D- block elements
The elements in which last electron enters in d-orbital of penultimate Shell i.s. (n-1)d orbital are called as d-block elements.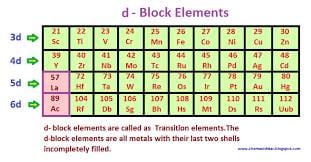
Transition Elements:-
The elements in which last electron enters in (n -1)d subshell and they having in completely filled (n -1)d subshell in their elementary state or common Oxidation state are called as transition elements.
Non transition elements:-
The elements having completely filled (n-1)d sub shell are called as non transition elements.
General electronic configuration:-
(n-1)d1-10,ns1-2
Note:- Chromium and copper shows Anomalous or abnormal electronic configuration.
General characters of transition elements:-
1) All are metals and shows metallic properties like malleability, ductility, Luster, etc.
2) They are good conductor of heat and electricity.
3) They form large number of complex compounds.
4) They having catalytic property.
5) They having high densities.
6) They having high melting point and boiling point.
7) They form organometallic compounds.
8) They have tendency to form coloured compounds.
9) They show variable oxidation States.

