 = 0.125
∴ Ratio of the number of Mg and S atoms = 0.125 : 0.125 = 1 : 1
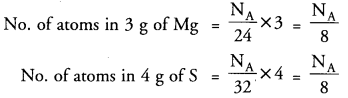
Hence the ratio of the number of Mg and S atoms is 1 : 1 and the formula is MgS.
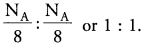
Q5: On analysing an impure sample of sodium chloride, the percentage of chlorine was found to be 45.5 What is the percentage of pure sodium chloride in the sample ?
Ans: Molar mass of pure NaCl = Atomic mass of Na + Atomic mass of Cl = 23 + 35.5 = 58.5 u
Percentage of chlorine in pure NaCl = (35.5 ÷ 58.5) × 100 = 60.68% (approximately).
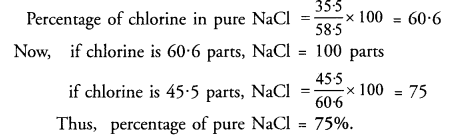
If the impure sample contains 45.5% chlorine by mass, then the percentage of NaCl present = (45.5 ÷ 60.68) × 100 ≈ 75.0%
Therefore the sample is about 75% pure sodium chloride.
Q6: A flask P contains 0.5 g mole of oxygen gas. Another flask Q contains 0.4 mole of ozone gas. Which of the two flasks contains greater number of oxygen atoms ?
Ans:
1 molecule of oxygen (O2) contains 2 oxygen atoms.
1 molecule of ozone (O3) contains 3 oxygen atoms.
In flask P : 0.5 mole of O2 contains 0.5 × 6.022 × 1023 molecules.
Number of oxygen atoms = 0.5 × 6.022 × 1023 × 2 = 1.0 × 6.022 × 1023 = 6.022 × 1023 atoms.
In flask Q : 0.4 mole of O3 contains 0.4 × 6.022 × 1023 molecules.
Number of oxygen atoms = 0.4 × 6.022 × 1023 × 3 = 1.2 × 6.022 × 1023 = 7.2264 × 1023 atoms.
∴ Flask Q contains the greater number of oxygen atoms.
Q7: Silicon forms a compound with chlorine in which 5.6 g of silicon is combined with 21.3 g of chlorine..
Calculate the formula of the compound (Atomic mass : Si = 28 : Cl = 35-5).
Ans:

The number of moles of Cl = 21.3 ÷ 35.5 = 0.60 mole
Ratio Si : Cl = 0.20 : 0.60 = 1 : 3
The simplest whole number ratio is Si : Cl = 1 : 3
The formula of the compound = SiCl3.
Q8: A colourless liquid is thought to be a pure compound. Analysis of three samples of the material yield the following results.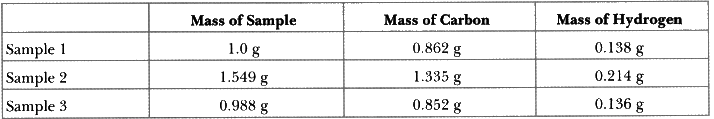
Could the material be a pure compound?
Ans: Analysis

Q9. What is the fraction of the mass of water due to neutrons?
Ans: Consider one molecule of water, H2O.
Mass numbers (approximate): H ≈ 1 (no neutron in the common protium isotope), O ≈ 16 (contains 8 neutrons).
Number of neutrons per H2O molecule = 0 + 8 = 8 neutrons.
Total mass of one water molecule ≈ 18 u (2 × 1 for H + 16 for O).
Fraction of mass due to neutrons = 8 ÷ 18 = 4 ÷ 9 ≈ 0.444... (about 44.4%).
Thus roughly 4/9 of the mass of water is due to neutrons.
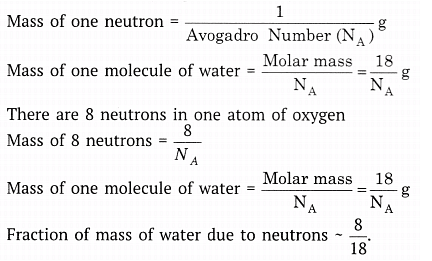
Q10: Calculate the number of electrons present in 15.4 g of carbon tetrachloride (CCl4).
Ans: Number of moles of CCl4 = 15.4 ÷ (12 + 4 × 35.5) = 15.4 ÷ 154 = 0.10 mole
∵ = 0.1 mole
Number of molecules in 0.1 mole = 0.1 × 6.022 × 1023 = 6.022 × 1022 molecules
Electrons per molecule of CCl4 = electrons in C + 4 × electrons in Cl = 6 + 4 × 17 = 74 electrons
∴ Total number of electrons = 74 × 6.022 × 1022 = 4.4563 × 1024 electrons (approximately).
Therefore, 15.4 g of CCl4 contains about 4.456 × 1024 electrons.
Q11: A big drop of water has volume 1.0 mL. How many molecules of water are there in this drop, If the density of water is lg/mL?
Ans: Volume of drop of water = 1.0 mL
Density of water = 1.0 g/mL
∴ Mass of drop of water = Volume × Density = 1.0 g
Molar mass of H2O = 2 × 1 + 16 = 18 g/mol
Number of moles in 1 g = 1 ÷ 18 = 0.055555... mole
Number of molecules = 0.055555... × 6.022 × 1023 ≈ 3.346 × 1022 molecules
Therefore the drop contains about 3.35 × 1022 water molecules.
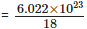
Q12: You are provided with a fine white coloured powder which is either sugar or salt. How would you identify it without tasting?
Ans: Two simple, safe tests are:
1. Heat test: On gentle heating the powder, sugar will char and turn brown or black (it decomposes), while common salt (sodium chloride) will not char.
2. Conductivity test: Dissolve a small portion of the powder in distilled water and test with a conductivity tester or a simple circuit. Salt solution conducts electricity (because it forms ions), whereas a sugar solution does not conduct appreciably.
Either of these tests will identify the substance without tasting.





