Revision Notes: Solutions
Introduction
In normal life we rarely come across pure substances. Most of these are mixtures containing two or more pure substances. Their utility or importance in life depends on their composition. The air around us is a mixture of gases primarily oxygen and nitrogen; the water we drink contains very small amounts of various salts dissolved in it. Our blood is a mixture of different components. Alloys such as brass, bronze, stainless steel, etc. are also mixtures. In this Unit, we will consider mostly liquid solutions and their properties.
Solutions
Definition
A solution is a homogeneous mixture of two or more than two components. For example, common salt in water.
Classification
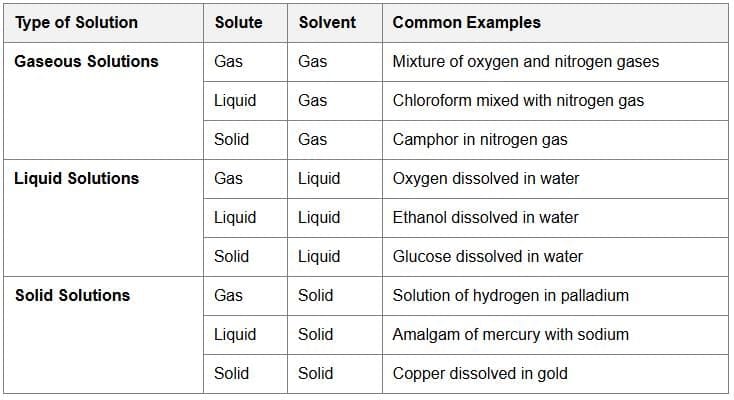
Solutions which contain two components in it are called Binary Solutions.
Substances which are used to prepare a solution are called as Components.
The component that is present in the largest quantity is known as Solvent. Solvent determines the physical state in which solution exists.
The other component present in lesser quantity in the solution is termed as Solute.
Each component may be solid, liquid or in gaseous state.
Strength of Solutions
The amount of solute dissolved per unit solution or solvent is called Strength of solution. There are various methods of measuring strength of a solution. :
- Mass Percentage (%w/w):
"It represents mass of a component present in 100 g of solution"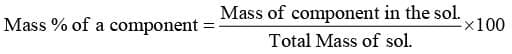
- Volume percentage (%v/v):
"It represents volume of a component in 100 mL of solution"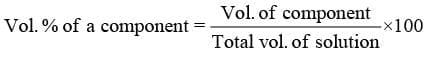
- Mass by volume percentage (%w/v):
"It represents mass of solute in grams present in 100 mL of solution"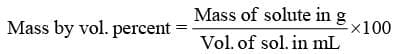
- Parts per Million (ppm)
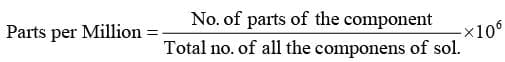
Concentration in parts per million can be expressed as mass to mass, volume to volume and mass to volume.
- Mole Fraction (x)
"It represents the moles of a solute present in one mole of solution"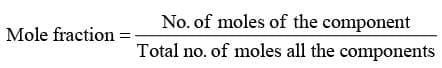
For example, in a binary mixture, if the number of moles of A and B are nA and nB respectively, the mole fraction of A will be
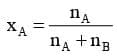
- Molarity, M
"It represents moles of solute present in 1 L of solution"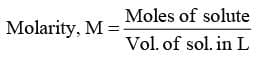
Units of Molarity are mol/L also represented by 'M' or 'Molar'.
"Density of a solution is mass of the solution per unit volume"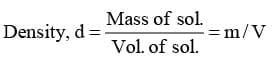
- Molality, m
"It represents moles of solute present per kg of solvent"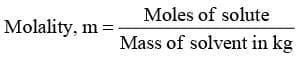
Units of molality are mol/kg which is also represented by 'm' or 'molal'.
- Normality, N
It represents no. of equivalents of solute present in 1 L of solution.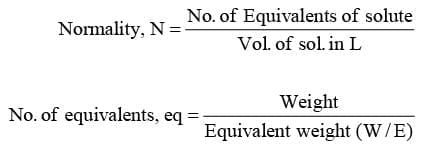 E = M/z (z is the valency factor)
E = M/z (z is the valency factor)
Some Important Relationships
- Dilution Law: If a solution is diluted by adding solvent to it, then the amount of solute remains constant and we can write:
M1V1 = M2V2 and N1V1 = N2V2 - Molarity and Normality
Normality = z x Molarity
IMPORTANT:
Mass %, ppm, mole fraction and molality are independent of temperature, whereas molarity & normality are a function of temperature. This is because volume depends on temperature and the mass does not.
Vapour Pressure
Definition
Vapour pressure of a liquid/solution is the pressure exerted by the vapours in equilibrium with the liquid/solution at a particular temperature. Vapour pressure ∝ escaping tendency
Vapour pressure of liquid solutions and Raoult's Law
(Raoult's law for volatile solutes)
Raoult's law states that for a solution of volatile liquids, the partial vapour pressure of each component in the solution is directly proportional to its mole fraction.
Consider a solution containing two volatile components 1 and 2 with mole fractions x1 and x2 respectively. Suppose at a particular temperature, their partial vapour pressures are p1 and p2 and the vapour pressure in pure state are 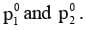
Thus, according to Raoult's Law, for component 1
Similarly, for component 2
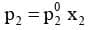
According to Dalton's law of partial pressure, the total pressure (ptotal) over the solution phase in the container will be the sum of the partial pressures of the components of the solution and is given as :
ptotal = p1 + p2
Substituting the values of p1 and p2, we get
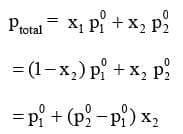
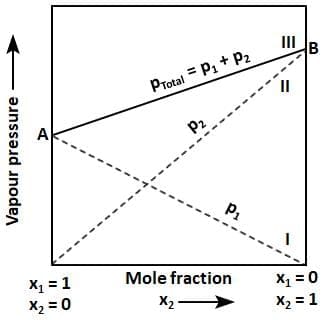
The plot of vapour pressure and mole fraction of an ideal solution at constant temperature. The dashed line I and II represent the partial pressure of the components. It can be seen from the plot that p1 and p2 are directly proportional to x1 and x2, respectively. The total vapour pressure is given by line marked III in the figure.
Mole fraction in vapour phase:
If y1 and y2 are the mole fractions of the components 1 and 2 respectively in the vapour phase then, using Dalton's law of partial pressures:
p1 = y1 ptotal
p2 = y2 ptotal
In general
pi = yi ptotal
Vapour pressures of solutions of solids in liquids and Raoult's Law:
(Raoult's law for non volatile solutes)
If a non-volatile solute is added to a solvent to give a solution, the number of solvent molecules escaping from the surface is correspondingly reduced, thus, the vapour pressure is also reduced.
The decrease in the vapour pressure of solvent depends on the quantity of non-volatile solute present in the solution, irrespective of its nature.
Raoult's law in its general form can be stated as, for any solution the partial vapour pressure of each volatile component in the solution is directly proportional to its mole fraction.
In a binary solution, let us denote the solvent by 1 and solute by 2. When the solute is non-volatile, only the solvent molecules are present in vapour phase and contribute to vapour pressure. Let p1 be the vapour pressure of the solvent, x1 be its mole fraction, p01 be its vapour pressure in the pure state. Then according to Raoult's law
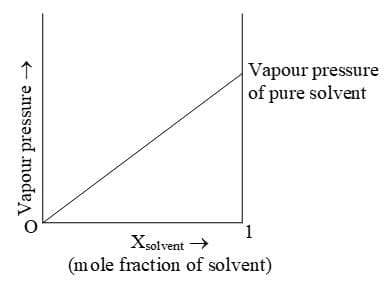
If a solution obeys Raoult's law for all concentrations, its vapour pressure would vary linearly from zero to the vapour pressure of the pure solvent.
Ideal and Non-ideal solutions
Ideal solutions: An ideal solution is the solution in which each component obeys Raoult's law under all conditions of temperatures and concentrations.
Properties of Ideal solutions :
- ΔHMIXING = 0
- ΔVMIXING = 0
- Intermolecular attractive forces between the A-A and B-B are nearly equal to those between A-B.
Eg. solution of benzene and toluene, solution of n-hexane and n-heptane
Non - ideal solutions: When a solution does not obey Raoult's law over the entire range of concentration, then it is called non-ideal solution.
Solutions showing positive deviation from Raoult's Law:
- Solvent-Solute(A-B) type of force is weaker than Solute-Solute(B-B) & Solvent-Solvent(A-A) forces.
- The vapour pressure is higher than predicted by the law.
- ΔHMIXING > 0
- ΔVMIXING > 0
Eg. ethanol and acetone, carbon disulphide and acetone
Pressure composition curve for solution showing positive deviation 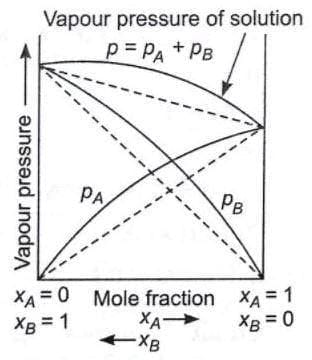
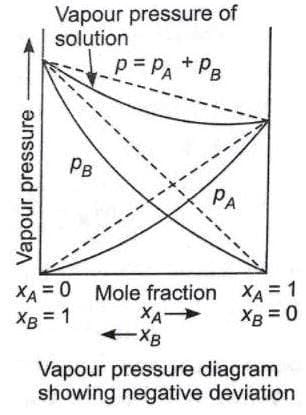
Solutions showing negative deviations from Raoult's law :
- Solvent-Solute(A-B) type of force is stronger than the other two.
- The vapour pressure is lower than predicted by the law
- ΔHMIXING < 0
- ΔVMIXING < 0
For example, phenol and aniline, chloroform and acetone etc.
Pressure composition curves for solution showing negative deviation
Azeotropes: Azeotropes are binary mixtures having the same composition in liquid and vapour phase and boil at a constant temperature.
Minimum boiling azeotrope
The solutions which show a large positive deviation from Raoult's law form minimum boiling azeotrope at a specific composition. For example, ethanol-water mixture containing approximately 95% of ethanol forms an azeotrope with boiling point 351.15 K.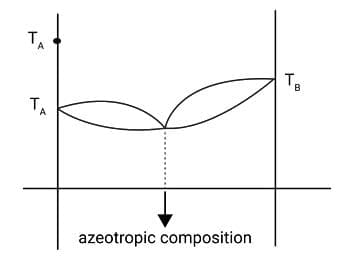
Boiling temperature - composition Diagram for solutions showing large positive deviations.
(Minimum boiling azeotrope)
Maximum boiling azeotrope: The solutions that show large negative deviation from Raoult's law form maximum boiling azeotrope at a specific composition. Nitric acid and water mixture containing 68% nitric acid forms an azeotrope with a boiling point of 393.5 K.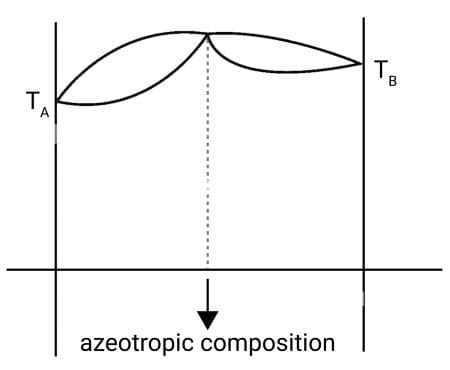
Boiling temperature - composition Diagram for solutions showing large negative deviations.
(Maximum boiling azeotrope)
Solubility
Solubility of a solid in liquid
Solubility of a substance is its maximum amount that can be dissolved in a specified amount of solvent.
Factors affecting the solubility of a solid in liquid :
- Nature of solute and solvent: Like dissolves like. For example, While sodium chloride and sugar dissolve readily in water, naphthalene and anthracene do not. On the other hand, naphthalene and anthracene dissolve readily in benzene but sodium chloride and sugar do not.
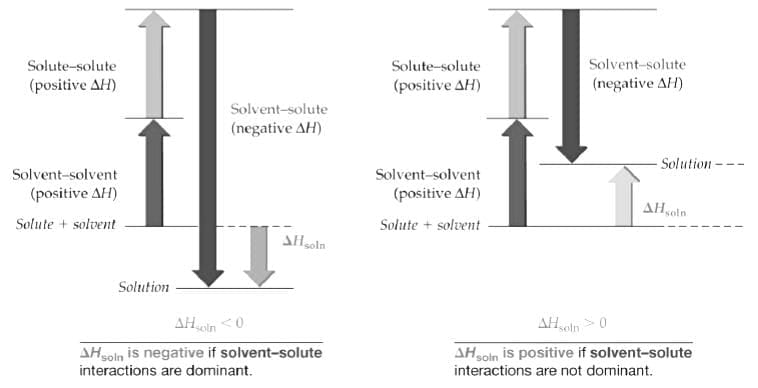
- Temperature: In a nearly saturated solution, If (Δsol H > 0), the solubility increases with rise in temperature and If (Δsol H < 0) the solubility decreases with rise in temperature.
Effect of pressure: Does not have any significant effect as solids and liquids are highly incompressible.
Henry's law: Henry's law states that at a constant temperature, the solubility of a gas in a liquid is directly proportional to the pressure of the gas.
The most commonly used form of Henry's law states that "the partial pressure of the gas in vapour phase (p) is proportional to the mole fraction of the gas (x) in the solution". This is expressed as:
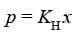
Here KH is the Henry's law constant.
Characteristics of KH:
- KH is a function of the nature of the gas.
- Higher the value of KH at a given pressure, the lower is the solubility of the gas in the liquid.
- KH values increase with increase of temperature indicating that the solubility of gases increases with decrease of temperature.
Applications of Henry's law:
- In the production of carbonated beverages.
- In the deep sea diving.
- For climbers or people at high altitudes.
Raoult's Law as a special case of Henry's Law
According to Raoult's law,
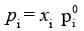
In the solution of a gas in a liquid, one of the components is so volatile that it exists as a gas. Its solubility according to Henry's law,
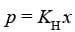
Thus, Raoult's law becomes a special case of Henry's law in which K_H becomes equal to p^0_i .
Colligative Properties
The properties that depend on the number of solute particles irrespective of their nature relative to the total number of particles present in the solution are called colligative properties.
There are four colligative properties:
- Relative Lowering of vapour Pressure
- Elevation in Boiling Point
- Depression in freezing point
- Osmotic pressure
Relative Lowering of vapour Pressure: When a non-volatile solute is added to a solvent, the vapour pressure decreases.
The lowering of vapour pressure w.r.t. the vapour pressure of the pure solvent is called "Relative lowering in vapour pressure".
According to Raoult's Law :
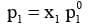
The reduction in the vapour pressure of solvent (Δp1) is given as:
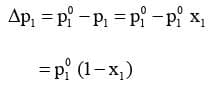
Knowing that x2 = 1 - x1, equation reduces to
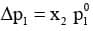
Equation can be written as
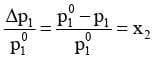
The expression on the left hand side of the equation as mentioned earlier is called relative lowering of vapour pressure and is equal to the mole fraction of the solute. The above equation can be written as :
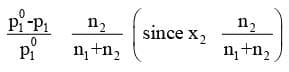
Here n1 and n2 are the number of moles of solvent and solute respectively present in the solution. For dilute solutions n2 << n1, hence neglecting n2 in the denominator we have
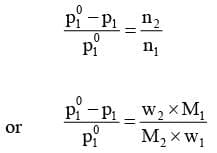
Here w1 and w2 are the masses and M1 and M2 are the molar masses of the solvent and solute respectively.
Elevation in Boiling Point: Boiling point of a liquid is the temperature at which the vapour pressure of the liquid becomes equal to the atmospheric pressure.
On addition of non-volatile solute the vapour pressure of the solvent decreases and therefore, to boil the solution the required temperature will be higher. So, there will be a rise in the boiling point of the solution.
The increase in boiling point 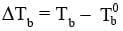 where T0b is the boiling point of pure solvent and Tb is the boiling point of solution is known as elevation of boiling point.
where T0b is the boiling point of pure solvent and Tb is the boiling point of solution is known as elevation of boiling point.
Expression:
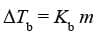
Kb is called Boiling Point Elevation Constant or Molal Elevation Constant (Ebullioscopic Constant).
Calculation of molar mass of solute:
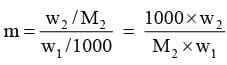
Substituting the value of molality in equation we get
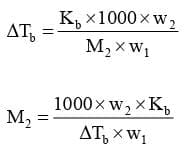
Kb: It is defined as the elevation in boiling point when the molality of the solution is unity.
The unit of Kb is K kg mol-1
Determination of Kb:
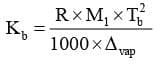
where: R = gas constant (8.314 JK/mol),
Tb = boiling temperature in K,
M1 = Molar mass of solvent in Kg/mol,
Δvap H = enthalpy of vapourisation of solvent in J/mol.
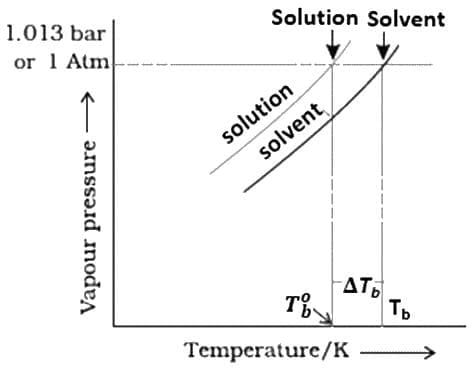 The vapour pressure curve for solution lies below the curve for pure water. The diagram shows that \Delta T_b denotes the elevation of boiling point of a solvent in solution.
The vapour pressure curve for solution lies below the curve for pure water. The diagram shows that \Delta T_b denotes the elevation of boiling point of a solvent in solution.
Depression in freezing point: The freezing point of a substance may be defined as the temperature at which the vapour pressure of the substance in its liquid phase is equal to its vapour pressure in the solid phase.
When a non-volatile solid is added to the solvent its vapour pressure decreases and now it would become equal to that of solid solvent at lower temperature. Thus, the freezing point of the solvent decreases.
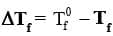 where T0f is the freezing point of pure solvent and Tf is its freezing point when non-volatile solute is dissolved is known as depression in freezing point.
where T0f is the freezing point of pure solvent and Tf is its freezing point when non-volatile solute is dissolved is known as depression in freezing point.Expression:
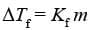
Kf is known as Freezing Point Depression Constant or Molal Depression Constant or Cryoscopic Constant.
Calculation of molar mass of solute :
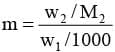
Substituting this value of molality in equation we get :
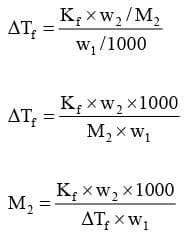
Kf: It is defined as the depression in freezing point when the molality of the solution is unity.
The unit of Kf is K kg mol-1.
Determination of Kf:
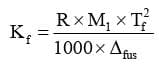
where : R = gas constant (8.314 JK/mol),
Tf = freezing temperature in K,
M1 = Molar mass of solvent in Kg/mol,
Δfus H= enthalpy of fusion of solvent in J/kg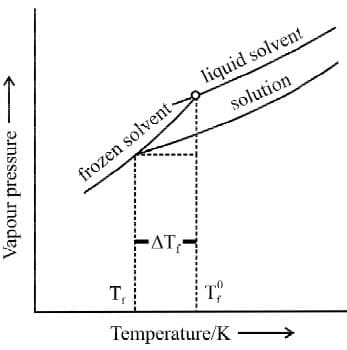 Diagram showing ΔTf, depression of the freezing point of a solvent in a solution.
Diagram showing ΔTf, depression of the freezing point of a solvent in a solution.
Osmosis
When a pure solvent and solution are kept with a semi-permeable membrane between them then the solvent particles pass through the membrane from the solvent side to the solution side. This phenomenon is called "Osmosis".
The semi-permeable membrane is a membrane that allows only small molecules to pass through and blocks the larger solute molecules.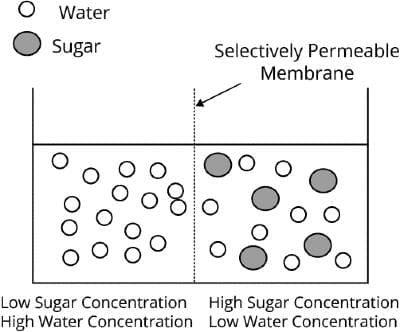
Osmotic pressure: The osmotic pressure of a solution is the excess pressure that must be applied to a solution to prevent osmosis, i.e., to stop the passage of solvent molecules through a semipermeable membrane into the solution.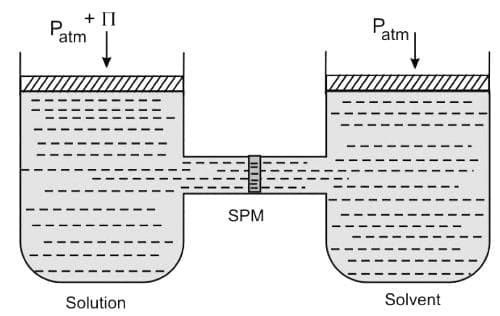 The excess pressure equal to osmotic pressure must be applied on the solution to prevent osmosis.
The excess pressure equal to osmotic pressure must be applied on the solution to prevent osmosis.
Expression:
For dilute solutions, osmotic pressure is proportional to the molarity, C of the solution at a given temperature T.
Thus:
π = C R T
Calculation of molar mass:
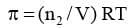
Here V is volume of a solution in litres containing n2 moles of solute. If w2 grams of solute, of molar mass, M2 is present in the solution, then n2 = w2 / M2 and we can write,
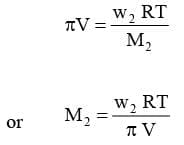
Isotonic solutions: Two solutions having same osmotic pressure at a given temperature are called isotonic solutions.
The solution with lower concentration or lower osmotic pressure is known as "Hypotonic" with respect to more concentrated solution.
The solution with higher concentration or higher osmotic pressure is known as "Hypertonic" with respect to dilute solution.
Reverse osmosis :
If a pressure larger than the osmotic pressure is applied to the solution side, the solvent will flow from the solution into the pure solvent through the semi permeable membrane. This phenomenon is called reverse osmosis.
Application:
Desalination of sea water: When pressure more than osmotic pressure is applied, pure water is squeezed out of the sea water through the membrane.Abnormal Molar Masses: When the molecular mass of a substance determined by studying any of the colligative properties comes out to be different than the theoretically expected value, the substance is said to show abnormal molar mass.
Abnormal Molar Masses are observed:
- When the solute undergoes association in the solution.
- When the solute undergoes dissociation in the solution.
van't Hoff Factor:
To calculate extent of association or dissociation, van't Hoff introduced a factor i, known as the van't Hoff Factor.

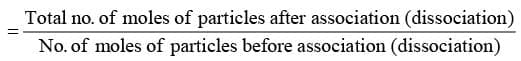
Association: Number of particles will always decrease due to association therefore i < 1.
Let initial particles (ni) = 1
Final number( nf) = 1 - α + α/n
van't Hoff factor, i = nf/ni = 1 - α + α/n
Dissociation: The number of particles will always increase due to dissociation and hence i > 1.
Initial particle = 1
Final particles = 1 - α + nα
i = 1 - α + nα
Modified Expressions:
Relative lowering of vapour pressure of solvent,
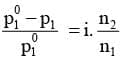
Elevation of Boiling point ΔTb = i Kb m
Depression of Freezing point, ΔTf = i Kf m
Osmotic pressure of solution, π\pi = i n2 RT/ V
Vapour Pressure
On increasing the temperature of the liquid the escaping tendency of the molecules increases and the vapour pressure increases.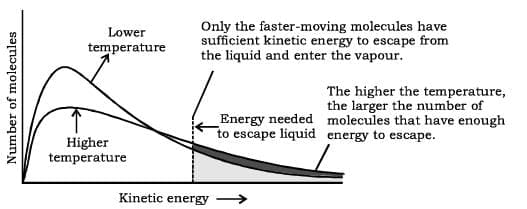
The distribution of molecular kinetic energies in a liquid
Claussius-Clapeyron Equation:
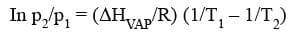
where ΔHVAP represents the enthalpy of vaporisation of the liquid.
Thermodynamics of Dissolution
If the interactions grow stronger the process is exothermic and if they go weaker during the formation of solution the process becomes endothermic. In general \Delta S is positive in dissolution process. If the mixing process is spontaneous/natural then ΔG has to be negative.
Boiling Point Elevation
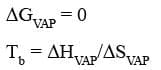
The non-volatile solute increases the randomness of the solution phase and the entropy of the vapours remains the same. Due to this, ΔSVAP decreases thus giving rise to the boiling point.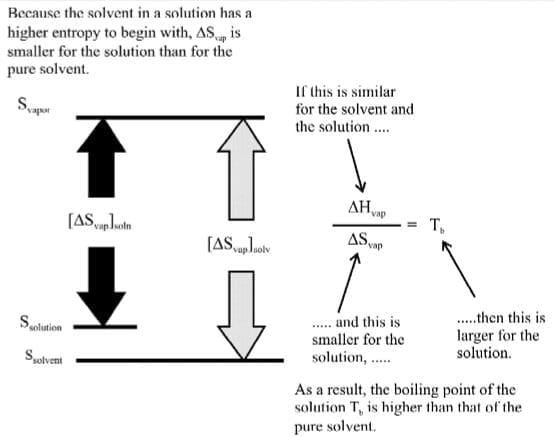
The higher boiling point of a solution relative to that of a pure solvent is due to a difference in their entropies of vapourization, ΔSvap.
Freezing Point Depression
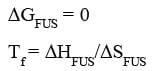
The entropy difference will increase in this case due to the increase in the entropy of solution. This increase in entropy will result in decrease of the freezing point according to the above relation.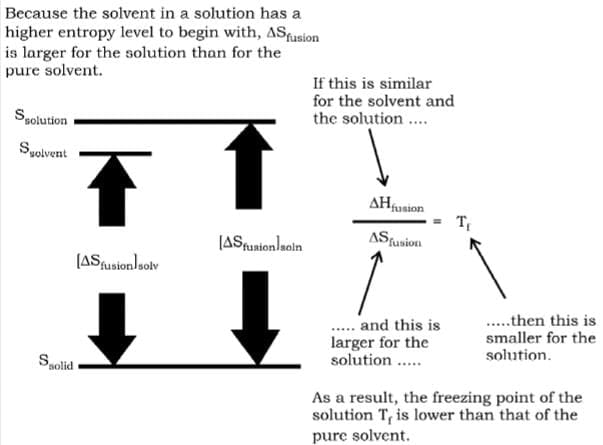
The lower freezing point of a solution relative to that of a pure solvent is due to a difference in their entropies of fusion, \ΔSfusion.
Ostwald Walker Method
This is a typical method to measure the relative lowering in vapour pressure of a solution. Dry air is passed successively through three systems: solution, pure solvent and then a drying agent.
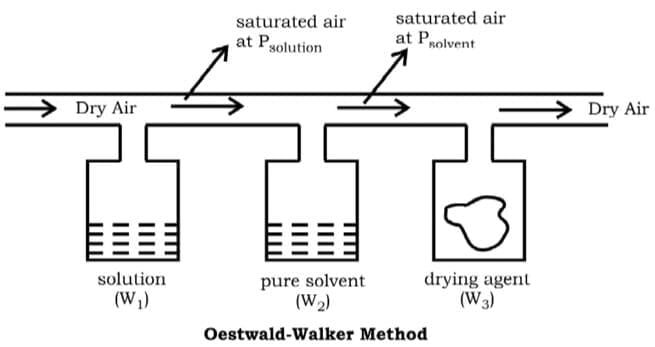
w1 and w2 represent the decrease in weight of the vessels and w3 represents the increase in weight of the third vessel due to absorption.
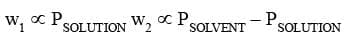
(as the air was already saturated)
w3 ∝ PSOLVENT
Using the above relations the relative lowering in vapour pressure can be calculated.
FAQs on Revision Notes: Solutions
| 1. What is vapour pressure in the context of solutions? |  |
| 2. How does solubility relate to solutions and colligative properties? |  |
| 3. What is the thermodynamics of dissolution? |  |
| 4. What is boiling point elevation and how does it occur? |  |
| 5. Can you explain freezing point depression? |  |






