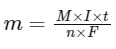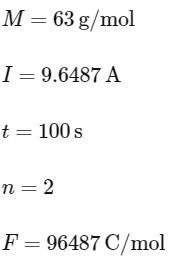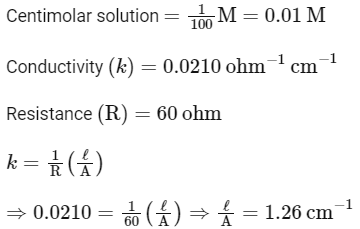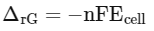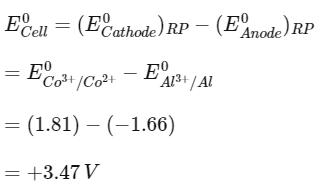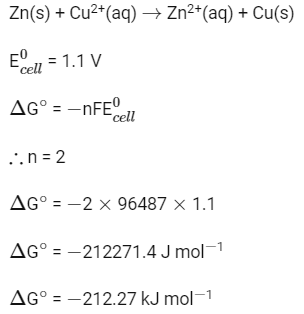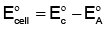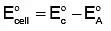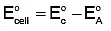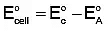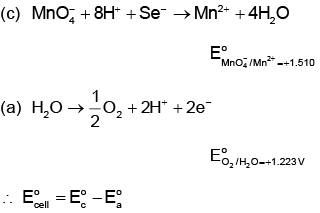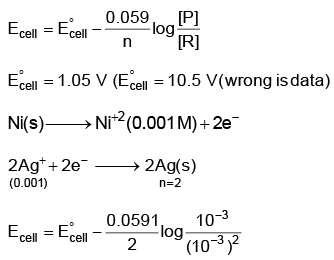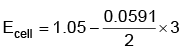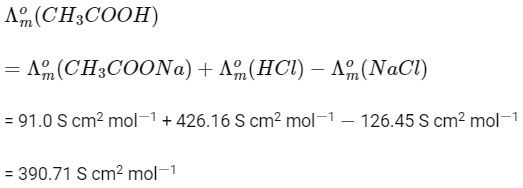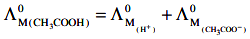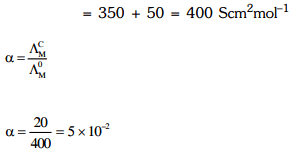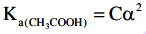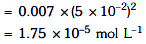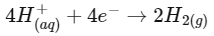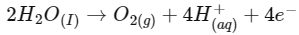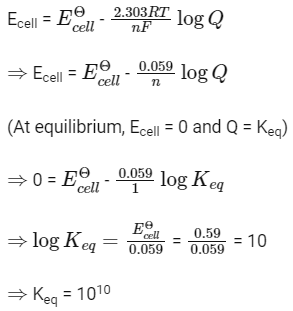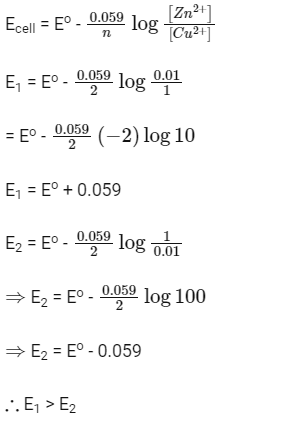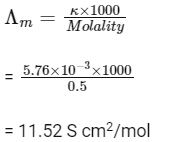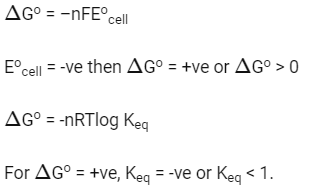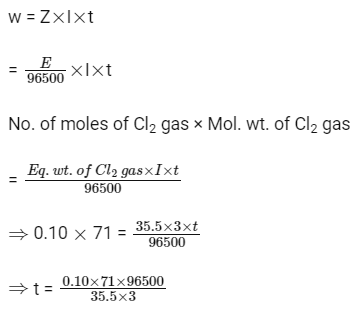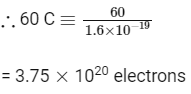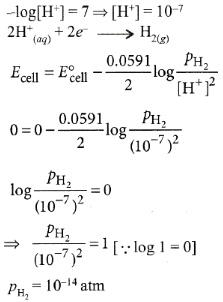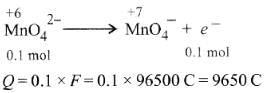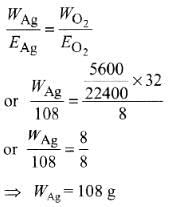Q4: The correct value of cell potential in volts for the reaction that occurs when the following two half-cells are connected is:
Fe²⁺(aq) + 2e⁻ → Fe(s), E° = -0.44 V
Cr₂O₇²⁻(aq) + 14H⁺ + 6e⁻ → 2Cr³⁺ + 7H₂O, E° = +1.33 V
(a) +1.77 V
(b) +2.65 V
(c) +0.01 V`
(d) +0.89 V (NEET 2023)
NEET Previous Year Questions (2014-2025): Electrochemistry
2025
Q1: If the molar conductivity (Λm) of a 0.050 mol L-1 solution of a monobasic weak acid is 90 S Cm2 mol-1, its extent (degree) of dissociation will be [Assume Λo+ = 349.6 S Cm2 mol-1 and Λo - = 50.4 S cm2 mol-1.] (NEET 2025)
(a) 0.225
(b) 0.215
(c) 0.115
(d) 0.125
2024
Q1: Match List I with List II.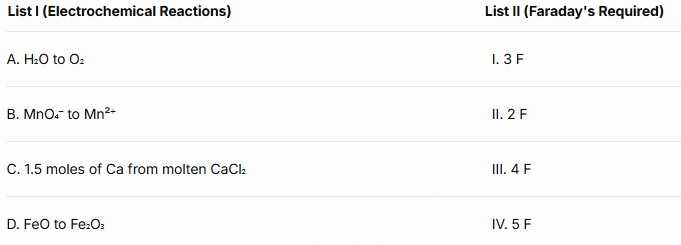
Choose the correct answer from the options given below :
(a) A-II, B-IV, C-I, D-III
(b) A-III, B-IV, C-I, D-II
(c) A-II, B-III, C-I, D-IV
(d) A-III, B-IV, C-II, D-I (NEET 2024)
Q2: Mass in grams of copper deposited by passing 9.6487 A current through a voltmeter containing copper sulphate solution for 100 seconds is (Given : Molar mass of Cu : 63 g mol-1,1 F = 96487C)
(a) 3.15 g
(b) 0.315 g
(c) 31.5 g
(d) 0.0315 g (NEET 2024)
Q3: Given below are two statements: (NEET 2024)
Statement I: 2 F electricity is required for the oxidation of 1 mole H₂O to O₂.
Statement II: To get 40.0 g of Aluminium from molten Al₂O₃, the required electricity is 4.44 F.
In the light of the above statements, choose the correct answer:
(a) Both Statement I and Statement II are true
(b) Both Statement I and Statement II are false
(c) Statement I is true but Statement II is false
(d) Statement I is false but Statement II is true
Q4: Select the one which is not an example of corrosion: (NEET 2024)
(a) Rusting of an iron object
(b) Production of hydrogen by electrolysis of water
(c) Tarnishing of silver
(d) Development of green coating on copper and bronze ornaments
Q5: The standard cell potential of the following cell: Zn | Zn²⁺ (aq) || Fe²⁺ (aq) | Fe is 0.32 V. (NEET 2024)
Calculate the standard Gibbs energy change for the reaction:
Zn (s) + Fe²⁺ (aq) → Zn²⁺ (aq) + Fe (s)
(Given: 1 F = 96487 C mol⁻¹)
(a) -61.75 kJ mol⁻¹
(b) +5.006 kJ mol⁻¹
(c) -5.006 kJ mol⁻¹
(d) +61.75 kJ mol⁻¹
2023
Q.1. The conductivity of centimolar solution of KCl at 25°C is 0.0210 ohm-1 cm-1 and the resistance of the cell containing the solution at 25°C is 60 ohm. The value of cell constant is (NEET 2023)(a) 1.34 cm-1
(b) 3.28 cm-1
(c) 1.26 cm-1
(d) 3.34 cm-1
Q.2. Given below are two statements: one is labelled as Assertion A and the other is labelled as Reason R (NEET 2023)
Assertion A : In equation △rG = -nFEcell' value of △rG depends on n.
Reasons R: Ecell is an intensive property and △rG is an extensive property.
In the light of the above statements, choose the correct answer from the options given below
(a) Both A and R are true and R is the correct explanation of A
(b) Both A and R are true and R is NOT the correct explanation of A
(c) A is true but R is false
(d) A is false but R is true
Q3: The molar conductance of an electrolyte increases with dilution according to the equation: (NEET 2023)
Λₘ = Λₘ⁰ - A√c
Consider the following four statements:
(A) This equation applies to both strong and weak electrolytes.
(B) The value of the constant A depends upon the nature of the solvent.
(C) The value of constant A is the same for both BaCl₂ and MgSO₄.
(D) The value of constant A is the same for both BaCl₂ and Mg(OH)₂.
Which of the above statements are correct?
(a) (A) and (B) only
(b) (A), (B), and (C) only
(c) (B) and (C) only
(d) (B) and (D) only
(a) 0.124 S cm² mol⁻¹
(b) 1.24 S m² mol⁻¹
(c) 124 S cm² mol⁻¹
(d) 124 S m² mol⁻¹
2022
Q1: Two half cell reactions are given below. (NEET 2022 Phase 2)
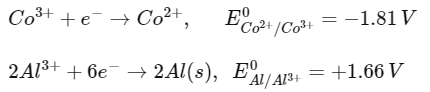
The standard EMF of a cell with feasible redox reaction will be :
(a) -3.47 V
(b) +7.09 V
(c) +0.15 V
(d) +3.47 V
Q2: Standard electrode potential for the cell with cell reaction (NEET 2022 Phase 2)
Zn(s) + Cu2+(aq) → Zn2+(aq) + Cu(s)
is 1.1 V. Calculate the standard Gibbs energy change for the cell reaction. (Given F = 96487 C mol-1)
(a) -200.27 J mol-1
(b) -200.27 kJ mol-1
(c) -212.27 kJ mol-1
(d) -212.27 J mol-1
Q3: At 298 K, the standard electrode potentials of Cu2+ / Cu, Zn2+/Zn, Fe2+/Fe and Ag+/Ag are 0.34 V, - 0.76 V, - 0.44 V and 0.80 V, respectively. On the basis of standard electrode potential, predict which of the following reaction can not occur?
(a) FeSO4(aq) + Zn(s) → ZnSO4(aq) + Fe(s)
(b) 2CuSO4(aq) + 2Ag(s) → 2Cu(s) + Ag2SO4(aq)
(c) CuSO4(aq) + Zn(s) → ZnSO4(aq) + Cu(s)
(d) CuSO4(aq) + Fe(s) → FeSO4(aq) + Cu(s) (NEET 2022 Phase 1)
Q4: Given below are half-cell reactions: (NEET 2022 Phase 1)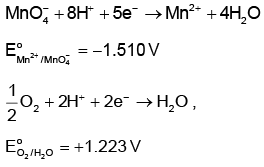
Will the permanganate ion,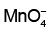 liberate O2 from water in the presence of an acid?
liberate O2 from water in the presence of an acid?
(a) Yes, because E°cell = + 2.733 V
(b) No, because E°cell = - 2.733 V
(c) Yes, because E°cell = + 0.287 V
(d) No, because E°cell = - 0.287 V
Q5: Find the emf of the cell in which the following reaction takes place at 298 K
Ni(s) + 2Ag+ (0.001 M) → Ni2+(0.001 M) + 2Ag(s)
(Given that 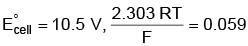 at 298 K)
at 298 K)
(a) 0.9615 V
(b) 1.05 V
(c) 1.0385 V
(d) 1.385 V (NEET 2022 Phase 1)
2021
Q1: The molar conductance of NaCl, HCI, and CH3COONa at infinite dilution are 126.45, 426.16, and 91.0 S cm mol-1 respectively. The molar conductance of CH3COOH at infinite dilution is. Choose the right option for your answer. (NEET 2021)
(a) 698.28 ohm-1 cm2 mol-1
(b) 540.48 ohm-1 cm2 mol-1
(c) 201.28 ohm-1 cm2 mol-1
(d) 390.71 ohm-1 cm2 mol-1
Q2: The molar conductivity of 0.007 M acetic acid is 20 S cm2 mol-1. What is the dissociation constant of acetic acid? Choose the correct option. (2021)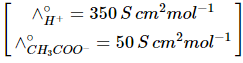
(a) 1.75 × 10-5 mol L-1
(b) 2.50 × 10-5 mol L-1
(c) 1.75 × 10-4 mol L-1
(d) 2.50 × 10-4 mol L-1
2020
Q1: On electrolysis of dil. sulphuric acid using platinum (Pt) electrode, the product obtained at the anode will be: (NEET 2020)
(a) H2S gas
(b) SO2 gas
(c) Hydrogen gas
(d) Oxygen gas
Q2: The number of Faradays (F) required to produce 20g of calcium from molten CaCl2 (Atomic mass of Ca = 40 g mol-1) is:
(a) 3
(b) 4
(c) 1
(d) 2 (NEET 2020)
2019
Q1: For a cell involving one electron E°cell = 0.59 V at 298 K, the equilibrium constant for the cell reaction is :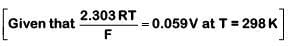 (NEET 2019)
(NEET 2019)
(a) 1.0 × 102
(b) 1.0 × 105
(c) 1.0 × 1010
(d) 1.0 × 1030
Q2: For the cell reaction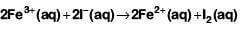
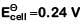 at 298 K. The standard Gibbs energy
at 298 K. The standard Gibbs energy 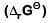 of the cell reaction is :[Given that Faraday constant F = 96500 C mol-1] (NEET 2019)
of the cell reaction is :[Given that Faraday constant F = 96500 C mol-1] (NEET 2019)
(a) - 46.32 kJ mol-1
(b) - 23.16 kJ mol-1
(c) 46.32 kJ mol-1
(d) 23.16 kJ mol-1
2018
Q1: Consider the change in the oxidation state of Bromine corresponding to different emf values as shown in the diagram below: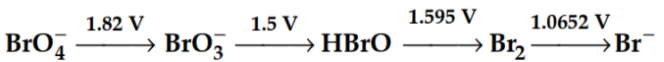 Then the species undergoing disproportionation is:- (NEET 2018)
Then the species undergoing disproportionation is:- (NEET 2018)
(a) BrO3-
(b) BrO4-
(c) Br2
(d) HBrO
2017
Q1: In the electrochemical cell : 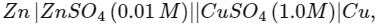 the emf of this Daniell cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that of CuSO4 changed to 0.01 M, the emf changes to E2. From the followings, which one is the relationship between E1 and E2? (Given, RT/F = 0.059) (NEET 2017)
the emf of this Daniell cell is E1. When the concentration of ZnSO4 is changed to 1.0 M and that of CuSO4 changed to 0.01 M, the emf changes to E2. From the followings, which one is the relationship between E1 and E2? (Given, RT/F = 0.059) (NEET 2017)
(a) E1 < E2
(b) E1 > E2
(c) E2 = 0 ≠ E1
(d) E1 = E2
2016
Q1: The molar conductivity of a 0.5 mol/dm3 solution of AgNO3 with electrolytic conductivity of 5.76
(a) 2.88 S cm2/mol
(b) 11.52 S cm2/mol
(c) 0.086 S cm2/mol
(d) 28.8 S cm2/mol (NEET 2016 Phase 2)
Q2: If the Eocell for a given reaction has a negative value, which of the following gives the correct relationships for the values of ΔGo and Keq ?
(a) ΔGo > 0; Keq < 1
(b) ΔGo > 0; Keq > 1
(c) ΔGo < 0; Keq > 1
(d) ΔGo < 0; Keq < 1 (NEET 2016 Phase 2)
Q3: During the electrolysis of molten sodium chloride, the time required to produce 0.10 mol of chlorine gas using a current of 3 amperes is (NEET 2016 Phase 2)
(a) 55 minutes
(b) 110 minutes
(c) 220 minutes
(d) 330 minutes
Q4: Zinc can be coated on iron to produce galvanized iron but the reverse is not possible. It is because
(a) zinc is lighter than iron
(b) zinc has lower melting point than iron
(c) zinc has lower negative electrode potential than iron
(d) zinc has higher negative electrode potential than iron (NEET 2016 Phase 2)
Q5: The number of electrons delivered at the cathode during electrolysis by a current of 1 ampere in 60 seconds is (charge on electron = 1.60 × 10-19C)
(a) 6 × 1023
(b) 6 × 1020
(c) 3.75 × 1020
(d) 7.48 × 1023 (NEET 2016 Phase 2)
Q6: The pressure of H2 required to make the potential of H2-electrode zero in pure water at 298 K is : (NEET 2016 Phase 1)
(a) 10-4 atm
(b) 10-14 atm
(c) 10-12 atm
(d) 10-10 atm
2015
Q1: Aqueous solution of which of the following compounds is the best conductor of electric current ?
(a) Hydrochloric acid, HCl
(b) Ammonia, NH3
(c) Fructose, C6H12O6
(d) Acetic acid, C2H4O2 (NEET / AIPMT 2015)
Q2: A device that converts the energy of combustion of fuels like hydrogen and methane, directly into electrical energy is known as (NEET / AIPMT 2015 Cancelled Paper)
(a) Ni-Cd cell
(b) Fuel Cell
(c) Electrolytic Cell
(d) Dynamo
2014
Q1: When 0.1 mol 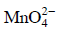 is oxidised the quantity of electricity required to completely oxidise
is oxidised the quantity of electricity required to completely oxidise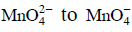 is :
is :
(a) 9650 C
(b) 96.50 C
(c) 96500 C
(d) 2 × 96500 C (NEET / AIPMT 2014)
Q2: The weight of silver (at. wt. = 108) displaced by a quantity of electricity that displaces 5600 mL of O2 at STP will be :
(a) 54.0 g
(b) 108.0 g
(c) 5.4 g
(d) 10.8 g (NEET / AIPMT 2014)
FAQs on NEET Previous Year Questions (2014-2025): Electrochemistry
| 1. What are the most important electrochemistry formulas that come in NEET exams? |  |
| 2. How do I solve galvanic cell and electrolytic cell questions that appear in NEET? |  |
| 3. Why do reduction potentials matter in electrochemistry NEET questions? |  |
| 4. What's the difference between standard cell potential and actual cell potential in electrochemistry? |  |
| 5. How are moles of products calculated using Faraday's laws in NEET electrochemistry problems? |  |