Important Formulas: Calorimetry & Heat Transfer
Calorimetry
Calorimetry is the study of heat transfer associated with changes in temperature and changes of state. The principal quantities are heat (Q), temperature (θ), specific heat (s) and latent heat (L). Heat is measured in joules (J) in SI units; older practical units such as calorie (cal) also appear in school-level problems (1 cal = 4.186 J).
Basic formulae and definitions
- Heat when temperature changes without change of state: Q = m s Δθ, where m is mass, s is specific heat and Δθ = (θfinal - θinitial).
- Heat when change of state occurs without change of temperature (latent heat): Q = m L, where L is the latent heat of the substance.
- Specific heat (s): heat required to raise the temperature of unit mass of a substance by 1 °C (or 1 K). Its SI unit is J kg-1 K-1. In older problems specific heat is sometimes given in cal g-1 °C -1. The specific heat of water is 1 cal g-1 °C -1 (measured between about 14.5 °C and 15.5 °C), equivalent to 4186 J kg-1 K-1.
- Heat capacity (c) of a body: c = m s; it is the heat required to raise the temperature of the whole body by 1 °C (or 1 K). Its SI unit is J K-1.
- Latent heat (L): heat required to convert unit mass of a substance from one phase to another at constant temperature. Units: J kg-1.
- Water equivalent of a vessel: the mass of water that would absorb the same amount of heat as the vessel for the same temperature rise. If the vessel has mass mv and specific heat sv, its water equivalent is mw = mv sv (expressed in kg of water or grams of water depending on units used).
Common applications and useful relations
- Mixture and thermal equilibrium (no heat lost to surroundings): Total heat gained = Total heat lost. For two bodies:
m1 s1 (θf - θ1) + m2 s2 (θf - θ2) = 0,
where θf is the final equilibrium temperature. - Calorimeter problems: Include heat absorbed or released by the calorimeter using its water equivalent. If mw is the water equivalent and θc is initial calorimeter temperature, then include mw (θf - θc) in the heat balance.
- Sign convention: Heat gained is positive, heat lost is negative. Ensure consistent units before calculation (convert grams to kilograms or calories to joules as required).
Worked example (mixing two liquids)
Problem: A mass m1 = 200 g of water at θ1 = 80 °C is mixed with m2 = 300 g of water at θ2 = 20 °C in an insulated container. Find the final temperature θf. Take specific heat of water s = 1 cal g-1 °C -1.
Solution
Write heat balance: heat lost by hotter water = heat gained by colder water.
m1 s (θ1 - θf) = m2 s (θf - θ2)
Substitute numbers: 200 × 1 × (80 - θf) = 300 × 1 × (θf - 20)
Expand: 16000 - 200 θf = 300 θf - 6000
Collect θf terms: 16000 + 6000 = 500 θf
So θf = 22000 / 500 = 44 °C.
Ans: θf = 44 °C.
Heat Transfer
Heat transfer occurs by three mechanisms: conduction, convection and radiation. In many Class 11 and competitive-exam problems the focus is on conduction and radiation; convection is treated qualitatively or in simple applications.
Conduction
Conduction is heat transfer through a material without bulk motion of the material. It is described, in steady state, by Fourier's law.
- Rate of heat flow (steady state) through a uniform rod: Q/t = K A (θ1 - θ2) / L,
where Q/t is heat flow per unit time (W), K (or κ) is thermal conductivity of the material (W m-1 K-1), A is cross-sectional area, L is the length between temperatures θ1 and θ2. - Thermal resistance of a uniform rod: R = L / (K A).
- Analogy with electrical resistance: Heat current = ΔT / R, where R is thermal resistance.
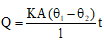
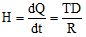
Radiation
Radiation is heat transfer by electromagnetic waves and requires no material medium. Key quantities are absorptive power, emissive power and emissivity.
- Absorptive power (absorptivity) a: fraction of incident radiation absorbed by the body. 0 ≤ a ≤ 1. For a perfect black body a = 1.
- Spectral absorptive power aλ: absorptivity at wavelength λ. 0 ≤ aλ ≤ 1.
- Emissive power (e): energy radiated per unit area per unit time. SI unit: W m-2.
- Spectral emissive power eλ: emissive power per unit wavelength at λ.
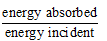
Spectral quantities describe the dependence on wavelength; total emissive or absorptive power is obtained by integrating spectral quantities over all wavelengths.
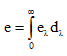
Stefan-Boltzmann law
Emissive power of a surface (Stefan's law): e = ε σ T4,
where ε is the emissivity (0 ≤ ε ≤ 1), σ is the Stefan-Boltzmann constant (σ = 5.670374419 × 10-8 W m-2 K-4), and T is absolute temperature in kelvin.
Total energy radiated in time t by area A: E = ε σ T4 A t.
Note: Emissivity ε is dimensionless and equals 1 for a perfect black body. Sometimes ε is called emittance or relative emissivity; do not confuse it with emissive power e which has units.
Kirchhoff's law of thermal radiation
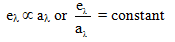
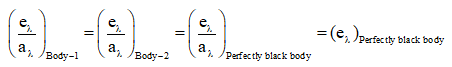
Kirchhoff's law states that, for a body in thermal equilibrium, the emissivity at a given wavelength equals the absorptivity at that wavelength. From this law we draw two practical conclusions:
- Good absorbers at a particular wavelength are also good emitters at that wavelength.
- At a given temperature, the ratio of emitted spectral power qλ to spectral absorptivity aλ for any body is equal to the spectral emissive power of a perfect black body at that temperature: qλ / aλ = eλ(black body).
Wien's displacement law
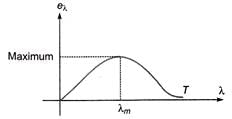
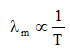
Wien's displacement law gives the wavelength λm at which the spectral emissive power of a black body is maximum:
λm T = b, where b is Wien's constant, b = 2.89 × 10-3 m·K.
The area under the spectral curve is proportional to T4 and gives total emissive power (leading to Stefan-Boltzmann law).
Cooling by radiation and Newton's law of cooling
Rate of cooling (qualitative): A body at temperature θ loses heat by radiation at a rate depending on its emissivity, surface area and fourth power of its absolute temperature; if the temperature difference with surroundings is small, the rate of cooling is approximately proportional to the temperature difference.
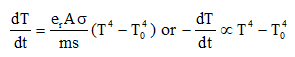
Newton's law of cooling (approximate): If the temperature difference between the body and the surroundings is small, the rate of change of temperature is proportional to that difference:
dθ/dt = -α (θ - θ0),
where θ0 is ambient temperature and α is a positive constant depending on geometry and heat-transfer properties.
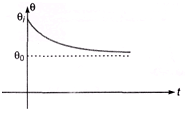
Solving this first-order linear differential equation for θ(t) with initial temperature θI at t = 0 gives the exponential cooling law:
θ(t) = θ0 + (θI - θ0) e-α t.
In many practical problems α is determined experimentally. For small temperature differences, radiative cooling approximately follows Newton's law because the T4 dependence can be linearised about the ambient temperature.
Remarks and applications
- Use the conduction formula Q/t = K A ΔT / L for steady-state heat flow along rods and for composite walls combine thermal resistances in series or parallel as in electrical circuits.
- Use calorimetry relations Q = m s Δθ and Q = m L for problems involving temperature changes and phase changes respectively; always include the calorimeter's water equivalent if present.
- For radiation problems, identify whether the surface behaves approximately like a black body (ε ≈ 1) or a poor emitter (ε ≪ 1); use Stefan-Boltzmann law for total radiative power and Wien's law to find peak wavelength.
- Newton's law of cooling is an approximation valid for small temperature differences or when convective heat transfer dominates; for large temperature differences use full radiative expression involving T4 or solve using appropriate boundary conditions.
Summary: Calorimetry provides formulas to compute heat exchanged in temperature changes and phase changes. Heat transfer by conduction uses Fourier's law and thermal resistance concepts. Thermal radiation is governed by emissivity, Kirchhoff's law, Wien's displacement law and Stefan-Boltzmann law; Newton's law of cooling is a useful approximation for small temperature differences.
FAQs on Important Formulas: Calorimetry & Heat Transfer
| 1. What is calorimetry? |  |
| 2. What are the key principles of heat transfer? |  |
| 3. How is specific heat capacity defined in calorimetry? |  |
| 4. What is the formula used to calculate heat transfer in calorimetry? |  |
| 5. How can calorimetry be applied in real-life scenarios? |  |






