JEE Exam > JEE Notes > Chemistry Main & Advanced > Revision Notes: Classification of Elements & Periodicity in Properties
Revision Notes: Classification of Elements & Periodicity in Properties
Some Important Points and Terms of the Chapter
- Dobereiner's Triads: In 1817 a German chemist Dobereiner identified certain groups of three elements. These groups of three elements having similar properties was called triads. When three elements were arranged in order of their increasing atomic masses, the atomic mass of the middle element was roughly the mean of the atomic masses of the other two elements.
- New Lands Law of octaves: When elements were arranged in order of their increasing relative atomic masses. The properties of every eight elements were similar to the first one, like the eighth note of a musical scale. This repetition in the properties of elements is just like the repetition of eighth note in an octave of music.
- Mendeleev's Periodic Law: The physical and chemical properties of elements are the periodic function of their atomic masses.
- Mendeleev's Periodic Table: When mendeleev started his work, 63 elements were known at that time. He selected hydrogen and oxygen as they are very reactive and formed compounds with most elements. Mendeleev's periodic table contains vertical columns called groups and horizontal rows called periods. There were 7 periods and 8 groups. Noble gases were not known at that time. So there was no group of noble gases. The elements in each group of the periodic tables are similar to one another in many properties. The similar properties of the elements are repeated periodically
(a) Merits of mendeleev's classification
- Mendeleev's periodic law predicted the existence of some elements that had not been discovered at that time.
- Could predict the properties of several elements on the basis of their position in the periodic table.
- Could accommodate noble gases when they were discovered.
(b) Limitations of mendeleev's classification :
- The correct position could not be assigned to the hydrogen in the periodic table.
- Wrong order of the atomic masses of some elements could not be explained.
- The position of isotopes could not be explained.
- Uncertainty in prediction of new elements was there.
- Modern periodic law: Properties of elements are the periodic function of their atomic number.
- Modern Periodic Table: This table was prepared was Bohr and is based upon the electronic configuration of elements. The table consists of 18 vertical columns called groups. Elements having similar outer electronic configurations in their atoms are arranged in vertical columns, referred to as groups. According to the recommendation of International Union of Pure and Applied Chemistry (IUPAC), the groups are numbered from 1 to 18 and the table consists of 7 horizontal rows called periods. The first period contains 2 elements. The subsequent periods consists of 8, 8, 18, 18 and 32 elements, respectively. The seventh period is incomplete and like the sixth period would have a theoretical maximum (on the basis of quantum numbers) of 32 elements. In this form of the Periodic Table, 14 elements of both sixth and seventh periods (lanthanoids and actinoids, respectively) are placed in separate panels at the bottom
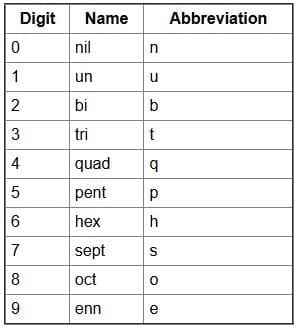
- Notation for IUPAC Nomenclature of Elements With Z > 100
- We can classify the elements into four blocks viz., s-block, p-block, d-block and f-block depending on the type of atomic orbital that are being filled with electrons.
- s-Block Elements : The elements of Group 1 (alkali metals) and Group 2 (alkaline earth metals) which have ns1 and ns2 outermost electronic configuration belong to the s-Block Elements.
- p-Block Elements The p-Block Elements comprise those belonging to Group 13 to 18 and these together with the s-Block Elements are called the Representative Elements or Main Group Elements. The outermost electronic configuration varies from ns2np1 to ns2np6 in each period.
- d-Block Elements These are the elements of Group 3 to 12 in the centre of the Periodic Table. These are characterised by the filling of inner d orbitals by electrons and are therefore referred to as d-Block Elements. These elements have the general outer electronic configuration (n-1)d1-10 ns0-2.
- f-Block Elements The two rows of elements at the bottom of the Periodic Table, called the Lanthanoids, Ce(Z = 58) - Lu(Z = 71) and Actinoids, Th(Z = 90) - Lr (Z = 103) are characterised by the outer electronic configuration (n-2)f1-14 (n-1)d0-1 ns2. The last electron added to each element is filled in f- orbital. These two series of elements are hence called the Inner-Transition Elements (f-Block Elements).
- Variation in Atomic Radius in Period : The atomic size generally decreases across a period It is because within the period the outer electrons are in the same valence shell and the effective nuclear charge increases as the atomic number increases resulting in the increased attraction of electrons to the nucleus.
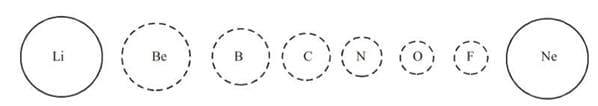
- Variation in Atomic Radius in Group: Within a family or vertical column of the periodic table, the atomic radius increases regularly with atomic number as), as we descend the groups, the principal quantum number (n) increases and the valence electrons are farther from the nucleus. This happens because the inner energy levels are filled with electrons, which serve to shield the outer electrons from the pull of the nucleus. Consequently the size of the atom increases as reflected in the atomic radii.
- The atomic radii of noble gases are not considered here. Being monatomic, their (non-bonded radii) values are very large. In fact radii of noble gases should be compared not with the covalent radii but with the van der Waals radii of other elements.
- A cation is smaller than its parent atom because it has fewer electrons while its nuclear charge remains the same. The size of an anion will be larger than that of the parent atom because the addition of one or more electrons would result in increased repulsion among the electrons and a decrease in effective nuclear charge. For example, the ionic radius of fluoride ion (F-) is 136 pm whereas the atomic radius of fluorine is only 64 pm. On the other hand, the atomic radius of sodium is 186 pm compared to the ionic radius of 95 pm for Na+.
- Isoelectronic species : Atoms and ions which contain the same number of electrons.. For example, O2-, F-, Na+ and Mg2+ have the same number of electrons (10). Their radii would be different because of their different nuclear charges. The cation with the greater positive charge will have a smaller radius because of the greater attraction of the electrons to the nucleus. Anion with the greater negative charge will have the larger radius. In this case, the net repulsion of the electrons will outweigh the nuclear charge and the ion will expand in size.
- Ionization Enthalpy: It represents the energy required to remove an electron from an isolated gaseous atom (X) in its ground state. In other words, the first ionization enthalpy for an element X is the enthalpy change (ΔH) for the reaction depicted in equation. X (g) → X+(g) + e-. The ionization enthalpy is expressed in units of kJ mol-1. We can define the second ionization enthalpy as the energy required to remove the second most loosely bound electron; it is the energy required to carry out the reaction shown in equation X+(g) → X2+(g) + e-. Energy is always required to remove electrons from an atom and hence ionization enthalpies are always positive. The second ionization enthalpy will be higher than the first ionization enthalpy because it is more difficult to remove an electron from a positively charged ion than from a neutral atom.
The document Revision Notes: Classification of Elements & Periodicity in Properties is a part of the JEE Course Chemistry for JEE Main & Advanced.
All you need of JEE at this link: JEE
FAQs on Revision Notes: Classification of Elements & Periodicity in Properties
| 1. What is the significance of the periodic table in the classification of elements? |  |
Ans. The periodic table is a systematic arrangement of elements based on their atomic number, electron configurations, and recurring chemical properties. It allows scientists to understand the relationships between elements, predict their behaviours, and identify trends in properties such as atomic size, ionisation energy, and electronegativity across periods and groups.
| 2. How does the concept of periodicity affect the properties of elements? |  |
Ans. Periodicity refers to the recurring trends that are observed in the properties of elements as one moves across a period or down a group in the periodic table. For instance, atomic radius tends to decrease across a period due to increased nuclear charge, while it increases down a group due to added electron shells. Similarly, ionisation energy generally increases across a period and decreases down a group, influencing the reactivity of elements.
| 3. What are the main categories of elements in the periodic table? |  |
Ans. The main categories of elements in the periodic table include metals, non-metals, and metalloids. Metals, found on the left side and in the centre, are typically good conductors of heat and electricity, malleable, and ductile. Non-metals, located on the right side, are usually poor conductors and can be gases, liquids, or solids at room temperature. Metalloids exhibit properties intermediate between metals and non-metals and are found along the zig-zag line separating the two categories.
| 4. What trends are observed in ionisation energy across periods and down groups? |  |
Ans. Ionisation energy is the energy required to remove an electron from an atom in its gaseous state. Across a period, ionisation energy generally increases due to a greater nuclear charge that holds the electrons more tightly. Conversely, down a group, ionisation energy decreases because the outermost electrons are further from the nucleus and experience increased shielding from inner electrons, making them easier to remove.
| 5. How do electronegativity values change across the periodic table? |  |
Ans. Electronegativity is the tendency of an atom to attract electrons towards itself in a chemical bond. It generally increases across a period from left to right due to increasing nuclear charge and a decreasing atomic radius. However, it decreases down a group as the increased number of electron shells results in greater distance between the nucleus and the bonding electrons, leading to a weaker attraction.
Related Searches
Extra Questions, practice quizzes, Exam, pdf , study material, Previous Year Questions with Solutions, Revision Notes: Classification of Elements & Periodicity in Properties, past year papers, MCQs, Viva Questions, Summary, ppt, Revision Notes: Classification of Elements & Periodicity in Properties, video lectures, shortcuts and tricks, Objective type Questions, Free, Revision Notes: Classification of Elements & Periodicity in Properties, Sample Paper, Important questions, mock tests for examination, Semester Notes;





