NEET Previous Year Questions (2014-2025): Organic Chemistry - Some Basic Principles & Techniques
2025
Q1: Which one of the following reactions does not belong to "Lassaigne's test"? (NEET 2025)
(a) 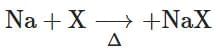
(b) 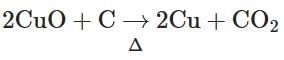
(c) 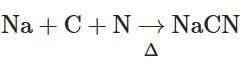
(d) 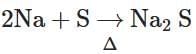
Ans: (b)
- Na + C + N → NaCN: This reaction forms sodium cyanide, used to detect nitrogen in the compound. This belongs to Lassaigne's test.
- 2Na + S → Na2S: This forms sodium sulfide, used to detect sulfur. This also belongs to Lassaigne's test.
- Na + X → NaX: This forms sodium halide, used to detect halogens like Cl, Br, and I. This belongs to Lassaigne's test.
- 2CuO + C → 2Cu + CO2: This reaction involves the reduction of copper oxide by carbon and is unrelated to Lassaigne's test for detecting N, S, or halogens.
Therefore, the reaction that does NOT belong to Lassaigne's test is 2CuO + C → 2Cu + CO2
Q2: Match List-I with List-II (NEET 2025)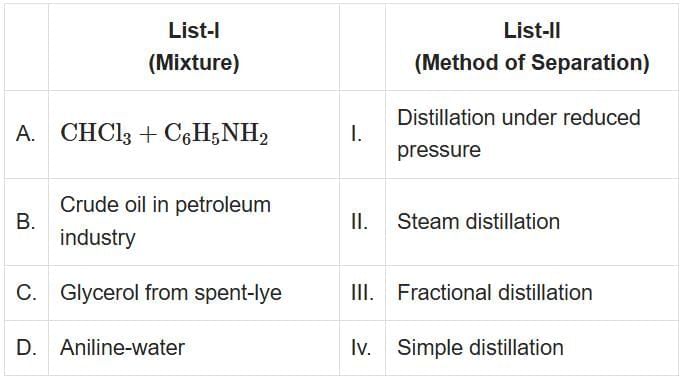
Choose the correct answer from the option given below:
(a) A-III, B-IV, C-I, D-II
(b) A-III, B-IV, C-II, D-I
(c) A-IV, B-III, C-I, D-II
(d) A-IV, B-III, C-II, D-I
Ans: (c)
- A. CHCl₃ + C₆H₅NH₂: These are heat-sensitive liquids. Therefore, Distillation under reduced pressure is used.
- B. Crude oil in petroleum industry: Crude oil is a complex mixture of hydrocarbons that have close boiling points. Therefore, Fractional Distillation is used.
- C. Glycerol from spent-lye: Glycerol is separated using Simple Distillation.
- D. Aniline-water: Aniline and water are separated using Steam Distillation as aniline is volatile in steam.
- A - I (Distillation under reduced pressure)
- B - III (Fractional Distillation)
- C - IV (Simple Distillation)
- D - II (Steam Distillation)
Therefore, the correct answer is A-I, B-III, C-IV, D-II.
2024
Q1: The most stable carbocation among the following is :(a)
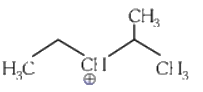
(b)
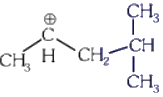
(c)
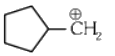
(d)
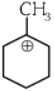 (NEET 2024)
(NEET 2024)Ans: (d)
The stability of carbocation can be described by the hyperconjugation. Greater the extent of hyperconjugation, more is the stability of carbocation.
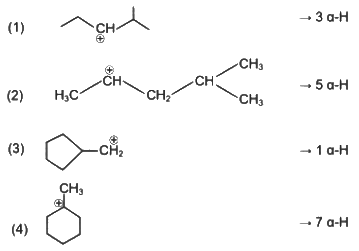
Stability order of carbocations = (4) > (2) > (1) > (3)
Q2: On heating, some solid substances change from solid to vapour state without passing through liquid state. The technique used for the purification of such solid substances based on the above principle is known as
(a) Crystallization
(b) Sublimation
(c) Distillation
(d) Chromatography (NEET 2024)
Ans: (b)
The technique described in the question is known as sublimation. Sublimation is a process where a solid turns directly into a gas without passing through the intermediate liquid state. This physical change occurs under specific conditions of temperature and pressure and is characteristic of certain substances.
Option B: Sublimation is the correct answer because it directly describes the process of solid substances transforming into vapour without becoming liquid first. For example, substances like iodine, naphthalene, and dry ice (solid carbon dioxide) sublimate when heated under normal atmospheric conditions. Sublimation is not only a fascinating chemical process but is also exploited in various scientific and industrial applications, including purification of substances, where impurities do not sublimate and can thus be easily separated from the vaporized material.
The other options provided do not describe this process:
- Crystallization (Option A) involves the formation of solid crystals from a homogeneous solution. It is typically used to purify solids wherein the impurities remain dissolved in the solvent.
- Distillation (Option C) is a process used to separate mixtures based on differences in the conditions required to change the phase (liquid phase) of the components of the mixture. It does not involve direct transition from solid to gas.
- Chromatography (Option D) is a method for separating components of a mixture based on differences in their movement through a stationary medium under the influence of a solvent or carrier gas. It does not involve the phase transition of solid to gas directly.
Q3: Given below are certain cations. Using inorganic qualitative analysis, arrange them in increasing group number from 0 to VI.
A. Al3+
B. Cu2+
C. Ba2+
D. Co2+
E. Mg2+
Choose the correct answer from the options given below.
(a) B, A, D, C, E
(b) B, C, A, D, E
(c) E, C, D, B, A
(d) E, A, B, C, D (NEET 2024)
Ans: (a) The correct order of group number of ions is
The correct order of group number of ions is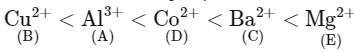
∴ The correct order is B, A, D, C, E
2023
Q1: The number of σ bonds, π bonds and lone pair of electrons in pyridine, respectively are: (NEET 2023)
(a) 12, 3, 0
(b) 11, 3, 1
(c) 12, 2, 1
(d) 11, 2, 0
Ans: (b)

Q2: In Lassaigne's extract of an organic compound, both nitrogen and sulphur are present, which gives blood red colour with Fe3+ due to the formation of (NEET 2023)
(a) Fe4[Fe(CN)6]3.xH2O
(b) NaSCN
(c) [Fe(CN)5NOS]4-
(d) [Fe(SCN)]2+
Ans: (d)
In case, nitrogen and sulphur both are present in organic compound, sodium thiocyanate is formed. It gives blood red colour and no Prussian blue since there are no free cyanide ions.
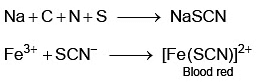
Q3: Amongst the given options which of the following molecules/ion acts as a Lewis acid? (NEET 2023)
(a) H2O
(b) BF3
(c) OH-
(d) NH3
Ans: (b)
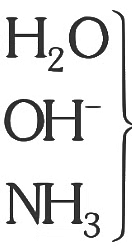
can not act as lewis acid because they does not contain vacant orbital
BF
Q4: Consider the following compounds/species: (NEET 2023)
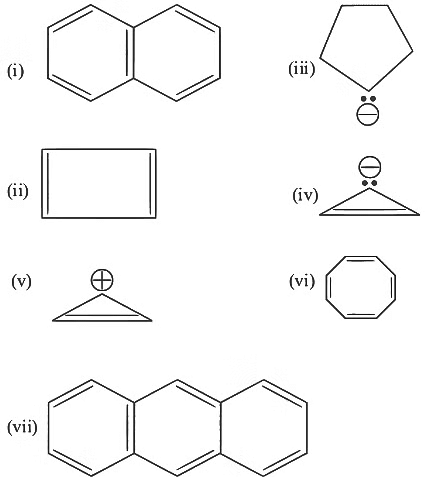
The number of compounds/species which obey Huckel's rule is ___________.
(a) 6
(b) 2
(c) 5
(d) 4
Ans: (d)
Huckle's rule = (4n + 2)π electrons
Comp (i), (ii), (v), (vii) obey Huckle's rule
2022
Q1: Given below are two statements : one is labelled as Assertion (A) and the other is labelled as Reason (R).
Assertion (A) : Chlorine is an electron withdrawing group but it is ortho, para directing in electrophilic aromatic substitution.
Reason (R) : Inductive effect of chlorine destabilises the intermediate carbocation formed during the electrophilic substitution, however due to the more pronounced resonance effect, the halogen stabilises the carbocation at ortho and para positioins.
In the light of the above statements, choose the most appropriate answer from the options given below :
(a) (A) is not correct but (R) is correct
(b) Both (A) and (R) are correct and (R) is the correct explanation of (A)
(c) Both (A) and (R) are correct but (R) is not the correct explanation of (A)
(d) (A) is correct but (R) is not correct (NEET 2022 Phase 2)
Ans: (d)
Cl has pronounced
Q2: Predict the order of reactivity of the following four isomers towards SN2 reaction. (NEET 2022 Phase 2)
(I) CH3CH2CH2CH2Cl
(II) CH3CH2CH(Cl)CH3
(III) (CH3)2CHCH2Cl
(IV) (CH3)3CCl
(a) (IV) > (II) > (III) > (I)
(b) (IV) > (III) > (II) > (I)
(c) (I) > (II) > (III) > (IV)
(d) (I) > (III) > (II) > (IV)
Ans: (d)
- Lesser the steric hinderance on halide carbon, more will be the reactivity of alkyl halide towards SN2 reaction.
- So correct order towards SN2 reactivity is :
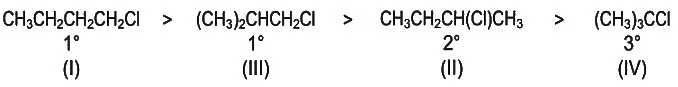
Q3: What is the hybridization shown by C1 and C2 carbons, respectively in the given compound?
OHC
(a) sp3 and sp3
(b) sp2 and sp3
(c) sp2 and sp2
(d) sp3 and sp2 (NEET 2022 Phase 2)
Ans: (b)
-COOCH3 has higher priority than
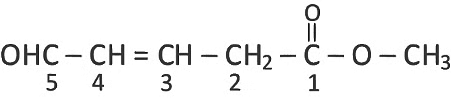
C1 = sp2
C2 = sp3
Q4: The Kjeldahl's method for the estimation of nitrogen can be used to estimate the amount of nitrogen in which one of the following compounds? (NEET 2022 Phase 1)
(a) 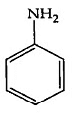
(b) 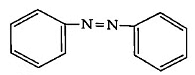
(c) 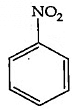
(d) 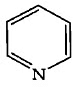
Ans: (a)
Kjeldahl method is not used for the organic compound containing
-NO2 group, -N = N- group,
And N- containing in the ring.
Q5: The correct IUPAC name of the following compound is: (NEET 2022 Phase 1)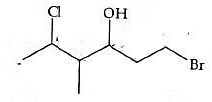
(a) 1-bromo-4-methyl-5-chlorohexan-3-ol
(b) 6-bromo-4-methyl-2-chlorohexan-4-ol
(c) 1-bromo-5-chloro-4-methylhexan-3-ol
(d) 6-bromo-2-chloro-4-methylhexan-4-ol
Ans: (c)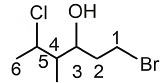
Q6: The incorrect statement regarding chirality is (NEET 2022 Phase 1)
(a) SN1 reaction yields 1 : 1 mixture of both enantiomers
(b) The product obtained by SN2 reaction of haloalkane having chirality at the reactive site shows inversion of configuration
(c) Enantiomers are superimposable mirror images on each other
(d) A racemic mixture shows zero optical rotation
Ans: (c)
The stereoisomers related to each other as non-superimposable mirror image are called enantiomers.
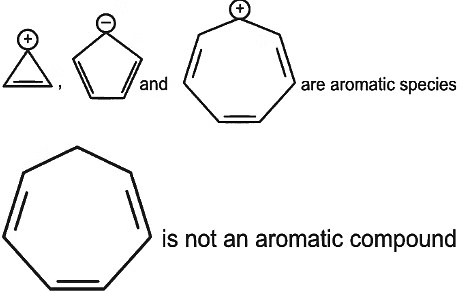
Q7: Which compound amongst the following is not an aromatic compound? (NEET 2022 Phase 1)
(a) 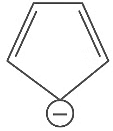
(b) 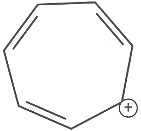
(c) 
(d) 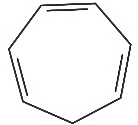
Ans: (d)
Planar, cyclic, conjugated species containing (4n + 2)π electrons will be aromatic in nature (n is an integer)
2021
Q1: The compound which shows metamerism is: (NEET 2021)
(a) C3H6O
(b) C4H10O
(c) C5H12
(d) C3H8O
Ans: (b)
C4H10O will have different alkyl group attached with polyvalent functional group that's why show metamerism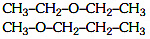
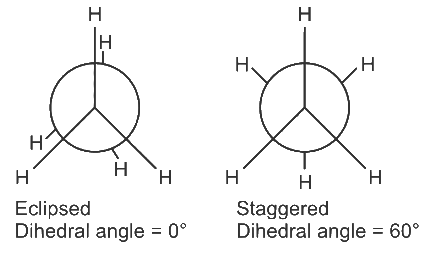
Q2: Dihedral angle of least stable conformer of ethane is :: (NEET 2021)
(a) 0∘
(b) 120∘
(c) 180∘
(d) 60∘
Ans: (a)
Ethane has two conformers (i) Eclipsed (ii) Staggered
Eclipsed conformer is least stable while staggered conformer is most stable. In eclipsed conformer the dihedral angle is 0∘.
2020
Q1: A tertiary butyl carbocation is more stable than a secondary butyl carbocation because of which of the following? (NEET 2020)
(a) -R effect of -CH3 groups
(b) Hyperconjugation
(c) -I effect of -CH3 groups
(d) + R effect of -CH3 groups
Ans: (b)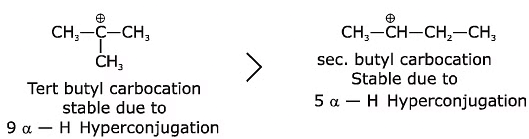
Q2: Paper chromatography is an example of : (NEET 2020)
(a) Partition chromatography
(b) Thin layer chromatography
(c) Column chromatography
(d) Adsorption chromatography
Ans: (a)
Paper chromatography is a type of partition chromatography in which a special quality paper known as chromatography paper is used.
2019
Q1: The compound that is most difficult to protonate is : (NEET 2019)
(a)
(b)
(c)
(d)
Ans: (d)
In Ph - OH, the lone pair of electrons on O atom causes delocalization which makes it difficult to protonate due to less availability of electrons.
2018
Q1: Which of the following carbocations is expected to be most stable ? (NEET 2018)
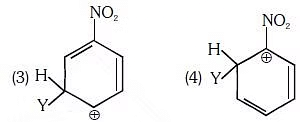 (a) 1
(a) 1
(b) 2
(c) 3
(d) 4
Ans: C
-NO2 group is meta-directing, thus will stabilize a electrophile at m-position.
Q2: Which of the following molecules represents the order of hybridisation sp2 , sp2 , sp, sp from left to right atoms?
(a) HC
(b) CH2 = CH - C
(c) CH2 = CH - CH = CH2
(d) CH3 - CH = CH - CH3 (NEET 2018)
Ans: (b)
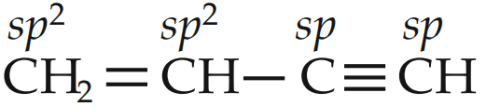
2017
Q1: The correct statement regarding electrophile is (NEET 2017)
(a) Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from another electrophile
(b) Electrophiles are generally neutral species and can form a bond by accepting a pair of electrons from a nucleophile
(c) Electrophile can be either neutral or positively charged species and can form a bond by accepting a pair of electrons from a nucleophile
(d) Electrophile is a negatively charged species and can form a bond by accepting a pair of electrons from a nucleophile
Ans: (c)
Electrophile can be either neutral or positively charged species and can form a bond by accepting a pair of electron from a nucleophile.
Q2: The IUPAC name of the compound (NEET 2017)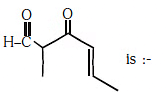
(a) 5-formylhex-2-en-3-one
(b) 5-methyl-4-oxohex-2-en-5-al
(c) 3-keto-2-methylhex-5-enal
(d) 3-keto-2-methylhex-4-enal
Ans: (d)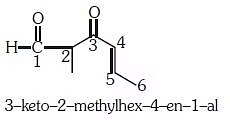
Q3: The most suitable method of separation of 1 : 1 mixture of ortho and para-nitrophenols is (NEET 2017)
(a) chromatography
(b) crystallisation
(c) steam distillation
(d) sublimation.
Ans: (c)
Steam distillation is the most suitable method of separation of 1 : 1 mixture of ortho and para nitrophenols as there is intramolecular hydrogen bonding in o-nitrophenol.
2016
Q1: The correct statement regarding the comparison of staggered and eclipsed conformations of ethane, is: (NEET 2016 Phase 1)
(a) The staggered conformation of ethane is more stable than eclipsed conformation, because staggered conformation has no torsional strain.
(b) The staggered conformation of ethane is less stable than eclipsed conformation, because staggered conformation has torsional strain.
(c) The eclipsed conformation of ethane is more stable than staggered conformation, because eclipsed conformation has no torsional strain.
(d) The eclipsed conformation of ethane is more stable than staggered conformation even though the eclipsed conformation has torsional strain.
Ans: (a)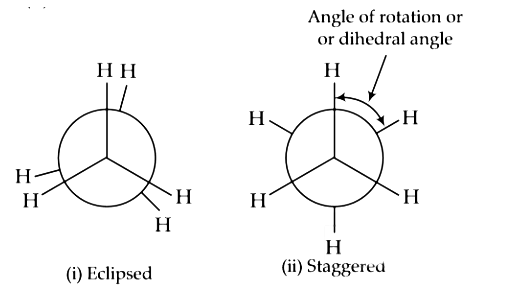
In staggered conformation any two hydrogen atoms on adjacent carbon atoms are as far apart as possible there by minimising repulsion between the electron clouds σ- of bonds of two non-bonded H-atomic (torsional strain)
Q2: Which among the given molecules can exhibit tautomerism? (NEET 2016 Phase 2)
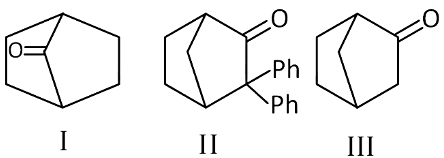
(a) III only
(b) Both I and III
(c) Both I and II
(d) Both II and III
Ans: (a)
α-hydrogen at bridge carbon never participates in tautomerism. Thus, only (III) exhibits tautomerism.
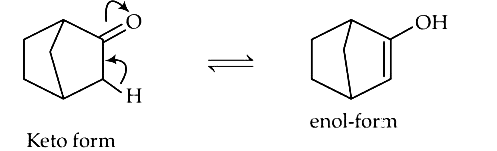
2015
Q1: The enolic form of ethyl acetoacetate as below has : (NEET / AIPMT 2015)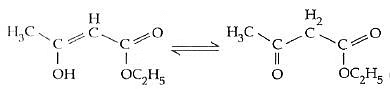 (a) 9 sigma bonds and 1 pi - bond(b) 18 sigma bonds and 2 pi - bonds
(a) 9 sigma bonds and 1 pi - bond(b) 18 sigma bonds and 2 pi - bonds
(c) 16 sigma bonds and 1 pi-bond
(d) 9 sigma bonds and 2 pi - bonds
Ans: (b)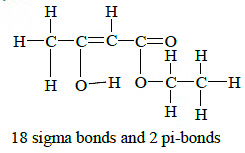 Q2: Consider the following compounds (NEET / AIPMT 2015)
Q2: Consider the following compounds (NEET / AIPMT 2015)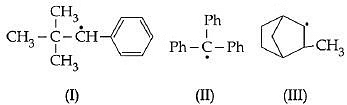 Hyperconjugation occurs in :(a) I and III
Hyperconjugation occurs in :(a) I and III
(b) I only
(c) II only
(d) III only
Ans: (d)
Hyperconjugation occurs through the H- atoms present on the carbon atom next to the double bond i.e alpha hydrogen atoms. There is no alpha -H in the structure I and II.
So, hyperconjugation occurs in structure III only ie.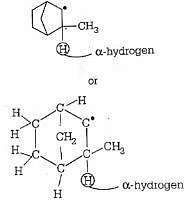 Q3: Which of the following is the most correct electron displacement for a nucleophilic reaction to take place ? (NEET / AIPMT 2015)
Q3: Which of the following is the most correct electron displacement for a nucleophilic reaction to take place ? (NEET / AIPMT 2015)
(a)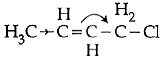
(b)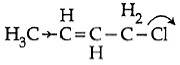
(c)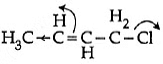
(d)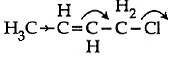
Ans: (d)
Allylic and benzylic halides show high reactivity towards Sn1 reaction Further, due to greater stabilisation of allyl and benzyl carbocations intermediates by resonance, primary allylic and primary benzylic halides show higher reactivity in Sn1 reactions than other simple primary halides.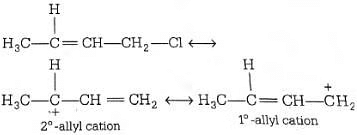 Hence, it undergoes nucleophilic reaction readily.
Hence, it undergoes nucleophilic reaction readily.
Q4: The total number of π -bond electrons in the following structure is : (NEET / AIPMT 2015)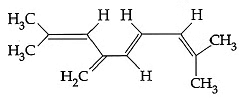 (a) 16
(a) 16
(b) 4
(c) 8
(d) 12
Ans: (c)
Q5: In which of the following compounds, the C - Cl bond ionisation shall give most stable carbonium ion ? (NEET / AIPMT 2015)
(a)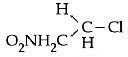
(b)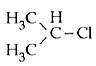
(c)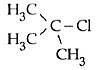
(d)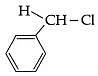
Ans: (c)
3° carbocation are more stable than benzylic carbocation.
Q6: In Duma's method for estimation of nitrogen, 0.25 g of an organic compound gave 40 mL of nitrogen collected at 300 K temperature and 725 mm pressure. If the aqueous tension at 300K is 25mm, the percentage of nitrogen in the compound is : (NEET / AIPMT 2015)
(a) 15.76
(b) 17.36
(c) 18.20
(d) 16.76
Ans: (d)
Q7: Given :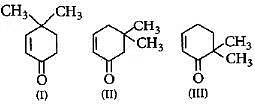 Q. Which of the given compounds can exhibit tautormerism ? (NEET / AIPMT 2015)
Q. Which of the given compounds can exhibit tautormerism ? (NEET / AIPMT 2015)
(a) I, II and III
(b) I and II
(c) I and III
(d) II and III
Ans: (a)
In keto-enol tautomerism,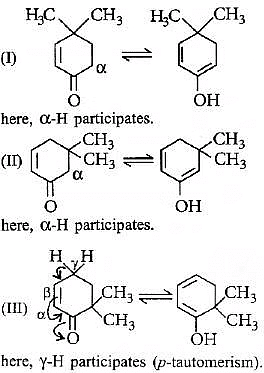
Q8:Which of the following statements is not correct for a nucleophile? (NEET / AIPMT 2015)
(a) Ammonia is a nucleophile.
(b) Nucleophiles attack low e- density sites.
(c) Nucleophiles are not electron seeking.
(d) Nucleophile is a Lewis acid.
Ans: (d)
Nucleophile is a species that provide electron while species which are deficient of electrons are termed as lewis acid, hence nucleophiles are usually lewis bases.
Q9: The possible stereo-structures of CH3CHOHCOOH, which are optically active, are called (NEET / AIPMT 2015)
(a) atropisomers
(b) enantiomers
(c) mesomers
(d) diastereomers
Ans: (b)
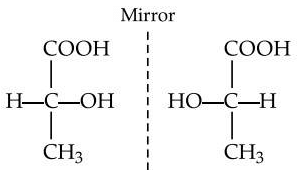
They are enantiomers (non-superimposable mirror images)
2014
Q1: In the Kjeldahl's method for estimation of nitrogen present in a soil sample, ammonia evolved from 0.75 g of sample neutralized 10 mL of 1M H2SO4. The percentage of nitrogen in the soil is : (NEET 2014)
(a) 35.33
(b) 43.33
(c) 37.33
(d) 45.33
Ans: C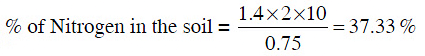
FAQs on NEET Previous Year Questions (2014-2025): Organic Chemistry - Some Basic Principles & Techniques
| 1. What are the main topics covered in organic chemistry previous year NEET questions from 2014 onwards? |  |
| 2. How do I identify structural isomers versus stereoisomers in NEET organic chemistry problems? |  |
| 3. What is the significance of hybridisation and bond angles in determining organic compound properties for NEET? |  |
| 4. Why do inductive and resonance effects matter in organic chemistry NEET questions about acid-base strength? |  |
| 5. How do I approach nomenclature and functional group identification in NEET organic chemistry previous year papers? |  |







