NCERT Exemplar: Biomolecules
Multiple Choice Questions -I
Q1. Glycogen is a branched chain polymer of α-D-glucose units in which chain is formed by C1- C4 glycosidic linkage whereas branching occurs by the formation of C1- C6 glycosidic linkage. Structure of glycogen is similar to __________.
(1) Amylose
(2) Amylopectin
(3) Cellulose
(4) Glucose
Ans. (2)
Solution. Structure of glycogen is similar to amylopectin. It is a branched chain polymer of α-D-glucose units in which the chain is formed by C1-C4 glycosidic linkage and branching occurs by C1-C6 glycosidic linkage.
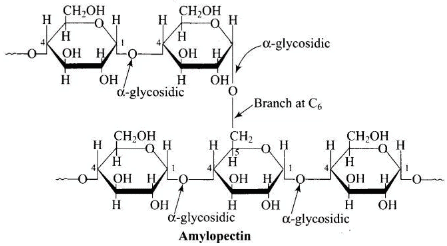
Q2. Which of the following polymer is stored in the liver of animals?
(1) Amylose
(2) Cellulose
(3) Amylopectin
(4) Glycogen
Ans. (4)
Solution. Glycogen is stored in the liver (and muscles) of animals.
Q3. Sucrose (cane sugar) is a disaccharide. One molecule of sucrose on hydrolysis gives _________.
(1) 2 molecules of glucose
(2) 2 molecules of glucose + 1 molecule of fructose
(3) 1 molecule of glucose + 1 molecule of fructose
(4) 2 molecules of fructose
Ans. (3)
Solution. Sucrose (cane sugar) is a disaccharide. One molecule of sucrose on hydrolysis gives one molecule of glucose andone molecule of fructose. 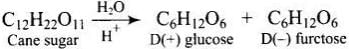
Note: Sucrose is dextrorotatory; on hydrolysis it produces a levorotatory mixture, so it is known as invert sugar. Sucrose is a non-reducing sugar while maltose and lactose are reducing sugars.
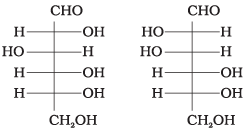
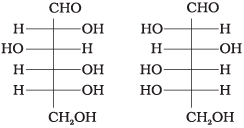
Q4. Which of the following pairs represents anomers?
(i)
(ii)
(iii)
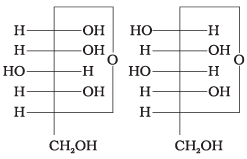
(iv)
Ans. (iii)
Solution. The isomers which differ only in the configuration of the hydroxyl group at C-1 are called anomers and are referred to as α- and β-forms.
Q5. Proteins are found to have two different types of secondary structures viz. α-helix and β-pleated sheet structure. α-helix structure of protein is stabilised by :
(1) Peptide bonds
(2) Van der Waals forces
(3) Hydrogen bonds
(4) Dipole-dipole interactions
Ans. (3)
Solution. α-helix structure of protein is stabilised by hydrogen bonds. A polypeptide chain forms hydrogen bonds by twisting into a right-handed helix with the -NH group of each amino acid residue hydrogen-bonded to the >C=O of an amino acid residue four positions earlier along the chain.
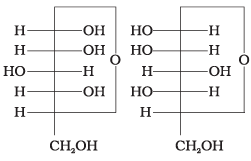
Q6. In disaccharides, if the reducing groups of monosaccharides i.e. aldehydic or ketonic groups are bonded, these are non-reducing sugars. Which of the following disaccharide is a non-reducing sugar?
(i)

(ii)
(iii)
(iv)
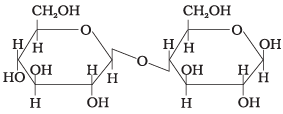
Ans. (ii)
Solution. This structure represents sucrose in which α-D-glucose and β-D-fructose are attached to each other by a C1-C2 glycosidic linkage. Since the reducing groups of glucose and fructose are involved in the glycosidic bond formation, sucrose is a non-reducing sugar.
Q7. Which of the following acids is a vitamin?
(1) Aspartic acid
(2) Ascorbic acid
(3) Adipic acid
(4) Saccharic acid
Ans. (2)
Solution. Ascorbic acid is vitamin C. Aspartic acid is an amino acid. Adipic acid and saccharic acid are dicarboxylic acids.
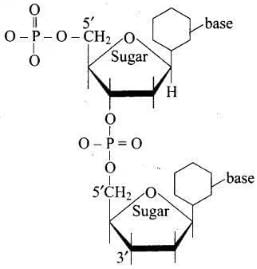
Q8. Dinucleotide is obtained by joining two nucleotides together by phosphodiester linkage. Between which carbon atoms of pentose sugars of nucleotides are these linkages present?
(1) 5′ and 3′
(2) 1′ and 5′
(3) 5′ and 5′
(4) 3′ and 3′
Ans. (1)
Solution. Phosphodiester linkages in dinucleotides connect the 5′-carbon of one nucleotide's sugar to the 3′-carbon of the next nucleotide's sugar (5′→3′ linkage).
Q9. Nucleic acids are the polymers of ______________.
(1) Nucleosides
(2) Nucleotides
(3) Bases
(4) Sugars
Ans. (2)
Solution. Nucleic acids are polymers of nucleotides in which nucleotides are linked together by phosphodiester linkages. Examples - DNA, RNA.
Q10. Which of the following statements is not true about glucose?
(1) It is an aldohexose.
(2) On heating with HI it forms n-hexane.
(3) It is present in furanose form.
(4) It does not give 2,4-DNP test.
Ans. (3)
Solution. Glucose is present predominantly in the pyranose form (six-membered ring), not the furanose form.
Q11. Each polypeptide in a protein has amino acids linked with each other in a specific sequence. This sequence of amino acids is said to be ____________.
(1) Primary structure of proteins.
(2) Secondary structure of proteins.
(3) Tertiary structure of proteins.
(4) Quaternary structure of proteins.
Ans. (1)
Solution. The sequence of amino acids in a polypeptide chain is called the primary structure of a protein.
Q12. DNA and RNA contain four bases each. Which of the following bases is not present in RNA?
(1) Adenine
(2) Uracil
(3) Thymine
(4) Cytosine
Ans. (3)
Solution. DNA contains adenine, guanine, thymine and cytosine. RNA contains adenine, uracil, guanine and cytosine. Thus, RNA does not contain thymine.
Q13. Which of the following B group vitamins can be stored in our body?
(1) Vitamin B1
(2) Vitamin B2
(3) Vitamin B6
(4) Vitamin B12
Ans. (4)
Solution. Vitamin B12 can be stored in our body because it is not freely water-soluble in the same way as other B vitamins and is stored in the liver.
Q14. Which of the following bases is not present in DNA?
(1) Adenine
(2) Thymine
(3) Cytosine
(4) Uracil
Ans. (4)
Solution. Uracil is not present in DNA; thymine is present instead.
Q15. Three cyclic structures of monosaccharides are given below which of these are anomers.
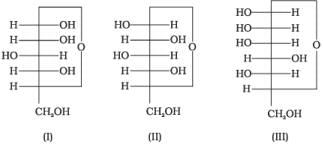
(1) I and II
(2) II and III
(3) I and III
(4) III is anomer of I and II
Ans. (1)
Solution. Cyclic structures of monosaccharides which differ in configuration at carbon-1 are anomers. Here, I and II differ only at C-1 and hence are anomers.
Q16. Which of the following reactions of glucose can be explained only by its cyclic structure?
(1) Glucose forms pentaacetate.
(2) Glucose reacts with hydroxylamine to form an oxime.
(3) Pentaacetate of glucose does not react with hydroxylamine.
(4) Glucose is oxidised by nitric acid to gluconic acid.
Ans. (3)
Solution. The pentaacetate of glucose does not react with hydroxylamine, indicating the absence of a free -CHO group in the acetylated derivative. This observation supports the cyclic (hemiacetal) structure of glucose.
Q17. Optical rotations of some compounds along with their structures are given below which of them have D configuration.
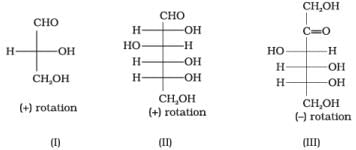
(1) I, II, III
(2) II, III
(3) I, II
(4) III
Ans. (1)
Solution. Structures I, II and III have D-configuration because the -OH group on the penultimate (last but one) asymmetric carbon is on the right, comparable to D-(+)-glyceraldehyde.
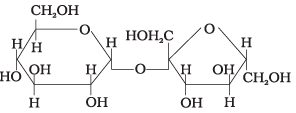
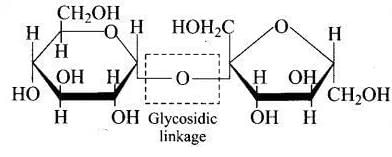
Q18. Structure of a disaccharide formed by glucose and fructose is given below. Identify anomeric carbon atoms in monosaccharide units.
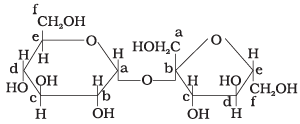
(1) 'a' carbon of glucose and ' a' carbon of fructose.
(2) 'a' carbon of glucose and 'e' carbon of fructose.
(3) 'a' carbon of glucose and ' b' carbon of fructose.
(4) 'f ' carbon of glucose and 'f ' carbon of fructose.
Ans. (3)
Solution. The anomeric carbon is the carbon adjacent to the ring oxygen in a cyclic sugar. In the given disaccharide, the anomeric carbon of glucose is marked 'a' and that of fructose is marked 'b'; both differ in the configuration of the hydroxyl group.
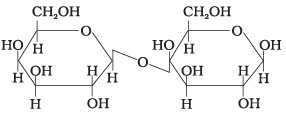
Q19. Three structures are given below in which two glucose units are linked. Which of these linkages between glucose units are between C1 and C4 and which linkages are between C1 and C6?
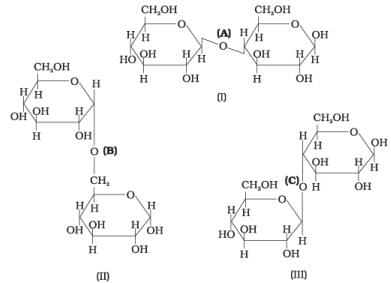
(1) (A) is between C1 and C4, (B) and (C) are between C1 and C6
(2) (A) and (B) are between C1 and C4, (C) is between C1 and C6
(3) (A) and (C) are between C1 and C4, (B) is between C1 and C6
(4) (A) and (C) are between C1 and C6, (B) is between C1 and C4
Ans. (3)
Solution. (A) and (C) show C1-C4 linkages; (B) shows a C1-C6 linkage.
Multiple Choice Questions -II
Note: In the following questions two or more options may be correct.
Q20. Carbohydrates are classified on the basis of their behaviour on hydrolysis and also as reducing or non-reducing sugar. Sucrose is a ________.
(1) Monosaccharide
(2) Disaccharide
(3) Reducing sugar
(4) Non-reducing sugar
Ans. (2, 4)
Solution. Sucrose is a disaccharide and a non-reducing sugar.
Q21. Proteins can be classified into two types on the basis of their molecular shape i.e., fibrous proteins and globular proteins. Examples of globular proteins are :
(1) Insulin
(2) Keratin
(3) Albumin
(4) Myosin
Ans. (1, 3)
Solution. Globular proteins are roughly spherical and soluble in water; insulin and albumin are examples. Keratin and myosin are fibrous proteins.
Q22. Which of the following carbohydrates are branched polymer of glucose?
(1) Amylose
(2) Amylopectin
(3) Cellulose
(4) Glycogen
Ans. (2, 4)
Solution. Amylopectin and glycogen are branched polymers of glucose. Amylose is largely unbranched; cellulose is a linear polymer of β-D-glucose.
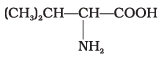
Q23. Amino acids are classified as acidic, basic or neutral depending upon the relative number of amino and carboxyl groups in their molecule. Which of the following are acidic?
(1)
(2)
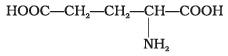
(3)
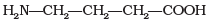
(4)
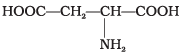
Ans. (2, 4)
Solution. Amino acids with more than one -COOH group relative to -NH2 are acidic. The structures in IMG_19 and IMG_21 represent acidic amino acids.
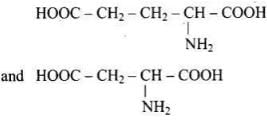
Q24. Lysine,
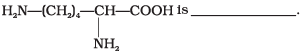
(1) α-Amino acid
(2) Basic amino acid
(3) Amino acid synthesised in body
(4) β-Amino acid
Ans. (1, 2)
Solution. Lysine is an α-amino acid. It is basic because it contains more -NH2 groups than -COOH groups. It is an essential amino acid and must be obtained from the diet.
Q25. Which of the following monosaccharides are present as five membered cyclic structure (furanose structure)?
(1) Ribose
(2) Glucose
(3) Fructose
(4) Galactose
Ans. (1, 3)
Solution. Ribose and fructose commonly adopt five-membered ring (furanose) forms. Glucose and galactose predominantly form six-membered (pyranose) rings.
Q26. In fibrous proteins, polypeptide chains are held together by ___________.
(1) Van der Waals forces
(2) Disulphide linkage
(3) Electrostatic forces of attraction
(4) Hydrogen bonds
Ans. (2, 4)
Solution. Fibrous proteins are stabilised by disulphide linkages (covalent S-S bonds) and hydrogen bonds; electrostatic interactions and van der Waals forces also contribute in some cases.
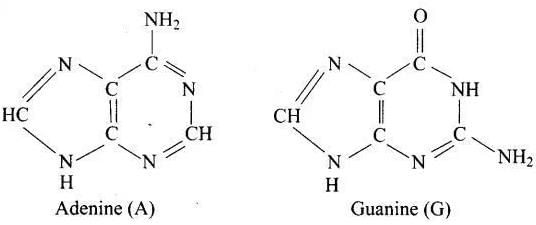
Q27. Which of the following are purine bases?
(1) Guanine
(2) Adenine
(3) Thymine
(4) Uracil
Ans. (1, 2)
Solution. Purines have fused six- and five-membered nitrogen heterocycles. Adenine and guanine are purines. Thymine and uracil are pyrimidines.
Q28. Which of the following terms are correct about enzyme?
(1) Proteins
(2) Dinucleotides
(3) Nucleic acids
(4) Biocatalysts
Ans. (1, 4)
Solution. Most enzymes are protein molecules and they act as biocatalysts for biochemical reactions.
Short Answer Type Questions
Q29. Name the sugar present in milk. How many monosaccharide units are present in it? What are such oligosaccharides called?
Ans. Lactose is present in milk. It contains two monosaccharide units: glucose and galactose. Such oligosaccharides with two monosaccharide units are called disaccharides.
Q30. How do you explain the presence of all the six carbon atoms in glucose in a straight chain?
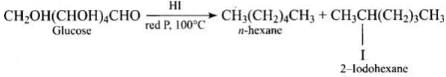
Ans. Glucose on prolonged heating with HI and red phosphorus gives n-hexane (HI in excess).
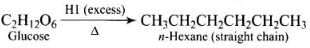
Q31. In nucleoside a base is attached at 1′ position of sugar moiety. Nucleotide is formed by linking of phosphoric acid unit to the sugar unit of nucleoside. At which position of sugar unit is the phosphoric acid linked in a nucleoside to give a nucleotide?
Ans. Phosphoric acid unit is linked preferentially at the 5′-position of the sugar moiety of a nucleoside to give a nucleotide.
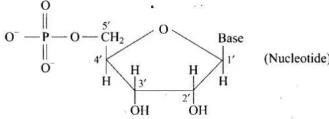
Q32. Name the linkage connecting monosaccharide units in polysaccharides.
Ans. The monosaccharide units are linked through glycosidic linkages in polysaccharides.
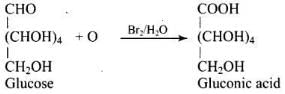
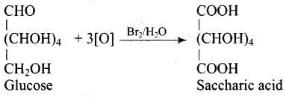
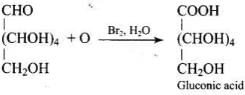
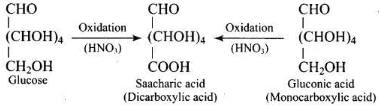
Q33. Under what conditions glucose is converted to gluconic and saccharic acid?
Ans. Glucose is converted to gluconic acid by oxidation with bromine water (Br2/H2O) and to saccharic (glucaric) acid by oxidation with concentrated HNO3.
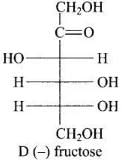
Q34. Monosaccharides contain carbonyl group hence are classified, as aldose or ketose. The number of carbon atoms present in the monosaccharide molecule are also considered for classification. In which class of monosaccharide will you place fructose?
Ans. Fructose has molecular formula C6H12O6; it contains six carbons and a keto group, so it is classified as a ketohexose.
Q35. The letters 'D' or 'L' before the name of a stereoisomer of a compound indicate the correlation of configuration of that particular stereoisomer. This refers to their relation with one of the isomers of glyceraldehyde. Predict whether the following compound has 'D' or 'L' configuration.
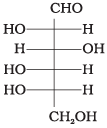
Ans. Since the -OH group at the penultimate chiral carbon atom (C5) is on the left, the given compound has the L-configuration.
Q36. Aldopentoses named as ribose and 2-deoxyribose are found in nucleic acids. What is their relative configuration?
Ans. Both the aldopentoses have D-configuration.
Q37. Which sugar is called invert sugar? Why is it called so?
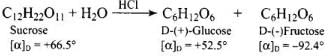
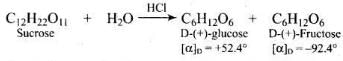
Ans. Sucrose is called invert sugar. Aqueous sucrose is dextrorotatory ([α]D = +66.5°). On hydrolysis (by dilute acids or the enzyme invertase) sucrose yields equimolar D-(+)-glucose and D-(-)-fructose.
Q38. Amino acids can be classified as α-, β-, γ-, δ- and so on depending upon the relative position of amino group with respect to carboxyl group. Which type of amino acids form polypetide chain in proteins?
Ans. α-Amino acids form polypeptide chains in proteins.
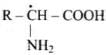
Q39. α-Helix is a secondary structure of proteins formed by twisting of polypeptide chain into right handed screw like structures. Which type of interactions are responsible for making the α-helix structure stable?
Ans. In the α-helix structure, stability arises from intramolecular hydrogen bonding between the N-H group of one peptide linkage and the >C=O group of an amino acid residue roughly four positions earlier in the chain.
Q40. Some enzymes are named after the reaction, where they are used. What name is given to the class of enzymes which catalyse the oxidation of one substrate with simultaneous reduction of another substrate.
Ans. Oxidoreductases.
Q41. During curdling of milk, what happens to sugar present in it?
Ans. The milk sugar lactose is fermented by lactic acid bacteria to produce lactic acid during curdling of milk.
Q42. How do you explain the presence of five -OH groups in glucose molecule?
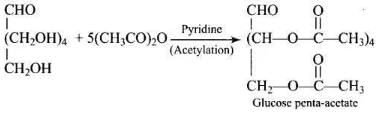
Ans. Glucose gives a penta-acetyl derivative on acetylation with acetic anhydride, confirming the presence of five -OH groups.
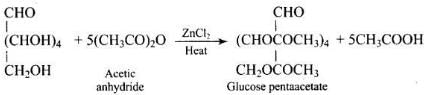
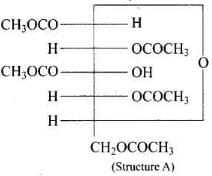
Q43. Why does compound (A) given below not form an oxime?
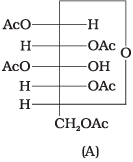
Ans. Glucose pentaacetate (structure A) does not have a free hemiacetal -OH at C-1 and therefore cannot open to the aldehyde (open-chain) form to give a free -CHO group; consequently it does not form an oxime with hydroxylamine.
Q44. Why must vitamin C be supplied regularly in diet?
Ans. Vitamin C is water soluble and excess amounts are readily excreted in urine; it is not stored in the body to any significant extent and therefore must be supplied regularly in the diet.
Q45. Sucrose is dextrorotatory but the mixture obtained after hydrolysis is laevorotatory. Explain.
Ans. Sucrose is dextrorotatory ([α]D = +66.5°). On hydrolysis it yields equimolar D-(+)-glucose and D-(-)-fructose.
Q46. Amino acids behave like salts rather than simple amines or carboxylic acids. Explain.
Ans. In aqueous solution, amino acids exist as zwitterions: the -COOH group loses a proton to become -COO⁻ and the -NH2 group gains a proton to become -NH3⁺, so they behave like internal salts.
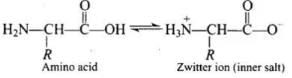
Q47. Structures of glycine and alanine are given below. Show the peptide linkage in glycylalanine.
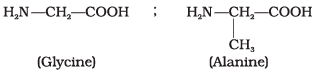
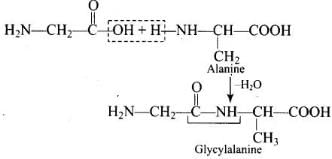
Ans. The carboxyl group of glycine reacts with the amino group of alanine to form glycylalanine via a peptide (-CO-NH-) linkage.
Q48. Protein found in a biological system with a unique three-dimensional structure and biological activity is called a native protein. When a protein in its native form, is subjected to a physical change like change in temperature or a chemical change like, change in pH, denaturation of protein takes place. Explain the cause.
Ans. Denaturation disturbs hydrogen bonds and other weak interactions (hydrophobic interactions, ionic bonds and sometimes disulphide bridges). As a result, the secondary and tertiary structure are disrupted, the globular protein unfolds and loses biological activity, while the primary sequence (peptide bonds) remains intact.
Q49. Activation energy for the acid catalysed hydrolysis of sucrose is 6.22 kJ mol-1, while the activation energy is only 2.15 kJ mol-1 when hydrolysis is catalysed by the enzyme sucrase. Explain.
Ans. Enzymes are biological catalysts that provide an alternative reaction pathway with a lower activation energy. Sucrase lowers the activation energy for sucrose hydrolysis from 6.22 kJ mol-1 to 2.15 kJ mol-1, increasing the reaction rate significantly.
Q50. How do you explain the presence of an aldehydic group in a glucose molecule?
Ans. Glucose reacts with hydroxylamine to form an oxime and adds HCN to give a cyanohydrin; on mild oxidation with bromine water glucose gives gluconic acid (oxidation of the aldehyde to carboxylic acid) indicating a free aldehydic group in the open chain form.
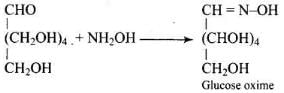
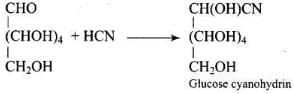
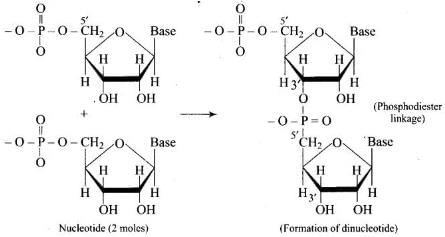
Q51. Which moieties of nucleosides are involved in the formation of phosphodiester linkages present in dinucleotides? What does the word diester in the name of linkage indicate? Which acid is involved in the formation of this linkage?
Ans. Nucleosides become nucleotides when a phosphoric acid group is esterified at the 5′-OH of the sugar. Two nucleotides are joined by a phosphodiester linkage between the 3′-OH of one sugar and the 5′-phosphate of the adjacent sugar (5′→3′). The term diester indicates that the phosphate is esterified to two alcohol groups (two ester bonds). Phosphoric acid (H₃PO₄) is involved in forming these linkages.
Q52. What are glycosidic linkages? In which type of biomolecules are they present?
Ans. Glycosidic linkages are ether-like (C-O-C) bonds formed between the anomeric carbon of one monosaccharide and a hydroxyl group of another monosaccharide with elimination of water. They are present in disaccharides, oligosaccharides and polysaccharides.
Q53. Which monosaccharide units are present in starch, cellulose and glycogen and which linkages link these units?
Ans. Starch and glycogen are composed of α-D-glucose units linked mainly by α-(1→4) glycosidic bonds, with branching via α-(1→6) bonds (amylopectin in starch; glycogen is more highly branched). Cellulose is composed of β-D-glucose units linked by β-(1→4) glycosidic bonds.
Q54. How do enzymes help a substrate to be attacked by the reagent effectively?
Ans. The active site of an enzyme binds the substrate and orients it in a favourable position and environment for reaction; this lowers activation energy and facilitates effective attack by reagents or catalytic groups in the enzyme.
Q55. Describe the term D- and L- configuration used for amino acids with examples.
Ans. The D- and L- notation refers to the configuration of a molecule relative to D-(+)-glyceraldehyde. In the Fischer projection, if the -OH group on the penultimate carbon (the chiral carbon furthest from the carbonyl) is on the right, the sugar is D; if on the left, it is L. Most naturally occurring sugars are D-series (e.g., D-glucose, D-ribose, D-fructose). For amino acids, the convention uses the same relative assignment; most naturally occurring α-amino acids are of the L-configuration.
Q56. How will you distinguish 1° and 2° hydroxyl groups present in glucose? Explain with reactions.
Ans. On acetylation with acetic anhydride (pyridine or traces of H2SO4), glucose gives a penta-acetate indicating five -OH groups present on different carbons; one is primary (1°) at C-6 (CH2OH) and four are secondary (2°) at C-2 to C-5.
Q57. Coagulation of egg white on boiling is an example of denaturation of protein. Explain it in terms of structural changes.
Ans. On boiling, the soluble globular protein albumin unfolds and aggregates to form insoluble fibrous material. Denaturation destroys the secondary and tertiary structures (and biological activity) while the primary sequence of amino acids (peptide bonds) remains intact.
Matching Type Questions
Note: Match the items of Column I and Column II in the following questions.
More than one option in Column II may match with the items given in Column I.
Q58. Match the vitamins given in Column I with the deficiency disease they cause given in Column II.
Column I (Vitamins) | Column II (Diseases) |
(i) Vitamin A | (a) Pernicious anaemia |
(ii) Vitamin B1 | (b) Increased blood clotting time |
(iii) Vitamin B12 | (c) Xerophthalmia |
(iv) Vitamin C | (d) Rickets |
(v) Vitamin D | (e) Muscular weakness |
(vi) Vitamin E | (f) Night blindness |
(vii) Vitamin K | (g) Beri Beri |
(h) Bleeding gums | |
(i) Osteomalacia |
Ans. (i → c, f), (ii → g), (iii → a), (iv → h), (v → d, i), (vi → e), (vii → b)
Q59. Match the following enzymes given in Column I with the reactions they catalyse given in Column II.
Column I (Enzymes) | Column II (Reactions) |
(i) Invertase | (a) Decomposition of urea into NH3 and CO2 |
(ii) Maltase | (b) Conversion of glucose into ethyl alcohol |
(iii) Pepsin | (c) Hydrolysis of maltose into glucose |
(iv) Urease | (d) Hydrolysis of cane sugar |
(v) Zymase | (e) Hydrolysis of proteins into peptides |
Ans. (i → d), (ii → c), (iii → e), (iv → a), (v → b)
ASSERTION AND REASON TYPE QUESTIONS
Note : In the following questions a statement of assertion followed by a statement of reason is given. Choose the correct answer out of the following choices.
Q60. Assertion: D (+) - Glucose is dextrorotatory in nature.
Reason: 'D' represents its dextrorotatory nature.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (iii)
Solution. 'D' corresponds to the configuration relative to D-glyceraldehyde (-OH on the right at the penultimate carbon), not directly to optical rotation. Thus D-glucose happens to be dextrorotatory, but 'D' does not mean dextrorotatory.
Q61. Assertion: Vitamin D can be stored in our body.
Reason: Vitamin D is fat soluble vitamin.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (i)
Solution. Vitamin D is fat soluble and can be stored in body fat and liver; the reason explains the assertion.
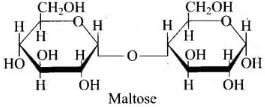
Q62. Assertion: β-glycosidic linkage is present in maltose,
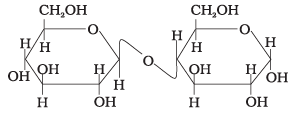
Reason: Maltose is composed of two glucose units in which C-1 of one glucose unit is linked to C-4 of another glucose unit.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (iv)
Solution. The linkage in maltose is an α-(1→4) glycosidic linkage, not β. The reason correctly states the connectivity but the assertion about β is wrong.
Q63. Assertion: All naturally occurring α-aminoacids except glycine are optically active.
Reason: Most naturally occurring amino acids have L-configuration.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (v)
Solution. All α-amino acids except glycine contain a chiral (asymmetric) α-carbon and are therefore optically active; most naturally occurring amino acids are L-configured. The reason is correct but does not explain why glycine is achiral.
Q64. Assertion: Deoxyribose, C5H10O4 is not a carbohydrate.
Reason: Carbohydrates are hydrates of carbon so compounds which follow Cx(H2O)y formula are carbohydrates.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (ii)
Solution. Deoxyribose is a carbohydrate (a pentose sugar). The simple formula Cx(H2O)y is not a reliable criterion for all carbohydrates; carbohydrates are better defined as polyhydroxy aldehydes or ketones or compounds that give such on hydrolysis.
Q65. Assertion: Glycine must be taken through diet.
Reason: It is an essential amino acid.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (ii)
Solution. Glycine is non-essential because it can be synthesised in the body; it does not need to be obtained from the diet.
Q66. Assertion: In presence of enzyme, substrate molecule can be attacked by the reagent effectively.
Reason: Active sites of enzymes hold the substrate molecule in a suitable position.
(i) Assertion and reason both are correct statements and reason explains the assertion.
(ii) Both assertion and reason are wrong statements.
(iii) Assertion is correct statement and reason is wrong statement.
(iv) Assertion is wrong statement and reason is correct statement.
(v) Assertion and reason both are correct statements but reason does not explain assertion.
Ans. (i)
Solution. The active site binds and orients the substrate, allowing effective interaction with catalytic groups which lowers the activation energy and enhances reaction rate.
Long Answer Type Questions
Q67. Write the reactions of D-glucose which can't be explained by its open-chain structure. How can cyclic structure of glucose explain these reactions?
Ans. The open-chain structure of glucose explains many reactions, but certain observations require a cyclic (hemiacetal) structure:
(i) Despite having an aldehydic (-CHO) group, glucose does not show some aldehyde characteristic reactions: it does not react with ammonia, does not form bisulfite addition products, and does not give Schiff's or 2,4-DNP tests in its usual form.
(ii) Glucose forms an oxime with hydroxylamine, but glucose pentaacetate does not form an oxime, showing acetylation removes the free hemiacetal hydroxyl and prevents ring opening to the aldehyde.
(iii) D-glucose exists as two stable crystalline stereoisomers, α and β, which have different melting points and optical rotations; this is explained by different configurations at the anomeric carbon in cyclic forms.
(iv) Aqueous glucose solutions show mutarotation: the specific rotation of α-glucose (+110°) changes towards +52.5° and that of β-glucose (+19.7°) also changes to the same equilibrium value; this is due to interconversion via the open chain form and equilibria between cyclic anomers.
(v) Glucose gives two methyl glucosides (α and β) on reaction with methanol and HCl, demonstrating that the anomeric carbon can form glycosides - a property of cyclic hemiacetals.
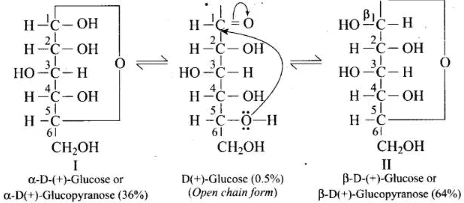
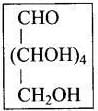
Q68. On the basis of which evidences D-glucose was assigned the following structure?
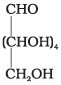
Ans. This open-chain structure for D-glucose was assigned based on the following evidences:
1. Molecular formula: Elemental analysis established the formula C6H12O6.
2. Straight chain evidence:
(i) Reduction of glucose with Na/Hg or NaBH4gives sorbitol (glucitol), a hexahydric alcohol, indicating six carbon atoms in a chain with hydroxyls.

(ii) Prolonged heating with HI and red phosphorus yields n-hexane (and 2-iodohexane), showing that the six carbons are arranged in a straight chain.
3. Presence of five hydroxyl groups: Acetylation gives a pentaacetate, confirming five free -OH groups located on different carbons (multiple -OH on one carbon would be unstable).
4. Presence of a primary alcoholic group: Oxidation to saccharic (glucaric) acid shows oxidation of a terminal primary alcohol (CH2OH), consistent with a terminal -CH2OH group.
5. Presence of an aldehyde (-CHO) group: Reaction with hydroxylamine to form an oxime and formation of cyanohydrin indicate a carbonyl functionality consistent with an aldehyde in the open chain form.
On the basis of these observations the open-chain CHO structure of glucose was proposed.
Q69. Carbohydrates are essential for life in both plants and animals. Name the carbohydrates that are used as storage molecules in plants and animals, also name the carbohydrate which is present in wood or in the fibre of cotton cloth.
Ans.
(i) Plants store carbohydrates mainly as starch (amylose and amylopectin) and sucrose is used for transport.
(ii) Animals store carbohydrates mainly as glycogen (often called animal starch) in liver and muscle; glycogen is broken down to glucose when required.
(iii) Cellulose is present in wood and the fibres of cotton cloth; it is a structural polysaccharide composed of β-(1→4) linked D-glucose units.
Q70. Explain the terms primary and secondary structure of proteins. What is the difference between α-helix and β-pleated sheet structure of proteins?
Ans. Primary structure: The primary structure of a protein is the linear sequence of amino acids in a polypeptide chain connected by peptide bonds. Any change in this sequence yields a different protein. Primary structure is determined by chemical or enzymatic hydrolysis into smaller fragments.
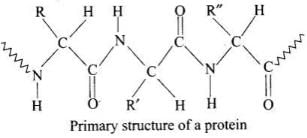

Secondary structure: The secondary structure describes local folding patterns of the polypeptide backbone stabilised by hydrogen bonds between the >C=O and N-H groups of the peptide linkages.
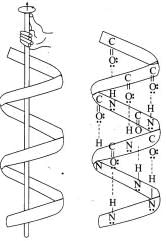
α-Helix: The polypeptide chain coils into a right-handed helix (3.6 residues per turn), with hydrogen bonds formed between the N-H of an amino acid and the >C=O of the residue four positions earlier, producing a stable helical structure often referred to as the 3.613helix.
β-Pleated sheet: Polypeptide chains lie side by side in an extended zigzag conformation, hydrogen bonded between neighbouring chains (or between different segments of the same chain). The arrangement resembles pleated sheets with alternating side chains above and below the sheet plane; multiple strands can stack to form a larger sheet.
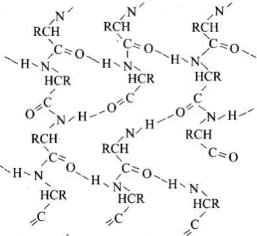
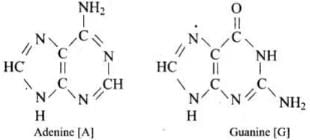
Q71. Write the structures of fragments produced on complete hydrolysis of DNA. How are they linked in DNA molecule? Draw a diagram to show pairing of nucleotide bases in double helix of DNA.
Ans. Complete hydrolysis of DNA yields:
(i) A pentose sugar (2-deoxyribose),
(ii) Phosphoric acid (H₃PO₄),
(iii) Nitrogenous bases: adenine (A), guanine (G), cytosine (C) and thymine (T).
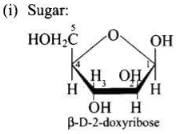
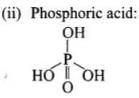
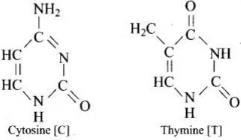
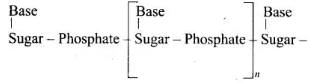
A base attached to the 1′-position of the sugar is a nucleoside. When the nucleoside is esterified with phosphoric acid at the 5′-position, a nucleotide is formed. Nucleotides are linked by phosphodiester bonds between the 3′-OH of one sugar and the 5′-phosphate of the next (5′→3′ backbone).
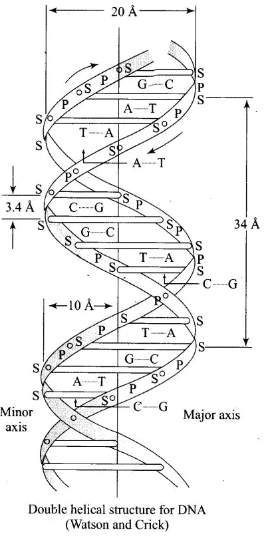
In the DNA double helix, two antiparallel polynucleotide chains coil about each other and are held together by hydrogen bonds between complementary bases: A pairs with T by two hydrogen bonds; G pairs with C by three hydrogen bonds.
FAQs on NCERT Exemplar: Biomolecules
| 1. What are the main types of biomolecules? |  |
| 2. How do enzymes function as biological catalysts? |  |
| 3. What is the significance of DNA and RNA in living organisms? |  |
| 4. What are the functions of carbohydrates in biological systems? |  |
| 5. How do lipids contribute to cellular structure and function? |  |







