Equilibrium in Physical Processes
What is Equilibrium?
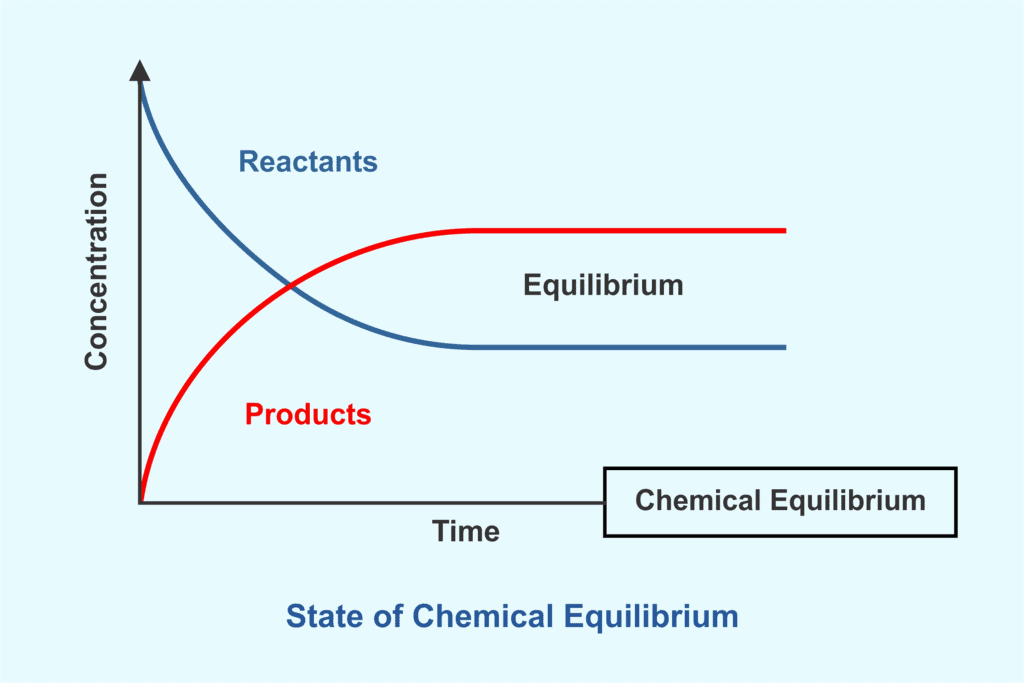
Equilibrium represents a state in a process where measurable properties such as pressure, temperature and concentration remain unchanged with time. It occurs when opposing processes or forces balance each other, producing a dynamic but stable condition in which microscopic changes continue to occur while macroscopic properties are constant.
Common Terms in Equilibrium
1. Equilibrium Mixture
An equilibrium mixture is a system in which both reactants and products are present in constant concentrations. This occurs in a reversible reaction where the forward and backward reactions proceed at the same rate.
2. Dynamic Equilibrium
Dynamic equilibrium describes a situation in which two opposing processes occur simultaneously and independently, producing a stable interchange between two states. The overall amounts of the species involved remain constant because the rates of the opposing processes are equal. A reversible process is commonly represented by a double-headed arrow:
A ⇌ B
3. Chemical Equilibrium
Chemical equilibrium is the state in which the rate of the forward chemical reaction equals the rate of the reverse reaction, so that the concentrations of reactants and products remain constant with time, although both reactions continue to occur.
4. Ionic Equilibrium
Ionic equilibrium occurs when an ionic compound dissociates into ions in a polar solvent and a balance establishes between the ions and the undissociated solute. In such systems the degree of dissociation and the concentrations of ions remain constant at equilibrium.
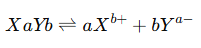
Equilibrium in Physical Processes (Physical Equilibrium)
Physical equilibrium is the equilibrium that develops between different phases or different physical states of a substance. In these processes the chemical composition of each phase does not change; only physical forms (phase, state, distribution) change or exchange.
Types of physical equilibria
1. Phase equilibrium - balance between different states of matter (solid, liquid, gas).
2. Solute-solid equilibrium - equilibrium between a solid and its saturated solution.
3. Gas-liquid equilibrium - balance between a gas and the gas dissolved in a liquid.
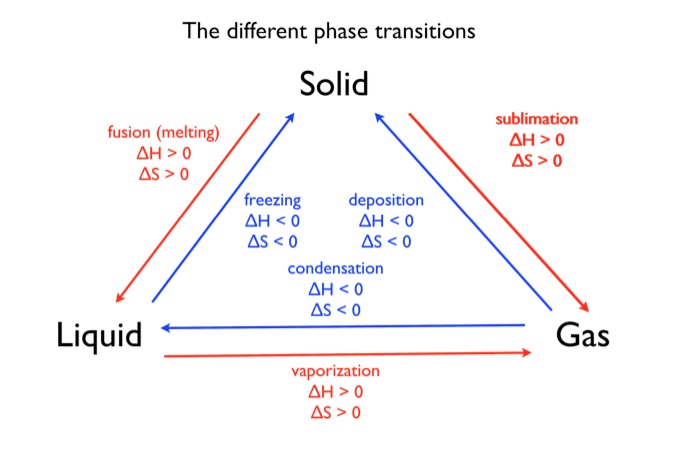
Substances undergo phase transformations such as:
- solid ⇌ liquid
- liquid ⇌ gas
- solid ⇌ gas
Solid-Liquid Equilibrium
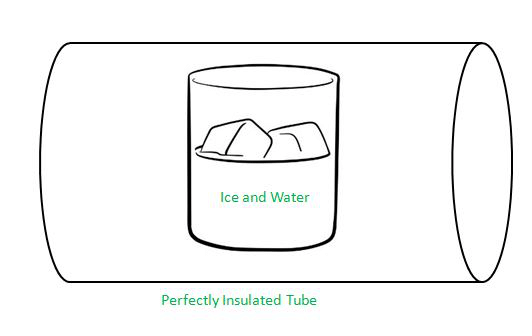
What happens if you keep ice and water in a perfectly insulated manner, such as in a thermos flask at a temperature of 273K and atmospheric pressure?
We see that the mass of ice and water do not change and that the temperature remains constant, indicating a state of equilibrium.
However, the equilibrium is not static because there is intense activity at the boundary between ice and water. Some ice molecules escape into liquid water and some molecules of water collide with ice and adhere to it. Despite this exchange, there is no change in the mass of ice and water. This is because the rates of transfer of ice molecules to water and the reverse process are equal at 273K and atmospheric pressure.
It is evident that ice and water are in equilibrium only at a particular pressure and temperature. Therefore,
For any pure substance at atmospheric pressure, the temperature at which the solid and liquid phases are at equilibrium is called the normal melting point or normal freezing point of the substance.
The system of ice and water is in dynamic equilibrium and we can conclude the following:
- Both opposing processes occur at the same time.
- The two processes occur at the same rate such that the amount of ice and water remains constant.
Liquid-Vapour Equilibrium

Liquid molecules at the surface may escape into the gaseous phase (evaporation) while gas molecules may collide with the surface and condense back to liquid. When the rate of evaporation equals the rate of condensation, the system attains liquid-vapour equilibrium.
Experiment (to demonstrate liquid-vapour equilibrium):
- Place a drying agent (for example anhydrous calcium chloride) for some hours in a transparent closed box fitted with a U-tube mercury manometer to remove moisture from the box.
- Remove the drying agent quickly and place a petri dish with water inside the box, then close the box immediately.
Observations:
- The mercury level in the manometer rises gradually and then reaches a constant value as water evaporates and the pressure of water vapour in the box increases.
- Initially there is no water vapour; evaporation reduces the liquid volume and raises vapour pressure, but as vapour density increases condensation also occurs.
- The rate of increase of vapour pressure falls with time because condensation begins to balance evaporation.
- Finally a steady state is reached where net evaporation = net condensation and the vapour pressure remains constant.
Conclusion:
- Equilibrium is reached when the rate of evaporation equals the rate of condensation; H2O(l) ⇌ H2O(g).
- At equilibrium the pressure exerted by the vapour at a given temperature is constant; this pressure is called the equilibrium vapour pressure of the liquid.
- The equilibrium vapour pressure of a liquid increases with increasing temperature.
Boiling point
The boiling point of a liquid is the temperature at which its equilibrium vapour pressure equals the external (usually atmospheric) pressure. A liquid with a higher vapour pressure at a given temperature is more volatile and has a lower boiling point than a liquid with lower vapour pressure.
Solid-Vapour Equilibrium
Some solids sublimate directly to vapour and the vapour may condense back to the solid. An example is iodine: when solid iodine is kept in a closed jar it sublimes to give violet vapour and, after some time, the colour intensity becomes constant as equilibrium is attained.
I2(s) ⇌ I2(g)
Other examples:
- Camphor (s) ⇌ Camphor (g)
- NH4Cl (s) ⇌ NH4Cl (g)
Try yourself: If both the forward and reverse reaction rates are equal in the equilibrium is said to be ____________
Equilibrium Involving Dissolution of Solids or Gases in Liquids
Solids in liquids
What happens when a concentrated sugar solution prepared at high temperature is allowed to cool to room temperature?
Crystals of sugar separate out. The cooled solution is a saturated solution at that temperature because no more solute can dissolve at that temperature. A dynamic equilibrium exists between the solute molecules in the solid phase and those in the solution:
Sugar (solution) ⇌ Sugar (solid)
At equilibrium the rate of dissolution equals the rate of crystallisation.
Thought experiment: add radioactive sugar to a saturated solution of non-radioactive sugar.
- Initially the solution contains no radioactive molecules, but because exchange between phases continues at equilibrium, radioactive molecules will appear both in solution and in the solid over time.
- The ratio of radioactive to non-radioactive sugar molecules in each phase changes until it reaches constant values consistent with dynamic equilibrium.
Gases in liquids
Opening a sealed bottle of a carbonated drink produces fizz and a hissing sound because CO2 dissolved under pressure escapes rapidly to reach a new equilibrium with the atmospheric partial pressure.
CO2(g) ⇌ CO2(in solution)

This gas-liquid equilibrium is described by Henry's law.
Henry's law states that, at a fixed temperature, the amount (or concentration) of a gas dissolved in a given volume (or mass) of a liquid is directly proportional to the partial pressure of the gas above the liquid. Mathematically, c = kH·p, where c is the concentration of the dissolved gas, p is the partial pressure of the gas and kH is Henry's constant. The solubility of most gases in liquids decreases with increasing temperature.
In a sealed carbonated bottle the high pressure above the liquid increases the solubility of CO2. On opening the bottle the pressure above the liquid drops and excess CO2 comes out of solution until a new equilibrium with the atmosphere is established. If left open, the drink eventually goes flat as CO2 escapes.
Some important points to remember:
- For solid ⇌ liquid equilibrium there is a specific temperature (melting point) at which both phases coexist under 1 atm pressure; without heat exchange the masses of the phases remain constant.
- For liquid ⇌ vapour equilibrium the vapour pressure remains constant at a given temperature.
- The solubility of a solid in a liquid is fixed at a given temperature for a saturated solution.
- The concentration of a gas dissolved in a liquid (solubility) is directly proportional to the partial pressure of that gas above the liquid (Henry's law).

Try yourself: When solid sugar dissolves in the solution __________ equilibrium is attained.
General Characteristics of Equilibrium in Physical Processes
The following characteristics are common to equilibrium states in physical processes:
- Equilibrium for physical processes is achieved only in a closed system at a given temperature; if matter can escape or enter the system the equilibrium will be disturbed.
- Opposing processes occur at the same rate and the system exhibits a dynamic but macroscopically stable condition during equilibrium.
- All measurable properties of the system (temperature, pressure, concentration, masses of coexisting phases) remain constant at equilibrium.
- Equilibrium in a physical process is usually characterised by a constant value of one parameter (for example vapour pressure, solubility or fraction of phases) at a given temperature.
- The extent to which a physical process has proceeded before reaching equilibrium is indicated by the magnitude of the relevant parameter (for example amount of dissolved solute or vapour pressure).
FAQs on Equilibrium in Physical Processes
| 1. What is equilibrium in physical processes and how does it differ from chemical equilibrium? |  |
| 2. Why does solid-liquid equilibrium happen at a specific temperature and how does pressure affect it? |  |
| 3. How do I identify when a physical process has reached equilibrium and what signs should I look for? |  |
| 4. What happens to vapour pressure during liquid-vapour equilibrium and why does it remain constant? |  |
| 5. Can solubility equilibrium reverse, and what role does temperature play in dissolving salts in water? |  |






