JEE Advanced Previous Year Questions (2018 - 2025): Haloalkanes and Haloarenes
2023
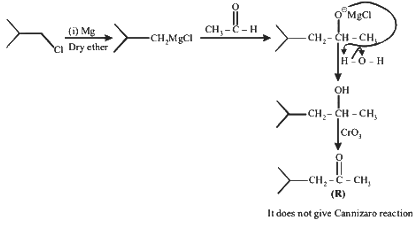
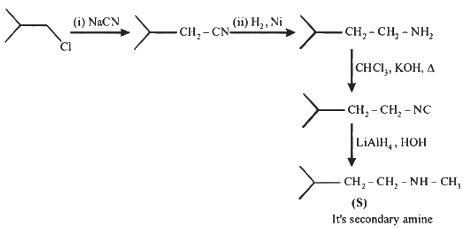
Q1: In the following reactions, P, Q, R, and S are the major products.
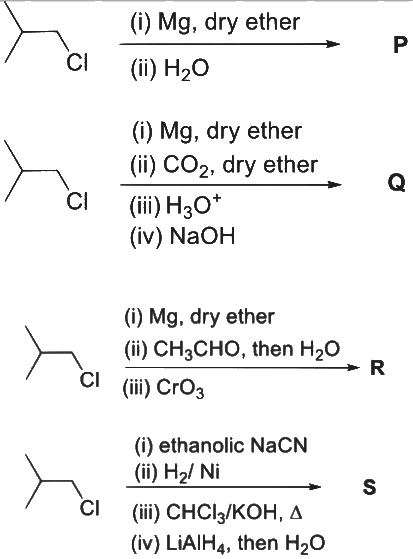
(a) P is a primary alcohol with four carbons.
(b) Q undergoes Kolbe's electrolysis to give an eight-carbon product.
(c) R has six carbons and it undergoes Cannizzaro reaction.
(d) S is a primary amine with six carbons. [JEE Advanced 2023 Paper 2]
Ans: (b)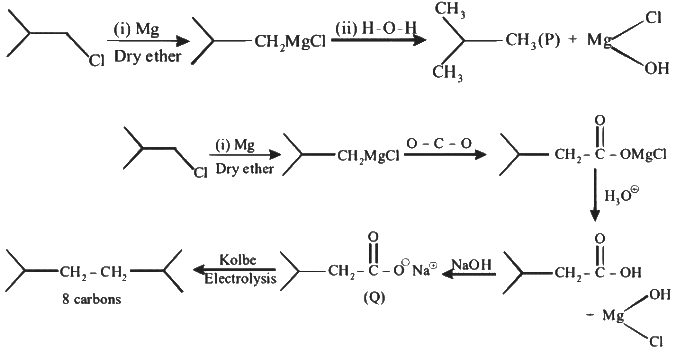
2021
Q1: In the reaction given below, the total number of atoms having sp2 hybridization in the major product P is _________. [JEE Advanced 2021 Paper 1]
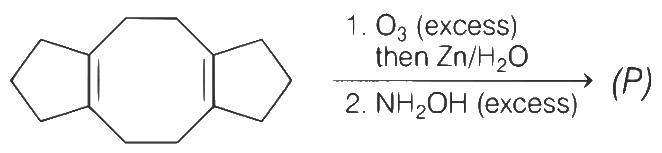
Ans: 12
Alkene on treatment with ozone (O3) followed by treatment with Zn/H2O gives carbonyl compounds.
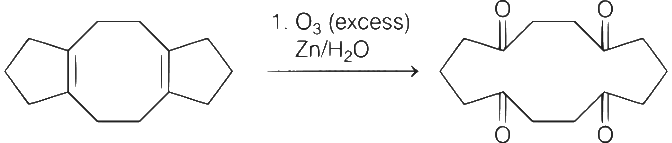 Carbonyl compounds react with NH2OH to give oximes.
Carbonyl compounds react with NH2OH to give oximes.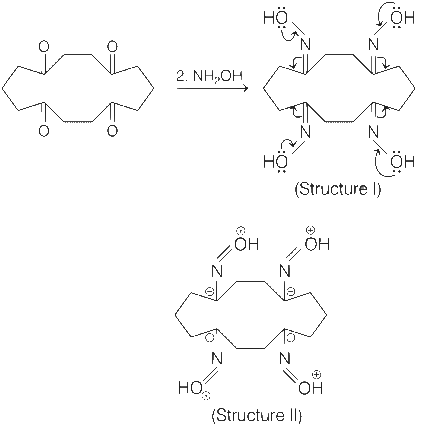
There are four sp2-carbon atoms, four sp2-nitrogen atoms and four sp2-oxygen atoms as shown in structures I and II.
Therefore, total number of atoms having sp2-hybridisation are twelve (12).
Q2: The major product formed in the following reaction of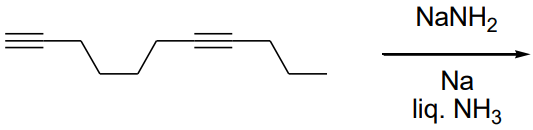
(a) 
(b) 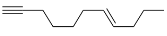
(c) 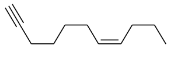
(d) 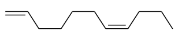 [JEE Advanced 2021 Paper 1]
[JEE Advanced 2021 Paper 1]
Ans: (b)
It is a case of Birch reduction. Alkynes on reaction with alkali metal in liq. NH3 gives trans-alkene. But terminal alkynes do not get reduced.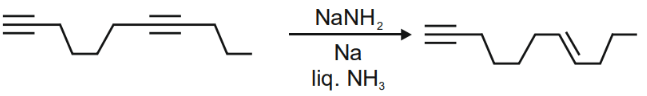
FAQs on JEE Advanced Previous Year Questions (2018 - 2025): Haloalkanes and Haloarenes
| 1. What are the main types of nucleophilic substitution reactions in haloalkanes that appear in JEE Advanced? |  |
| 2. How do I identify whether a haloarene will undergo nucleophilic aromatic substitution in JEE Advanced problems? |  |
| 3. What's the difference between elimination and substitution reactions for haloalkanes in competitive exams? |  |
| 4. Why do secondary haloalkanes show competing SN1 and SN2 pathways in JEE problems? |  |
| 5. What reaction mechanisms do haloalkanes and haloarenes commonly show in JEE Advanced exams from 2018-2025? |  |





