NCERT Exemplar: Some Basic Principles & Techniques
MULTIPLE CHOICE QUESTIONS
Q.1. Which of the following is the correct IUPAC name?
(1) 3-Ethyl-4, 4-dimethylheptane
(2) 4,4-Dimethyl-3-ethylheptane
(3) 5-Ethyl-4, 4-dimethylheptane
(4) 4,4-Bis(methyl)-3-ethylheptane
Ans: (1)
Explanation: In IUPAC nomenclature substituent names are cited in alphabetical order, and multiplicative prefixes (di, tri, etc.) are ignored for alphabetical ordering. Therefore 'ethyl' comes before 'methyl' and option (1) is the correct name.
Q.2. The IUPAC name for
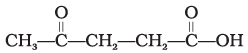
is ________.
(1) 1-hydroxypentane-1,4-dione
(2) 1,4-dioxopentanol
(3) 1-carboxybutan-3-one
(4) 4-oxopentanoic acid
Ans: (4)
Explanation: When more than one functional group is present the highest-priority functional group is chosen as the principal suffix. A carboxylic acid has higher priority than a ketone or alcohol and is therefore given the suffix -oic acid; other carbonyl groups are treated as oxo substituents with appropriate locants. The correct name corresponding to the structure is 4-oxopentanoic acid (option 4).
Q.3. The IUPAC name for
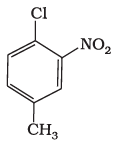
(1) 1-Chloro-2-nitro-4-methylbenzene
(2) 1-Chloro-4-methyl-2-nitrobenzene
(3) 2-Chloro-1-nitro-5-methylbenzene
(4) m-Nitro-p-chlorotoluene
Ans: (2)
Explanation: Number the ring so that substituents receive the lowest possible set of locants; after locating the principal substituent at position 1, continue numbering in the direction that gives the next substituents the lowest numbers. Substituents are then cited in alphabetical order (chloro before methyl before nitro). Following these rules yields 1-chloro-4-methyl-2-nitrobenzene (option 2).
Q.4. Electronegativity of carbon atoms depends upon their state of hybridisation. In which of the following compounds, the carbon marked with asterisk is most electronegative?
(1) CH3 - CH2 - *CH2 - CH3
(2) CH3 - *CH = CH - CH3
(3) CH3 - CH2 - C ≡ *CH
(4) CH3 - CH2 - CH = *CH2
Ans: (3)
Explanation: Electronegativity of a carbon atom increases with the s-character of its hybrid orbitals. sp3 (25% s) < sp2 (33% s) < sp (50% s). The asterisked carbon in option (3) is sp hybridized (as in an alkyne) and therefore is the most electronegative among the choices, so option (3) is correct.
Q.5. In which of the following, functional group isomerism is not possible?
(1) Alcohols
(2) Aldehydes
(3) Alkyl halides
(4) Cyanides
Ans: (3)
Explanation: Functional isomers have the same molecular formula but different functional groups. Examples: alcohols ↔ ethers, aldehydes ↔ ketones, cyanides ↔ isocyanides. Alkyl halides do not have a different functional group that can be formed without changing the halogen atom into a different functional group, therefore alkyl halides do not show functional group isomerism (option 3).
Q.6. The fragrance of flowers is due to the presence of some steam volatile organic compounds called essential oils. These are generally insoluble in water at room temperature but are miscible with water vapour in vapour phase. A suitable method for the extraction of these oils from the flowers is: (1) Distillation
(2) Crystallisation
(3) Distillation under reduced pressure
(4) Steam distillation
Ans: (4)
Explanation: Steam distillation is used to isolate steam-volatile, water-immiscible organic compounds at temperatures below their normal boiling points. Steam is passed through the plant material; the volatile oils co-distil with water vapour, are condensed and then separated from water using a separatory funnel. This prevents decomposition of heat-sensitive natural products (option 4).
Q.7. During hearing of a court case, the judge suspected that some changes in the documents had been carried out. He asked the forensic department to check the ink used at two different places. According to you which technique can give the best results?
(1) Column chromatography
(2) Solvent extraction
(3) Distillation
(4) Thin layer chromatography
Ans: (4)
Explanation: Thin-layer chromatography (TLC) is a rapid, sensitive analytical method to separate and compare components of mixtures. It requires only microgram amounts, resolves many ink components, and allows direct comparison of Rf values for questioned and reference samples. Because of its speed, sensitivity and ease of visual comparison, TLC is the preferred technique for ink analysis in forensic work (option 4).
Q.8. The principle involved in paper chromatography is
(1) Adsorption
(2) Partition
(3) Solubility
(4) Volatility
Ans: (2)
Explanation: Paper chromatography is an example of partition chromatography. Separation occurs by continuous differential partitioning of solutes between the stationary phase (a thin film of water retained in the paper) and the mobile solvent phase. Components distribute according to their partition coefficients (option 2).
Q.9. What is the correct order of decreasing stability of the following cations.
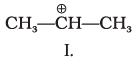
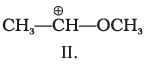
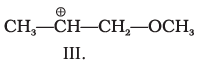
(1) II > I > III
(2) II > III > I
(3) III > I > II
(4) I > II > III
Ans: (1)
Explanation: Stability of carbocations increases when the positive charge is delocalised or when electron-releasing groups stabilise the cation. In structure II the +ve centre is stabilised by resonance (+R) from an OCH3 group. In I the cation is stabilised by electron-releasing alkyl groups (+I and hyperconjugation). In III the substituent exerts an overall electron-withdrawing effect that destabilises the positive centre. Thus II > I > III (option 1).
Q.10. Correct IUPAC name for 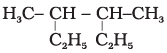 is______________.
is______________.
(1) 2- ethyl-3-methylpentane
(2) 3,4- dimethylhexane
(3) 2-sec-butylbutane
(4) 2, 3-dimethylbutane
Ans: (2)
Explanation: Choose the longest continuous carbon chain that gives the substituents the lowest possible locants while including substituents. The correct longest chain gives two methyl groups at positions 3 and 4 of a hexane backbone, hence 3,4-dimethylhexane (option 2).
Q.11. In which of the following compounds the carbon marked with asterisk is expected to have greatest positive charge?
(1) *CH3-CH2-Cl
(2) *CH3-CH2-Mg+Cl-
(3) *CH3-CH2-Br
(4) *CH3-CH2-CH3
Ans: (1)
Explanation: The magnitude of partial positive charge on a carbon atom increases when it is bonded to a more electronegative atom which withdraws electron density. Electronegativity order here is Cl > Br > C > Mg. Therefore the carbon next to Cl will bear the greatest partial positive charge (option 1).
Q.12. Ionic species are stabilised by the dispersal of charge. Which of the following carboxylate ion is the most stable?
(1) 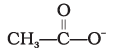
(2) 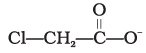
(3) 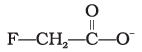
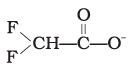
(4) 
Ans: (4)
Explanation: Stabilisation of a carboxylate ion depends on dispersal of the negative charge by resonance and by inductive effects of substituents. Electron-withdrawing halogens stabilise the negative charge by inductive effect; more electronegative substituents provide greater stabilization. Structure (4) has two fluorine atoms which give the greatest dispersal of negative charge, so it is the most stable (option 4).
Q.13. Electrophilic addition reactions proceed in two steps. The first step involves the addition of an electrophile. Name the type of intermediate formed in the first step of the following addition reaction.
H3C-HC = CH2 + H+ →?
(1) 2° Carbanion
(2) 1° Carbocation
(3) 2° Carbocation
(4) 1° Carbanion
Ans: (3)
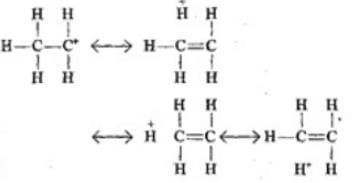
Explanation: When the electrophile attacks CH3 - CH = CH2, delocalisation of electrons can take place in two possible ways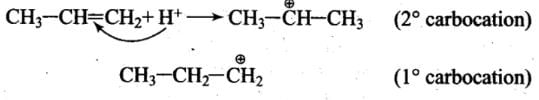
As 2° carbocation is more stable than 1° carbocation, the first addition is more feasible.
Q.14. Covalent bond can undergo fission in two different ways. The correct representation involving a heterolytic fission of CH3-Br is
(1)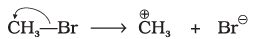
(2) 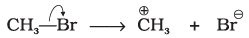
(3) 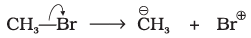
(4) 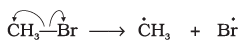
Ans: (2)
Explanation: In heterolytic fission the shared electron pair goes to the more electronegative atom. Bromine is more electronegative than carbon, so the bond breaks to give CH3+ and Br-. That is represented by option (2).
Q.15. The addition of HCl to an alkene proceeds in two steps. The first step is the attack of H+ ion to  portion which can be shown as
portion which can be shown as
(1) 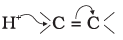
(2) 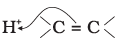
(3) 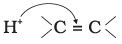
(4) All of these are possible
Ans: (2)
Explanation: Electrophilic attack of H+ occurs such that the more stable carbocation intermediate is formed. The π electrons of the double bond attack the proton and the direction of attack leads to the carbocation shown in option (2), which is the more stable intermediate for this alkene.
MULTIPLE CHOICE QUESTIONS II
In the following questions two or more options may be correct.
Q.16. Which of the following compounds contain all the carbon atoms in the same hybridisation state?
(i) H-C ≡ C-C ≡ C-H
(ii) CH3-C ≡ C-CH3
(iii) CH2 = C = CH2
(iv) CH2 = CH-CH = CH2
Ans: (1,4)
Explanation: In (i) all carbons are sp hybridised (each carbon in an alkyne linkage), and in (iv) all carbons in butadiene are sp2 hybridised. In (ii) terminal methyl carbons are sp3 while internal carbons are sp, and in (iii) the central carbon is sp hybridised while terminal carbons are sp2, so those do not have all carbons in the same hybridisation.
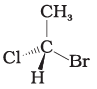
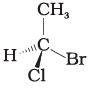
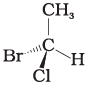
Q.17. In which of the following representations given below spatial arrangement of group/ atom different from that given in structure 'A'?
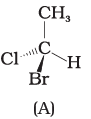
(1) 
(2) 
(3) 
(4) 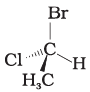
Ans: (1,3,4)
Explanation: To compare spatial arrangements, perform two interchanges to bring the hydrogen atom below the plane and then observe the sequence of remaining substituents (from highest to lowest priority). Structure (2) retains the same spatial arrangement as (A) after such operations; structures (1), (3) and (4) are different stereochemical arrangements.
Q.18. Electrophiles are electron seeking species. Which of the following groups contain only electrophiles?
(1) BF3, NH3 , H2O
(2) AlCl3, SO3, NO+2
(3) 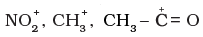
(4) 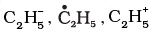
Ans: (2,3)
Explanation: Electrophiles are electron-deficient species or Lewis acids. AlCl3 and SO3 act as Lewis acids; NO2+ is a positively charged electrophile. The structures shown in option (3) represent positively charged species and are electrophilic. Option (1) contains NH3 and H2O which are nucleophilic, so (1) is incorrect.
Q.19. Which of the following pairs are position isomers?
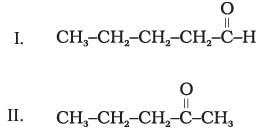
(1) I and II
(2) II and III
(3) II and IV
(4) III and IV
Ans: (2)
Explanation: Position isomers differ in the position of a functional group or multiple bond while keeping the same molecular formula. Pentan-2-one and pentan-3-one differ only by the position of the keto group, so II and III are position isomers (option 2).
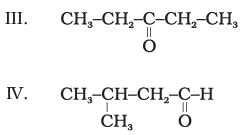
Q.20. Which of the following pairs are not functional group isomers?
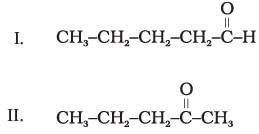
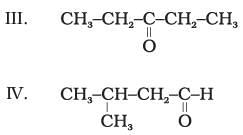
(1) II and III
(2) II and IV
(3) I and IV
(4) I and II
Ans. (1,2)
Explanation: Functional group isomers have the same molecular formula but different functional groups. In the given set, compounds II and III both have the ketonic group (so they are not functional isomers). Likewise II and IV both have the same functional group type in this set. Compounds I and IV are aldehydes while II and III are ketones; these pairs are functional isomers of each other.
Q.21. Nucleophile is a species that should have
(1) a pair of electrons to donate
(2) positive charge
(3) negative charge
(4) electron deficient species
Ans: (1,3)
Explanation: Nucleophiles are electron-rich species; they either carry a negative charge or possess a lone pair of electrons available for donation to an electrophile. Therefore options (1) and (3) are correct.
Q.22. Hyperconjugation involves delocalisation of ___________.
(1) electrons of carbon-hydrogen σ bond of an alkyl group directly attached to an atom of unsaturated system.
(2) electrons of carbon-hydrogen σ bond of alkyl group directly attached to the positively charged carbon atom.
(3) π-electrons of carbon-carbon bond
(4) lone pair of electrons
Ans. (1, 2)
Explanation: Hyperconjugation is delocalisation of electrons from a C-H σ bond (or other σ bonds of an alkyl group) into an adjacent empty p-orbital or π system. It stabilises carbocations and conjugated systems. Options (1) and (2) correctly describe hyperconjugation.
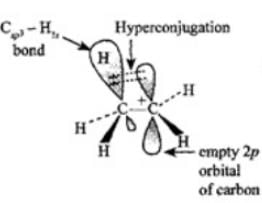
SHORT ANSWER TYPE QUESTIONS
Q.23.
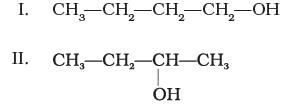
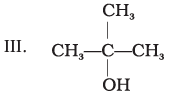
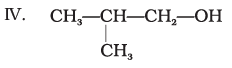
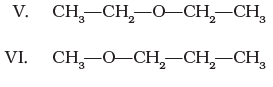
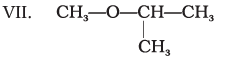
Which of the above compounds form pairs of metamers?
Ans: V and VI, VI and VII, and V and VII form pairs of metamers.
Explanation: Metamers are ethers (or related compounds) that have the same molecular formula but differ in the alkyl groups attached to the heteroatom (-O-). The listed pairs have different alkyl chains on either side of the ether oxygen and therefore are metamers of each other.
Q.24.



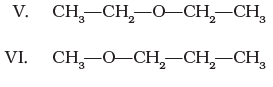

Identify the pairs of compounds which are functional group isomers.
Ans: Each of the alcohols I-IV is a functional group isomer of each of the ethers V-VII.
Explanation: The compounds I-IV contain -OH (alcohols) while V-VII contain -O- (ethers) and all share the molecular formula C4H10O. Therefore every alcohol (I-IV) forms a functional-group isomeric pair with every ether (V-VII). For example: I-V, I-VI, I-VII; II-V, II-VI, II-VII; III-V, III-VI, III-VII; IV-V, IV-VI, IV-VII.
Q.25.



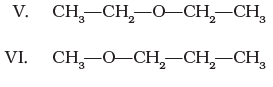

Identify the pairs of compounds that represents position isomerism.
Ans: I and II, III and IV, VI and VII are position isomers.
Explanation: In each indicated pair the functional group (-OH or -O-) occupies different positions on the same carbon skeleton while the overall molecular formula remains the same, therefore they are position isomers.
Q.26.



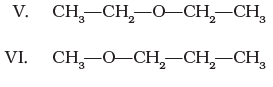

Identify the pairs of compounds that represents chain isomerism.
Ans: I and III, I and IV, II and III, II and IV.
Explanation: Chain isomers differ in the carbon skeleton (branching pattern) while retaining the same molecular formula. The listed pairs show the same molecular formula but different chain structures, hence they are chain isomers.
Q.27. For testing halogens in an organic compound with AgNO3 solution, sodium extract (Lassaigne's extract) is acidified with dilute HNO3. What will happen if a student acidifies the extract with dilute H2SO4 in place of dilute HNO3?
Ans. On adding dilute H2SO4 the silver ions react to give white precipitate of Ag2SO4, which interferes with the test for halides because Ag2SO4 can be mistaken for AgCl. Therefore dilute HNO3 is used to avoid formation of sulfate precipitate and obtain a reliable test for halogens.
Q.28. What is the hybridization of each carbon in H2C = C = CH2?
Ans. In H2C=C=CH2 (allene), the terminal carbons C1 and C3 are sp2 hybridised (each forms three σ bonds: two to H and one to the central carbon), while the central carbon C2 is sp hybridised (it forms two σ bonds and has two π bonds). The molecule as a whole is non-planar.
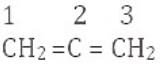
Q.29. Explain, how is the electronegativity of carbon atoms related to their state of hybridisation in an organic compound?
Ans. Electronegativity increases with increasing s-character of the hybrid orbitals because s-electrons are held more tightly by the nucleus than p-electrons. Thus:
sp3 - 25% s-character (least electronegative)
sp2 - 33% s-character
sp - 50% s-character (most electronegative)
Hence the order is sp3 < sp2 < sp.
Q.30. Show the polarization of carbon-magnesium bond in the following structure.
CH3-CH2-CH2-CH2-Mg-X
Ans. The C-Mg bond is polarized toward carbon because carbon is more electronegative than magnesium. The bonded electron pair is closer to carbon, giving it partial negative charge and magnesium a partial positive charge: Cδ- - Mgδ+-X. Thus the organomagnesium carbon behaves nucleophilically (carbon is electron-rich).
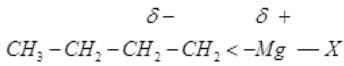
Q.31. Compounds with same molecular formula but differing in their structures are said to be structural isomers. What type of structural isomerism is shown by

Ans. These are position isomers.
Explanation: The two thioethers differ in how the alkyl groups are attached to the sulphur atom: methyl-n-propyl thioether versus methyl-isopropyl thioether. The connectivity differs by the position of branching in the carbon chain, so they are position isomers, not metamers (the number of carbons on either side of S remains the same).
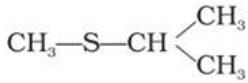
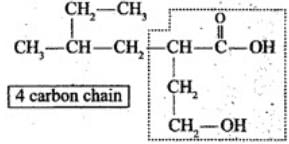
Q.32. Which of the following selected chains is correct to name the given compound according to IUPAC system.
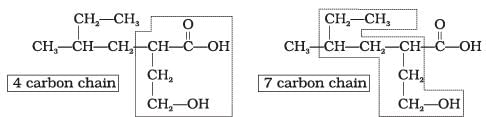
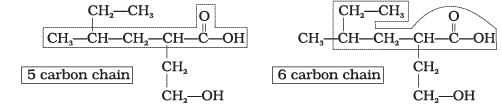
Ans. The correct selected chain is the one that includes the maximum number of carbon atoms and also includes both functional groups so that they receive the lowest possible locants.
Explanation: According to IUPAC rules the principal chain should contain the maximum number of principal functional groups. Only the selected chain of four carbon atoms that includes both functional groups is correct; the other choices omit one of the functional groups from the main chain and are therefore not correct choices.
Q.33. In DNA and RNA, nitrogen atom is present in the ring system. Can Kjeldahl method be used for the estimation of nitrogen present in these? Give reasons.
Ans: No, Kjeldahl method cannot be reliably used to estimate nitrogen in DNA or RNA.
Explanation: In nucleic acids the nitrogen atoms are part of heterocyclic rings (bases) rather than being present as simple substituents that can be converted quantitatively to ammonium salts during digestion. The Kjeldahl method relies on converting nitrogen into ammonium sulphate, which works well for many organic compounds but is unsuitable for nitrogen trapped in stable heterocyclic ring systems like purines and pyrimidines.
Q.34. If a liquid compound decomposes at its boiling point, which method (s) can you choose for its purification. It is known that the compound is stable at low pressure, steam volatile and insoluble in water.
Ans. Steam distillation is appropriate.
Explanation: Because the compound is steam volatile and immiscible with water, steam distillation allows it to vaporise at a lower temperature in the presence of steam, avoiding decomposition at its normal boiling point. The vapour is condensed and the compound separated from water.
Note: Answer Questions 35-38 on the basis of information given below: "Stability of carbocations depends upon the electron releasing inductive effect of groups adjacent to positively charged carbon atom, involvement of neighbouring groups in hyperconjugation and resonance."
Q.35. "Stability of carbocations depends upon the electron releasing inductive effect of groups adjacent to positively charged carbon atom involvement of neighbouring groups in hyperconjugation and resonance."
Draw the possible resonance structures for 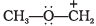 and predict which of the structures is more stable. Give reason for your answer.
and predict which of the structures is more stable. Give reason for your answer.
Ans: Two canonical structures are possible for the shown carbocation.
Q.36. Which of the following ions is more stable? Use resonance to explain your answer.
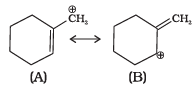
Ans. Structure A is more stable than structure B.
Explanation: Carbocation A is planar and its positive charge can be delocalised into the aromatic ring (conjugation), so resonance stabilisation operates. In contrast, B is non-planar (or less able to conjugate) and cannot delocalise the positive charge effectively; the positive charge remains localized. Therefore A, which can be stabilised by resonance with the ring, is the more stable ion.
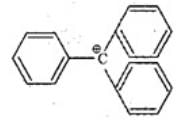
Q.37. The structure of triphenylmethyl cation is given below. This is very stable and some of its salts can be stored for months. Explain the cause of high stability of this cation.
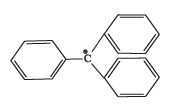
Ans. Triphenylmethyl cation is unusually stable because the positive charge is delocalised over three phenyl rings.
Explanation: The central carbocation is conjugated with each of the three benzene rings; each ring can delocalise the positive charge to its ortho and para positions, producing multiple canonical structures. In total there are several resonance forms (nine major contributors when considering the three rings), which spread the positive charge over a large volume and greatly stabilise the cation. This extensive resonance delocalisation explains the exceptional stability of the triphenylmethyl cation.
Q.38. Write structures of various carbocations that can be obtained from 2-methylbutane. Arrange these carbocations in order of increasing stability.
Ans. 2-Methylbutane gives four distinct carbocations on removal of a hydrogen atom from different carbon atoms: two primary carbocations, one secondary and one tertiary.
Explanation: Label the carbons of 2-methylbutane and consider H removal from each carbon:
- Primary carbocations (1°): removal from terminal primary carbons gives two distinct primary cations.
- Secondary carbocation (2°): removal from the carbon adjacent to the branching point gives a secondary cation.
- Tertiary carbocation (3°): removal from the branched carbon (bearing two methyls) gives a tertiary cation.
Stability order: 1° < 2° < 3°. Thus, listing the four specific cations in increasing stability: the primary cations < secondary cation < tertiary cation.
Numerically: I < IV < II < III (1° < 1° < 2° < 3°).
Q.39. Three students, Manish, Ramesh and Rajni were determining the extra elements present in an organic compound given by their teacher. They prepared the Lassaigne's extract (L.E.) independently by the fusion of the compound with sodium metal. Then they added solid FeSO4 and dilute sulphuric acid to a part of Lassaigne's extract. Manish and Rajni obtained prussian blue colour but Ramesh got red colour. Ramesh repeated the test with the same Lassaigne's extract, but again got red colour only. They were surprised and went to their teacher and told him about their observation. Teacher asked them to think over the reason for this. Can you help them by giving the reason for this observation. Also, write the chemical equations to explain the formation of compounds of different colours.
Ans. The difference is due to the composition of the Lassaigne's extract produced during fusion with sodium.
Explanation: When an organic compound contains both nitrogen and sulphur, fusion with sodium can yield different products depending on conditions and amount of sodium:
- If insufficient sodium is available, sodium thiocyanate (NaSCN) may be formed preferentially. On treatment with FeSO4 and acid this gives ferric thiocyanate, a red complex (Fe(SCN)3), producing a red colour.
- If sodium is in excess and cyanide is produced (NaCN), subsequent treatment leads to formation of Prussian blue (a ferric ferrocyanide complex), giving a blue colour.
Chemical reactions:
If Na is in excess then : 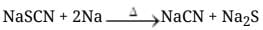
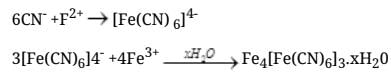
Prussian blue
Q.40. Name the compounds whose line formulae are given below :
(i) 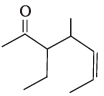
(ii) 
Ans: (i) 3-Ethyl-4-methylhept-5-en-2-one.
(ii) 3-Nitrocyclohex-1-en.
Explanation: (i) Choose the longest chain including the carbonyl (C=O) so that the >C=O group gets the lowest possible locant; assign substituents accordingly. (ii) For the cyclohexene, number the ring so that the double bond carbons receive the lowest numbers and the nitro substituent gets the correct locant (3-nitrocyclohex-1-en).
Q.41. Write structural formulae for compounds named as
(a) 1 - Bromoheptane
(b) 5 - Bromoheptanoic acid
Ans. (a) 1-Bromoheptane: CH2Br-CH2-CH2-CH2-CH2-CH2-CH3.
(b) 5-Bromoheptanoic acid: HOOC-CH2-CH2-CH(Br)-CH2-CH2-CH3 (showing the bromo on carbon-5 counting from the carboxyl carbon).
Q.42. Draw the resonance structures of the following compounds;
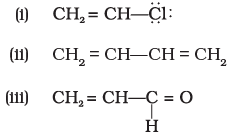
Ans. (i) Vinyl chloride (CH2=CH-Cl) has a small resonance contribution where a lone pair on Cl can donate into the π-system:
CH2=CH-Cl ↔ -CH2-CH=Cl+ (minor contributor).
(ii) 1,3-Butadiene (CH2=CH-CH=CH2) can be represented by two major resonance (conjugated) structures in which the π bonds are delocalised: CH2=CH-CH=CH2 ↔ CH2-CH=CH-CH2 (showing the different locations of double bonds).
(iii) Propenal (CH2=CH-CHO) shows resonance between the C=C and C=O systems: CH2=CH-CHO ↔ +CH2-CH=CH-O- (charge-separated minor contributor).
Q.43. Identify the most stable species in the following set of ions giving reasons:
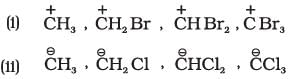
Ans. (i) CH3+ is more stable relative to analogous species bearing electron-withdrawing substituents; introduction of strongly electron-withdrawing groups increases instability of a positively charged carbon.
(ii) CCl3- is the most stable among comparable carbanions because the negative charge is dispersed by the strong -I (inductive) effect of three chlorine atoms; greater electron-withdrawing substituents stabilise a negative charge.
Explanation: Electron-withdrawing substituents stabilise negative charges but destabilise positive charges; electron-releasing groups stabilise positive charges and destabilise negative charges. Use inductive and resonance arguments to compare stability.
Q.44. Give three points of differences between inductive effect and resonance effect.
Ans:
| S.No. | Inductive effect | Resonance effect |
| 1. | It involves displacement of σ electrons in saturated compounds. | It involves displacement (delocalisation) of π electrons or lone pair electrons in unsaturated and conjugated systems. |
| 2. | Inductive effect produces a small shift of σ electrons giving rise to partial positive or negative charges along the chain. | Resonance involves redistribution of π electrons (or lone pairs) and often results in full formal charges in canonical forms, leading to greater charge separation in individual contributors. |
| 3. | Inductive effect is a short-range effect and weakens rapidly (typically up to 3-4 bonds away). | Resonance can extend along the entire conjugated system and delocalise charge over many atoms. |
Q.45. Which of the following compounds will not exist as resonance hybrid. Give reason for your answer:
(i) CH3OH (ii) R-CONH2 (iii) CH3CH = CHCH2NH2
Ans. (i) and (iii) do not show significant resonance; (ii) does.
Explanation: (i) CH3OH lacks a π system or adjacent empty p-orbital for delocalisation, so it does not show resonance. (ii) Amides (RCONH2) show resonance because the lone pair on nitrogen can delocalise into the C=O π system, producing resonance structures. (iii) In CH3CH=CHCH2NH2 the lone pair on the nitrogen is not conjugated with the C=C double bond (there is no continuous overlap), so significant resonance between the N lone pair and the double bond is not present.
Q.46. Why does SO3 act as an electrophile?
Ans: SO3 acts as an electrophile because the sulphur atom is electron-deficient due to the strongly electronegative oxygen atoms bonded to it. Resonance structures of SO3 show partial positive character on S; consequently S can accept electron pairs from nucleophiles, behaving as an electrophile.
Q.47. Resonance structures of propenal are given below. Which of these resonating structures is more stable? Give reason for your answer.
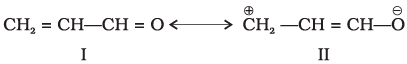
Ans. Structure I (the neutral form with full octets and no formal charges) is more stable than Structure II (charge-separated form).
Explanation: Resonance contributors that minimise formal charge separation and maintain full octets for all atoms are more important. Structure I has covalent bonds and complete octets, while Structure II involves charge separation and a carbon with incomplete octet in some visualisations; hence Structure I is the major contributor and is more stable.
Q.48. By mistake, an alcohol (boiling point 97°C) was mixed with a hydrocarbon (boiling point 68°C). Suggest a suitable method to separate the two compounds. Explain the reason for your choice.
Ans. Simple (fraction) distillation is suitable because the boiling points differ significantly.
Explanation: When two liquids differ by more than about 20°C in boiling point, simple distillation effectively separates them: the lower-boiling hydrocarbon (68°C) will vaporise and distil over first, leaving the higher-boiling alcohol (97°C) behind. If very pure separation is required, a single simple distillation will usually suffice given this substantial boiling-point difference.
Q.49. Which of the two structures (A) and (B) given below is more stabilised by resonance? Explain.
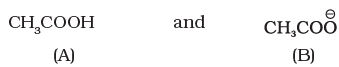
Ans. Structure (B) is more stable.
Explanation: Structure B does not involve charge separation in its canonical forms, whereas structure A requires separation of charges in at least one major contributor. Resonance forms without charge separation generally contribute more to the resonance hybrid, so B is more stabilised.
IV. Matching Type Question.
In the following questions more than one correlation is possible between options of Column I and Column II. Make as many correlations as you can.
Q.50. Match the type of mixture of compounds in Column I with the technique of separation/purification given in Column II.
| Column I | Column II |
| (i) Two solids which have different solubilities in a solvent and which do not undergo reaction when dissolved in it. | (a) Steam distillation |
| (ii) Liquid that decomposes at its boiling point | (b) Fractional distillation |
| (iii) Steam volatile liquid | (c) Simple distillation |
| (iv) Two liquids which have boiling points close to each other | (d) Distillation under reduced pressure |
| (v) Two liquids with large difference in boiling points. | (e) Crystallisation |
Explanation: (i) Different solubilities: crystallisation separates a soluble solid from impurities. (ii) Liquid that decomposes at its boiling point: distillation under reduced pressure lowers the boiling point to avoid decomposition. (iii) Steam-volatile liquid: steam distillation is used. (iv) Liquids with close boiling points: fractional distillation using a fractionating column is required. (v) Liquids with widely different boiling points: simple distillation will separate them.
| Column - I | Column II |
| (i) Two solids which have different solubility's in a solvent and which do not undergo reaction when dissolved in it. | On dissolving in solvent, the solid which is insoluble is separated out by filtration and the soluble solid is separated by crystallization. |
| (ii) Liquid that decomposes at its boiling point | Distillation under reduced pressure is done, because at low pressure vapours are formed below its boiling point and decomposition does not take place. |
| (iii) Steam volatile liquid | Liquid is converted into vapours by passing steam followed by condensation. |
| (iv) Two liquids which have boiling points close to each other | The vapours of such liquids are formed within the same temperature range and are condensed simultaneously. The technique of fractional distillation is used in such cases. In this technique, vapours of a liquid mixture are passed through a fractionating column before condensation. The fractionating column is fitted over the mouth of the round bottom flask. |
| (v) Two liquids with large difference in boiling points. | Liquids having different boiling points vaporize at different temperatures. The vapours are cooled and the liquids so formed are collected separately. |
Q.51. Match the terms mentioned in Column I with the terms in Column II.
| Column I | Column II |
| (a) Carbocation | (1) Cyclohexane and 1-hexene |
| (b) Nucleophile | (2) Conjugation of electrons of C - H σbond with empty p-orbital present at adjacent positively charged carbon. |
| (c) Hyperconjugation | (3) sp2 hybridised carbon with empty p-orbital |
| (d) Isomers | (4) Ethyne |
| (e) sp hybridization | (5) Species that can receive a pair of electrons |
| (f) Electrophile | (6) Species that can supply a pair of electrons |
| Column I | Column II | Explanation |
| (a) Carbocation | sp2-hybridised carbon with empty p-orbital | Loss of an electron pair from carbon leads to an sp2 centre with an empty p-orbital (e.g., CH3+). |
| (b) Nucleophile | Species that can supply a pair of electrons | Electron-rich species that donate an electron pair to electrophiles. |
| (c) Hyperconjugation | Conjugation of electrons of C - H σbond with empty p-orbital present at adjacent positively charged carbon | Delocalisation of σ (C-H) electrons into an adjacent empty p-orbital stabilises carbocations. |
| (d) Isomers | Cyclohexane and 1-hexene | Same molecular formula but different structures (example pair). |
| (e) sp hybridization | Ethyne | HC≡CH is an example of sp hybridisation. |
| (f) Electrophile | Species that receive a pair of electrons | Electron-deficient species or Lewis acids accept an electron pair from a nucleophile. |
Q.52. Match Column I with Column II.
| Column I | Column II |
| (a) Dumas method | (1) AgNO3 |
| (b) Kjeldahl method | (2) Silica gel |
| (c) Carius method | (3) Nitrogen gel |
| (d) Chromatography | (4) Free radicals |
| (e) Homolysis | (5) Ammonium sulphate |
| Column I | Column II | Explanation |
| (a) Dumas method | Nitrogen gel | A classical method for determining nitrogen content. |
| (b) Kjeldahl method | Ammonium sulphate | Organic nitrogen is converted to ammonium sulphate during digestion and then quantified. |
| (c) Carius method | AgNO3 | Used for halogen estimation; organic compound is heated with AgNO3 in sealed tube. |
| (d) Chromatography | Silica gel | Silica gel is a commonly used stationary phase in adsorption chromatography. |
| (e) Homolysis | Free radicals | Homolytic bond cleavage gives free radicals. |
Q.53. Match the intermediates given in Column I with their probable structure in
Column II.
| Column I | Column II |
| (i) Free radical | (a) Trigonal planar |
| (ii) Carbocation | (b) Pyramidal |
| (iii) Carbanion | (c) Linear |
| Column I | Column II |
| (i) Free radical | Free radical is formed as a result of homolytic fission and the radical carbon is sp2 hybridized; its shape is trigonal planar. |
| (ii) Carbocation | In a carbocation the positively charged carbon is sp2 hybridized and the geometry is trigonal planar. |
| (iii) Carbanion | In a carbanion the negatively charged carbon is typically sp3 with a lone pair, giving a pyramidal shape. |
Q.54. Match the ions given in Column I with their nature given in Column II.
| Column I | Column II |
(i) 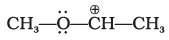 | (a) Stable due to resonance |
(ii) 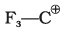 | (b) Destabilised due to inductive effect |
(iii) 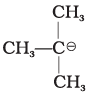 | (c) Stabilised by hyperconjugation |
(iv) 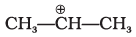 | (d) A secondary carbocation |
| Column I | Column II |
(i) 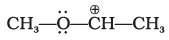 | Stabilised due to resonance; the negative or positive charge is delocalised over a conjugated system. |
(ii) 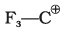 | The presence of electron-withdrawing CF3 groups exerts a strong -I effect, reducing electron density and destabilising the ion. |
(iii)  | In (CH3)C-, CH3 groups show +I effect which increases electron density on the negatively charged carbon and thus destabilises the carbanion. |
(iv) 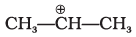 | This is a secondary carbocation; it is stabilised by hyperconjugation and the +I effect of two adjacent methyl groups. |
V. Assertion and Reason Type
In the following questions a statement of Assertion (A) followed by a statement of Reason (R) is given. Choose the correct option out of the choices given below each question.
Q.55. Assertion (A) : Simple distillation can help in separating a mixture of propan-1-ol (boiling point 97°C) and propanone (boiling point 56°C).
Reason (R) : Liquids with a difference of more than 20°C in their boiling points can be separated by simple distillation.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (i) Both A and R are correct and R is the correct explanation of A.
Explanation:
(i) Assertion: Simple distillation can separate propan-1-ol and propanone because their boiling points differ markedly.
(ii) Reason: When two liquids differ by more than about 20°C in boiling point, their vapours form at sufficiently different temperatures for simple distillation to separate them.
(iii) Justification: Both statements are true and the reason correctly explains why the assertion holds (option i).
Q.56. Assertion (A) : Energy of resonance hybrid is equal to the average of energies of all canonical forms.
Reason (R) : Resonance hybrid cannot be presented by a single structure.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (iv) A is not correct but R is correct.
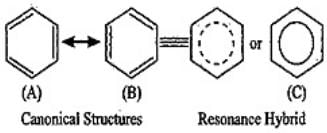
Explanation:
(i) Assertion: The energy of a resonance hybrid is not simply the numerical average of energies of its canonical forms; instead, the resonance hybrid is lower in energy (more stable) than any individual canonical form. So the assertion is incorrect.
(ii) Reason: A resonance hybrid cannot be represented by a single Lewis structure and must be depicted as a hybrid of canonical forms - this statement is correct. Therefore (iv) is the correct choice: A is not correct but R is correct.
Q.57. Assertion (A) : Pent- 1- ene and pent- 2- ene are position isomers.
Reason (R) : Position isomers differ in the position of functional group or a substituent.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (i) Both A and R are correct and R is the correct explanation of A.
Explanation:
(i) Assertion: Pent-1-ene and pent-2-ene have the same molecular formula but differ in the position of the double bond; this is position isomerism - true.
(ii) Reason: Position isomers differ in the location of a functional group, substituent or multiple bond on the carbon skeleton - true and it correctly explains the assertion. Hence option (i) is correct.
Q.58. Assertion (A): All the carbon atoms in H2C=C=CH2 are sp2 hybridized
Reason (R): In this molecule, all the carbon atoms are attached to each other by double bonds.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (iv) A is not correct but R is correct.
Explanation:
(i) Assertion: Not all carbon atoms in H2C=C=CH2 are sp2; the terminal carbons are sp2 but the central carbon is sp hybridised because it forms two σ and two π bonds. Thus the assertion is incorrect.
(ii) Reason: It is true that the carbons are connected by double bonds, but this alone does not imply all carbons are sp2; the nature and number of σ bonds on each carbon determine hybridisation. Therefore the correct choice is (iv): A is not correct but R is correct.
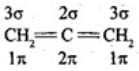
If C has 3 σ bonds it is sp2 hybridized; if C has 2 σ bonds it is sp hybridized.
Q.59. Assertion (A): Sulphur present in an organic compound can be estimated quantitatively by Carious method.
Reason (R): Sulphur is separated easily from other atoms in the molecule and gets precipitated as light yellow solid.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (iii) Both A and R are not correct.
Explanation:
(i) Assertion: Sulfur is estimated by the Carius method, but the product obtained for quantitative estimation is barium sulfate (BaSO4), which is white and insoluble; thus the statement that sulphur is precipitated as a light yellow solid is incorrect.
(ii) Reason: The reasoning given is incorrect; therefore both A and R are not correct (option iii).
Q.60. Assertion (A): Components of a mixture of red and blue inks can be separated by distributing the components between stationary and mobile phases in paper chromatography.
Reason (R): The colored components of inks migrate at different rates because paper selectively retains different components according to the difference in their partition between the two phases.
(i) Both A and R are correct and R is the correct explanation of A.
(ii) Both A and R are correct but R is not the correct explanation of A.
(iii) Both A and R are not correct.
(iv) A is not correct but R is correct.
Ans. (i) Both A and R are correct and R is the correct explanation of A.
Explanation:
(i) Assertion: Paper chromatography separates components of inks by partitioning between a stationary aqueous phase held by the paper and a mobile solvent phase - true.
(ii) Reason: The components travel at different rates because of their differing partition coefficients between the stationary and mobile phases; this correctly explains separation by paper chromatography. Hence both statements are correct and R explains A (option i).
VI. Long Answer Type
Q.61. What is meant by hybridisation? Compound CH2 = C = CH2 contains sp or sp2 hybridised carbon atoms. Will it be a planar molecule?
Ans: Hybridisation is the mixing of atomic orbitals of nearly equal energy on the same atom to form a new set of equivalent hybrid orbitals that are used in bond formation. These hybrid orbitals have characteristic geometries and energy.
Explanation: In CH2=C=CH2 (allene) the terminal carbons (C1 and C3) are sp2 hybridised (each has three σ bonds), while the central carbon (C2) is sp hybridised (it forms two σ bonds and two π bonds). Because the two terminal CH2 groups lie in mutually perpendicular planes (due to the sp hybridised central carbon and orthogonal p orbitals), the allene molecule is non-planar overall.
Q.62. Benzoic acid is a organic compound. Its crude sample can be purified by crystallization from hot water. What characteristic differences in the properties of benzoic acid and the impurity make this process of purification suitable?
Ans. Crystallisation from hot water is suitable because benzoic acid has moderate solubility in hot water but low solubility in cold water, while common impurities are either much more soluble (remain in solution on cooling) or insoluble (removed by filtration) at the crystallisation temperature.
Explanation: Upon dissolving the crude sample in hot water and cooling, benzoic acid preferentially crystallises out while impurities remain in the mother liquor (if they are more soluble) or are removed by prior filtration (if insoluble). This difference in solubility between benzoic acid and impurities makes recrystallisation an effective purification method.
Q.63. Two liquids (A) and (B) can be separated by the method of fractional distillation. The boiling point of liquid (A) is less than boiling point of liquid (B). Which of the liquids do you expect to come out first in the distillate? Explain.
Ans. Liquid A (lower boiling point) will distil first.
Explanation: In fractional distillation the vapours rising through the fractionating column become progressively enriched in the more volatile component. Because A has a lower boiling point than B it is more volatile and therefore its vapour reaches the top of the column first and condenses to give the first fraction collected.
Q.64. You have a mixture of three liquids A, B and C. There is a large difference in the boiling points of A and rest of the two liquids i.e., B and C. Boiling point of liquids B and C are quite close. Liquid A boils at a higher temperature than B and C and boiling point of B is lower than C. How will you separate the components of the mixture? Draw a diagram showing set up of the apparatus for the process.
Ans. Separate A first by simple distillation (since A's boiling point differs widely from B and C), then separate B and C by fractional distillation because their boiling points are close.
Explanation: Heat the mixture and collect the lower-boiling fraction (B or C depending on which has the lower boiling point) using fractional distillation for the close-boiling pair. Since A boils higher and is well separated by temperature, it can be left behind or collected separately by raising the temperature later. The apparatus includes a round-bottom flask, heating mantle, fractionating column for B/C separation, condenser and receiving flasks for collected fractions.
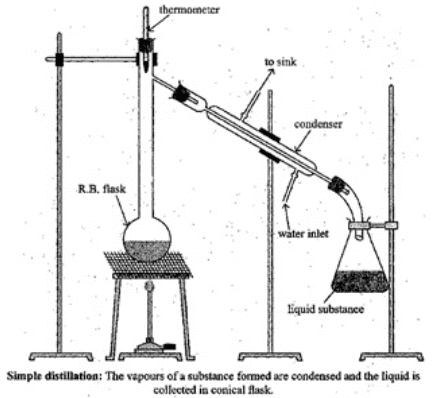
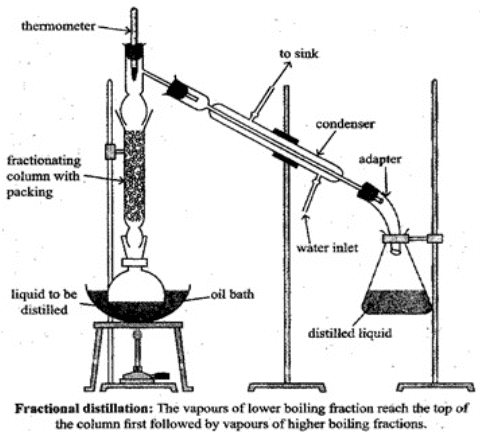
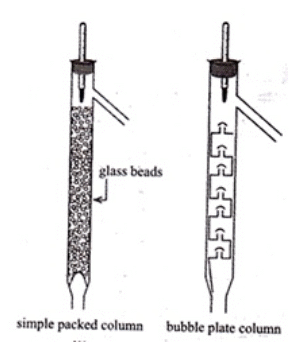
Q.65. Draw a diagram of bubble plate type fractionating column. When do we require such type of a column for separating two liquids. Explain the principle involved in the separation of components of a mixture of liquids by using fractionating column. What industrial applications does this process have?
Ans. A bubble-plate (tray) column contains a series of perforated plates (trays) stacked along the column height. Vapour rising from the reboiler bubbles through the liquid on each plate; repeated vapour-liquid contact increases the degree of separation. Use such a column when the boiling points of two liquids are close and efficient repeated equilibration stages are needed.
Explanation: Fractional distillation works on the principle of repeated vapourisation and condensation (the more volatile component preferentially vapourises and is enriched in the vapour phase). A fractionating column (packed or tray) provides many theoretical plates (equilibrium stages) so that the mixture is progressively enriched in the more volatile component as vapour rises. Industrial applications include petroleum refining (fractional distillation of crude oil), production of pure solvents, and separation of air into oxygen and nitrogen.
Q.66. A liquid with high boiling point decomposes on simple distillation but it can be steam distilled for its purification. Explain how is it possible?
Ans. Steam distillation allows vapourisation of a steam-volatile compound at a temperature lower than its normal boiling point by co-distilling it with water.
Explanation: In steam distillation the compound and water form immiscible vapour mixtures whose total vapour pressure allows evaporation at a temperature lower than the compound's decomposition temperature. Thus the compound vapourises safely at a lower temperature, is condensed and then separated from water, enabling purification without thermal decomposition.
FAQs on NCERT Exemplar: Some Basic Principles & Techniques
| 1. What are the basic principles and techniques covered in the NCERT Exemplar on Some Basic Principles & Techniques? |  |
| 2. How can the NCERT Exemplar on Some Basic Principles & Techniques help students in their exams? |  |
| 3. Are the principles and techniques covered in the NCERT Exemplar applicable to all subjects? |  |
| 4. Can the NCERT Exemplar on Some Basic Principles & Techniques be used as a stand-alone study material? |  |
| 5. Where can I access the NCERT Exemplar on Some Basic Principles & Techniques? |  |






