Revision Notes: Electrochemistry
Introduction
Electrochemistry is the study of production of electricity from the energy released during a spontaneous chemical reaction and the use of electrical energy to bring about non-spontaneous chemical transformations.
Electrochemical Cells
A spontaneous chemical process is the one which can take place on its own and in such a process the Gibb's energy of the system decreases. It is this energy that gets converted to electrical energy. The reverse process is also possible in which we can make non-spontaneous processes occur by supplying external energy in the form of electrical energy. These inter conversions are carried out in equipments called Electrochemical Cells.
Types
Electrochemical Cells are of two types:
- Galvanic Cells: Converts chemical energy into electrical energy
- Electrolytic Cells: Converts electrical energy into chemical energy.
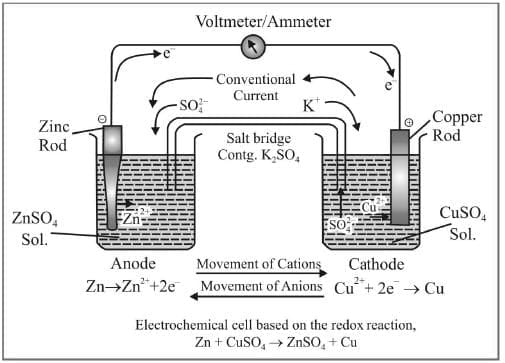
Galvanic Cell
Cell energy is extracted from a spontaneous chemical process or reaction and it is converted to electric current. For example, Daniell Cell is a Galvanic Cell in which Zinc and Copper are used for the redox reaction to take place.
Zn (s) + Cu2+ (aq) → Zn2+ (aq) + Cu(s)
Oxidation Half: Zn (s) → Zn2+ (aq) + 2e-
Reduction Half: Cu2+(aq) + 2e- → Cu(s)
Zn is the reducing agent and Cu2+ is the oxidising agent. The half cells are also known as Electrodes. The oxidation half is known as Anode and the reduction half is called Cathode. Electrons flow from anode to cathode in the external circuit. Anode is assigned negative polarity and cathode is assigned positive polarity. In Daniell Cell, Zn acts as the anode and Cu acts as the cathode.
Electrolytic Cell
These electrodes are dipped in and electrolytic solution containing cations and anions. On supplying current the ions move towards electrodes of opposite polarity and simultaneous reduction and oxidation takes place.
Preferential Discharge of ions
Where there are more than one cation or anion the process of discharge becomes competitive in nature. Discharge of any ion requires energy and in case of several ions being present the discharge of that ion will take place first which requires the energy.
Electrode Potential
It may be defined as the tendency of an element, when it is placed in contact with its own ions to either lose or gain electrons and in turn become positively or negatively charged. The electrode potential will be named as oxidation or reduction potential depending upon whether oxidation or reduction has taken place.

Characteristics
(a) Both oxidation and reduction potentials are equal in magnitude but opposite in sign.
(b) It is not a thermodynamic property, so values of E are not additive.
Standard Electrode Potential (E0)
It may be defined as the electrode potential of an electrode determined relative to standard hydrogen electrode under
standard conditions. The standard conditions taken are :
(i) 1M concentration of each ion in the solution.
(ii) A temperature of 298 K.
(iii) 1 bar pressure for each gas.
Electrochemical Series
The half cell potential values are standard values and are represented as the standard reduction potential values as shown in the table at the end which is also called Electrochemical Series.
Cell Potential or Emf of a Cell
The difference between the electrode potentials of two half cells is called cell potential. It is known as electromotive force (EMF) of the cell if no current is drawn from the cell.
Ecell = Ecathode + Eanode
For this equation we take oxidation potential of anode and reduction potential of cathode.
Since anode is put on left and cathode on right, it follows
therefore,
= ER + EL
For a Daniel cell, therefore
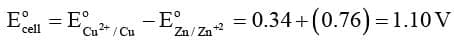
Cell Diagram or Representation of a Cell
The following conventions or notations are applied for writing the cell diagram in accordance with IUPAC recommendations.
The Daniel cell is represented as follows :
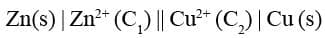
(a) Anode half cell is written on the left hand side while cathode half cell on right hand side.
(b) A single vertical line separates the metal from aqueous solution of its own ions.

(c) A double vertical line represents salt bridge
(d) The molar concentration (C) is placed in brackets after the formula of the corresponding ion.
(e) The value of e.m.f. of the cell is written on the extreme right of the cell. For example,
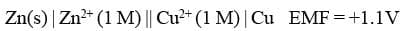
(f) If an inert electrode like platinum is involved in the construction of the cell, it may be written along with the working electrode in bracket say for example, when a zinc anode is connected to a hydrogen electrode.
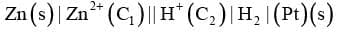
Salt Bridge
Salt bridge is used to maintain the charge balance and to complete the circuit by facilitating the flow of ions through it. It contains a gel in which an inert electrolyte like Na2SO4 or KNO3 etc. are mixed. Negative ions flow to the anode and positive ions flow to the cathode through the salt bridge and charge balance is maintained and cell keeps on functioning.
Spontaneity of a Reaction
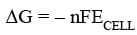
For a spontaneous cell reaction ΔG should be negative and cell potential should be positive.
If we take standard value of cell potential in the above equation we will obtain standard value of ΔG as well.
Types of Electrodes
Metal-Metal Ion Electrodes: metal rod/plate is dipped in an electrolyte solution containing metal ions. There is a potential difference between these two phases and this electrode can act as a cathode or anode both.
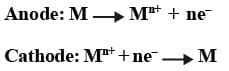
Gas Electrodes: Electrode gases like H2, Cl2 etc are used with their respective ions. For example, H2 gas is used with a dilute solution of HCl (H+ ions). The metal should be inert so that it does not react with the acid.
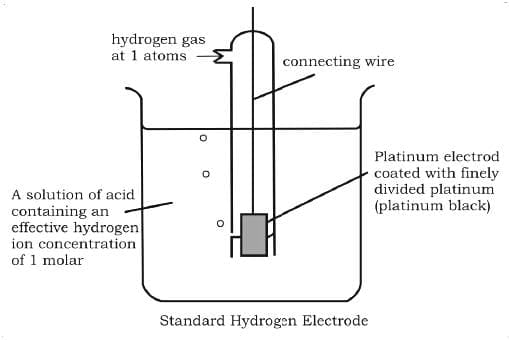
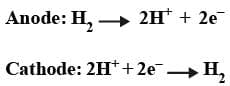
The hydrogen electrode is also used as the standard to measure other electrode potentials. Its own potential is set to 0 V as a reference. When it is used as a reference the concentration of dil. HCl is taken as 1 M and the electrode is called "Standard Hydrogen Electrode (SHE)".
Metal-Insoluble salt electrode: We use salts of some metals which are sparingly soluble with the metal itself as electrodes. For example, if we use AgCl with Ag there is a potential gap between these two phases which can be identified in the following reaction:
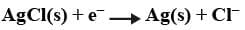
This electrode is made by dipping a silver rod in a solution containing AgCl(s) and Cl- ions.
Calomel Electrode: Mercury is used with two other phases, one is a calomel paste (Hg2Cl2) and electrolyte containing Cl- ions.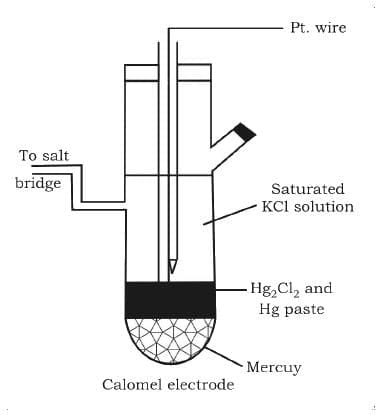
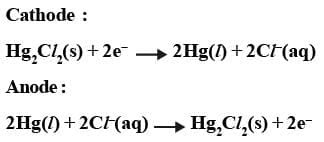
This electrode is also used as another standard to measure other potentials. Its standard form is also called Standard Calomel Electrode (SCE).
Redox Electrode: In these electrodes two different oxidation states of the same metal are used in the same half cell. For example,
Fe2+ and Fe3+ are dissolved in the same container and an inert electrode of platinum is used for the electron transfer.
Following reactions can take place:
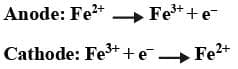
Nernst Equation
It relates electrode potential with the concentration of ions. Thus, the reduction potential increases with the increase in the concentration of ions. For a general electrochemical reaction of the type.

Nernst equation can be given as
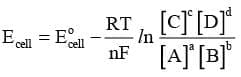
Substituting the values of R and F we get
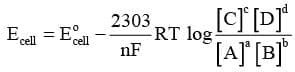
Applications of Nernst Equation
Equilibrium Constant from Nernst Equation:
For a Daniel cell, at equilibrium
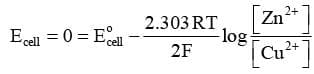
or
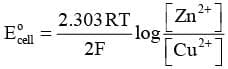
But at equilibrium,
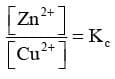

In general,
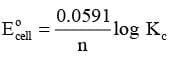
or,
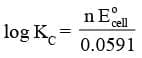
Concentration Cells
If two electrodes of the same metal are dipped separately into two solutions of the same electrolyte having different concentrations and the solutions are connected through salt bridge, such cells are known as concentration cells. For example
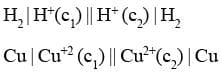
These are of two types :
1. Electrode concentration cells:
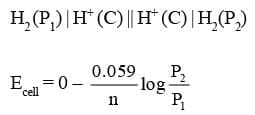
where P2 > P1 for spontaneous reaction
2. Electrolyte concentration cell
The EMF of concentration cell at 298 K is given by
Zn | Zn²⁺ (c₁) || Zn²⁺ (c₂) | Zn
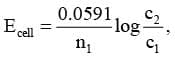
where c2 > c1 for spontaneous reaction
Cases of Electrolysis
Electrolysis of molten sodium chloride:
2NaCl (l) → 2Na⁺ (l) + 2Cl⁻ (l)
The reactions occurring at the two electrodes may be shown as follows :
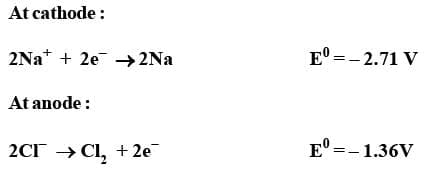
Overall reaction :
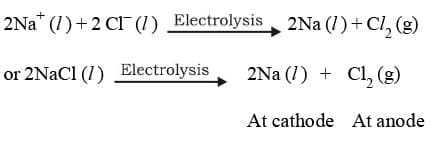
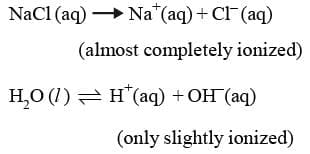
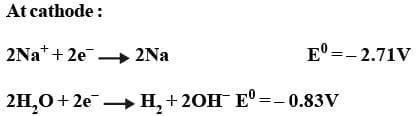
Thus H₂ gas is evolved at cathode value Na⁺ ions remain in solution.
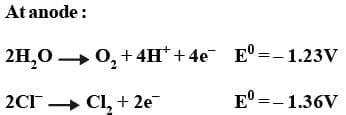
Thus, Cl₂ gas is evolved at the anode by over voltage concept while OH⁻ ions remain in the solution.
Batteries
When Galvanic cells are connected in series to obtain a higher voltage the arrangement is called Battery.
Primary Batteries
Primary cells are those which can be used so long the active materials are present. Once they get consumed the cell will stop functioning and cannot be re-used. Example Dry Cell or Leclanche cell and Mercury cell.
Dry cell
Anode: Zn container
Cathode: Carbon (graphite) rod surrounded by powdered MnO₂ and carbon.
Electrolyte: NH₄Cl and ZnCl₂
Reaction :
Anode: Zn → Zn²⁺ + 2e⁻
Cathode: MnO₂ + NH₄⁺ + e⁻ → MnO(OH) + NH₃
The standard potential of this cell is 1.5 V and it falls as the cell gets discharged continuously and once used it cannot be recharged.
Mercury cells
These are used in small equipments like watches, hearing aids.
- Anode: Zn - Hg Amalgam
Cathode: Paste of HgO and carbon
Electrolyte: Paste of KOH and ZnO
Anode: Zn (Hg) + 2OH⁻ → ZnO (s) + H₂O + 2e⁻
Cathode: HgO (s) + H₂O + 2e⁻ → Hg (l) + 2OH⁻
Overall Reaction: Zn (Hg) + HgO (s) → ZnO (s) + Hg (l)
The cell potential is approximately 1.35V and remains constant during its life.
Secondary Batteries
Secondary cells are those which can be recharged again and again for multiple uses. e.g. lead storage battery and Ni - Cd battery.
Lead Storage Battery
- Anode: Lead (Pb)
- Cathode: Grid of lead packed with lead oxide (PbO₂)
- Electrolyte: 38% solution of H₂SO₄
- Discharging Reactions
- Anode: Pb(s) + SO₄²⁻(aq) → PbSO₄(s) + 2e⁻
- Cathode: PbO₂(s) + 4H⁺(aq) + SO₄²⁻(aq) + 2e⁻ → PbSO₄(s) + 2H₂O(l)
- Overall Reaction : Pb(s) + PbO₂(s) + 2H₂SO₄(aq) → 2PbSO₄(s) + 2H₂O(l)
To recharge the cell, it is connected with a cell of higher potential and this cell behaves as an electrolytic cell and the reactions are reversed. Pb(s) and PbO₂(s) are regenerated at the respective electrodes. These cells deliver an almost consistent voltage.
Recharging Reaction: 2PbSO₄(s) + 2H₂O(l) → Pb(s) + PbO₂(s) + 2H₂SO₄(aq)
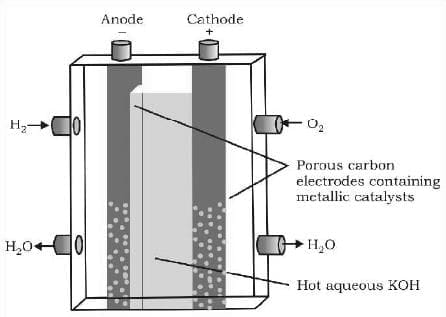
Fuel Cells
A fuel cell differs from an ordinary battery in the sense that the reactants are not contained inside the cell but are externally supplied from an external reservoir. Fuel cell is used in space vehicles and in this cell the two gases are supplied from external storages. In this cell carbon rods are used as electrodes with KOH as the electrolyte.
Cathode: O₂ (g) + 2H₂O (l) + 4e⁻ → 4OH⁻ (aq)
Anode: 2H₂ (g) + 4OH⁻ (aq) → 4H₂O (l) + 4e⁻
overall Reaction: 2H₂(g) + O₂ (g) → 2H₂O (l)
Corrosion
It involves a redox reaction and formation of an electrochemical cell on the surface of iron or any other metal.
At one location oxidation of iron takes place (anode) and at another location reduction of oxygen to form water takes place (cathode). First Fe gets oxidised to Fe²⁺ and then in the presence of oxygen it forms Fe³⁺ which then reacts with water to form rust which is represented by Fe₂O₃.xH₂O.
Anode: 2Fe (s) → 2 Fe²⁺ + 4e⁻ Eº = + 0.44 V
Cathode: O₂ (g) + 4H⁺ + 4e⁻ → 2H₂O (l) Eº = 1.23 V
Overall R × N: 2Fe (s) + O₂ (g) + 4H⁺ → 2Fe²⁺ + 2H₂O Eºcell = 1.67 V
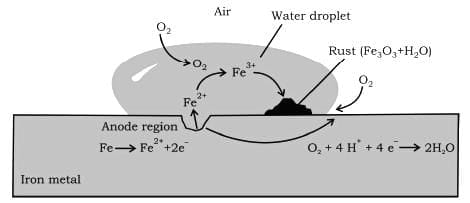
Rusting of iron can be avoided by painting it or by coating it with some other metals like Zinc. The latter process is known as Galvanisation. As the tendency of Zn to get oxidised is more than iron it gets oxidised in preference and iron is protected. This method of protecting one metal by the other is also called Cathodic Protection.
Conductance (G)
It is the reciprocal of resistance and may be defined as the ease with which the electric current flows through a conductor.
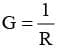
SI unit is Siemen (S).
1 S = 1 ohm⁻¹ (mho)
Conductivity (κ)
It is the reciprocal of resistivity (ρ).
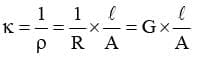
Now if ℓ = 1 cm and A = 1 cm², then к = G .
Hence, conductivity of an electrolytic solution may be defined as the conductance of a solution of 1 cm length with area of cross-section equal to 1 cm².
Factors Affecting Electrolytic Conductance
Electrolyte:
An electrolyte is a substance that dissociates in solution to produce ions and hence conducts electricity in dissolved or molten state.
Examples: HCl, NaOH, KCl (Strong electrolytes).
CH3-COOH, NH4OH (Weak electrolytes).
The conductance of electricity by ions present in the solutions is called electrolytic or ionic conductance. The following factors govern the flow of electricity through a solution of electrolyte.
(i) Nature of electrolyte or interionic attractions : Lesser the solute-solute interactions, greater will be the freedom of movement of ions and higher will be the conductance.
(ii) Solvation of Ions : Larger the magnitude of solute-solvent interactions, greater is the extent of solvation and lower will be the electrical conductance.
(iii) The nature of solvent and its viscosity : Larger the solvent-solvent interactions, larger will be viscosity and more will be the resistance offered by the solvent to flow of ions and hence lesser will be the electrical conductance.
(iv) Temperature : As the temperature of electrolytic solution rises solute-solute, solute-solvent and solvent-solvent interactions decreases, this results in the increase of electrolytic conductance.
Measurement of Conductance
As we know,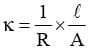 The value of к could be known, if we measure l, A and R . The value of the resistance of the solution R between two parallel electrodes is determined by using 'Wheatstones' bridge method.
The value of к could be known, if we measure l, A and R . The value of the resistance of the solution R between two parallel electrodes is determined by using 'Wheatstones' bridge method.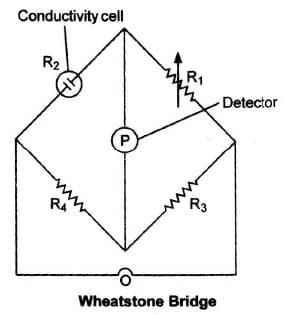
It consists of two fixed resistance R3 and R4, a variable resistance R1 and the conductivity cell having the unknown resistance R2. The bridge is balanced when no current passes through the detector. Under these conditions,
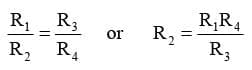
Molar Conductivity (Λm)
It may be defined as the conducting power of all the ions produced by dissolving one mole of an electrolyte placed between two large electrodes at one centimeter apart.
Mathematically,
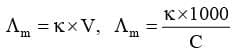
where, V is the volume of solution in cm³ containing 1 mole of electrolyte and C is the molar concentration.
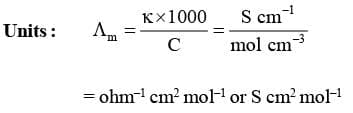
Equivalent Conductivity (Λeq)
It is conducting power of one equivalent of electrolyte placed between two large electrodes at one centimeter apart.
Mathematically :
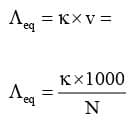
Where, v is the volume of solution in cm³ containing 1 equivalent of electrolyte and N is normality.
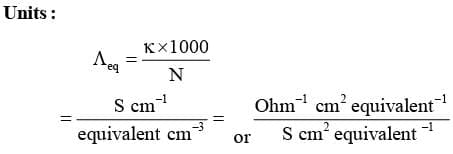
Variation of Conductivity and Molar Conductivity With Dilution
Conductivity decreases with decrease in concentration, this is because the number of ions per unit volume that carry the current in the solution decreases on dilution.
Molar conductivity 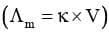 increases with decrease in concentration. This is because the total volume V of solution containing one mole of electrolyte also increases.
increases with decrease in concentration. This is because the total volume V of solution containing one mole of electrolyte also increases.
It has been found that the decrease in к on dilution of a solution is more than compensated by increases in its volume.
Graphic representation of the variation of 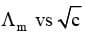
Limiting Molar Conductivity (Λom)
The value of molar conductivity when the concentration approaches zero is known as limiting molar conductivity or molar conductivity at infinite dilution. It is possible to determine the molar conductivity at infinite dilution 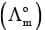 in case of strong electrolyte by extrapolation of curve of
in case of strong electrolyte by extrapolation of curve of 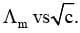 On contrary, the value of molar conductivity of weak electrolyte at infinite dilution cannot be determined by extrapolation of the curve as the curve becomes almost parallel to y-axis when concentration approaches to zero.
On contrary, the value of molar conductivity of weak electrolyte at infinite dilution cannot be determined by extrapolation of the curve as the curve becomes almost parallel to y-axis when concentration approaches to zero.
The mathematical relationship between 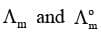 for strong electrolyte was developed by Debye, Huckel and Onsagar. In simplified form the equation can be given as
for strong electrolyte was developed by Debye, Huckel and Onsagar. In simplified form the equation can be given as
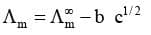
where 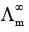 is the molar conductivity at infinite dilution and b is a constant which depends on the nature of the solvent and temperature.
is the molar conductivity at infinite dilution and b is a constant which depends on the nature of the solvent and temperature.
Kohlrausch's Law
It states that the limiting molar conductivity of an electrolyte can be represented as the sum of the individual contributions of the anion and cation of the electrolyte. In general, if an electrolyte on dissociation gives v+ cations and v- anions then its limiting molar conductivity is given by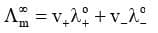
Here,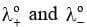 are the limiting molar conductivities of cations and anions respectively.
are the limiting molar conductivities of cations and anions respectively.
Applications of Kohlrausch's Law
(i) Calculation of molar conductivities of weak electrolyte at infinite dilution
For example, molar conductivity of acetic acid at infinite dilution can be obtained from the knowledge of molar conductivities at infinite dilution of strong electrolyte like HCl, CH₃COONa and NaCl as illustrated below.
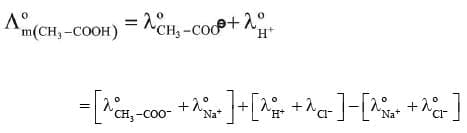
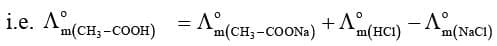
(ii) Determination of Degree of Dissociation of Weak Electrolytes
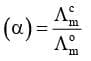
(iii) Determination of Dissociation Constant (K) of Weak Electrolytes:
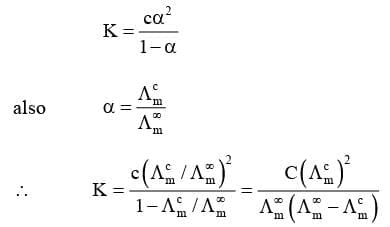
Use of Δg in Relating EMF Values of Half Cell Reactions
When we have two half cell reactions such that on adding them we obtain another half cell reaction then their emfs cannot be added directly. But in any case thermodynamic functions like ΔG can be added and emf values can be related through them. Consider the following three half cell reactions:
Fe²⁺ + 2e⁻ → Fe E₁
Fe³⁺ + 3e⁻ → Fe E₂
Fe³⁺ + e⁻ → Fe²⁺ E₃
We can easily observe that the third reaction can be obtained by subtracting the first reaction from the second. But the same relation does not apply on the emf values. That is, E₃ ≠ E₂ - E₁. But the ΔG values can be related according to the reactions. That is,
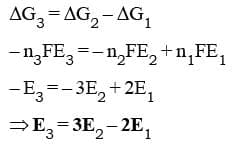
NOTE
We should always remember that emf values are additive only when two half cell reactions are added to give a complete balanced cell reaction. In any other case we will be using ΔG values to obtain relations between emf values.
FAQs on Revision Notes: Electrochemistry
| 1. What are the main types of electrochemical cells? |  |
| 2. What is a salt bridge and its function in electrochemical cells? |  |
| 3. How is standard electrode potential (E⁰) determined? |  |
| 4. What is the Nernst equation and its significance? |  |
| 5. What are concentration cells and how do they operate? |  |






