UPSC Exam > UPSC Notes > Science & Technology CSE > NCERT Summary: Summary of Chemistry- 1
NCERT Summary: Summary of Chemistry- 1
Acid, Base and Salts
Acid
- The word acid is derived from Latin and means "sour". The sour taste of many fruits and some vegetables is due to acids present in them. Digestive fluids of many animals and humans also contain acids.
- An acid is a compound which, on dissolving in water, produces hydronium ions (H3O+) as the positive ions responsible for the characteristic properties of acidity.
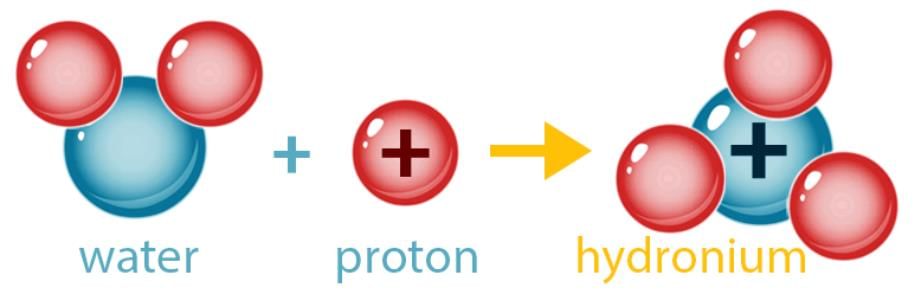
- Many acids contain hydrogen. Examples are hydrochloric acid (HCl), sulphuric acid (H2SO4) and nitric acid (HNO3). However, not every hydrogen-containing compound is an acid (for example, water, H2O, and methane, CH4).
- Acids may be classified by:
- strength (strong or weak);
- basicity (number of replaceable hydrogen atoms);
- concentration (dilute or concentrated);
- presence of oxygen (oxyacids versus non-oxyacids).
- The strength of an acid refers to the extent of dissociation in water, i.e. the concentration of hydronium ions. Some acids dissociate completely (strong acids) while others dissociate only partially (weak acids).
- Strong acid: dissociates almost completely in water; nearly all hydrogen ions exist as hydronium ions. Examples: HCl, H2SO4, HNO3.
- Weak acid: dissociates only partially in water; many molecules remain undissociated. Examples: acetic acid, formic acid, carbonic acid.

Comparing strong and weak acids
- Acids are generally sour in taste. Indicators are substances that show different colours in acidic and basic media, and are used to test whether a solution is acidic or basic. Natural indicators include turmeric, litmus and petals of china rose (Gudhal).
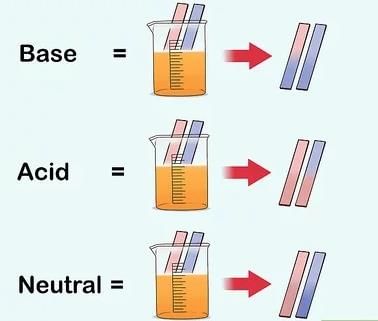
- Litmus is the most commonly used natural indicator, obtained from lichens. It is mauve in distilled water, turns red in acidic solutions and blue in basic solutions. Litmus is available as a solution or as strips of paper (red and blue litmus papers).
Change in color of litmus paper
- Solutions that do not change the colour of either red or blue litmus paper are called neutral solutions; such substances are neither acidic nor basic.
- Many acids are corrosive and can burn skin and dissolve metals; appropriate safety precautions are required when handling them.
Bases and Alkalis
- A base is a substance that produces hydroxide ions (OH-) when dissolved in water. Common bases are metal hydroxides (MOH). Examples: sodium hydroxide (NaOH) and calcium hydroxide (Ca(OH)2). A base that dissolves in water giving OH- ions is called an alkali.
- Bases neutralise acids. A base can therefore be defined as a substance that reacts with an acid to give a salt and water. Alkalis may be strong or weak; stronger alkalis produce more OH- ions in solution.
- The acidic property of an acid is due to H+ (hydrogen) ions, while the basic property of a base is due to OH- ions. When an acid and a base react, H+ combines with OH- to form neutral water.
- Strong base: dissociates completely in water and produces a high concentration of OH-. Examples: NaOH, KOH, Ca(OH)2.
- Weak base: dissociates only partially in water. Examples: Mg(OH)2, ammonium hydroxide (NH4OH).
- Bases are usually bitter in taste and feel soapy or slippery to touch. Strong alkalis such as NaOH and KOH are highly corrosive (caustic) and can damage organic tissue. NaOH is often called caustic soda and KOH as caustic potash.
pH
- A scale for measuring hydrogen ion concentration in a solution, called pH scale has been developed. The p in pH stands for 'potenz' in German, meaning power. On the pH scale we can measure pH from 0 (very acidic) to 14 (very alkaline). pH should be thought of simply as a number that indicates the acidic or basic nature of a solution. Higher the hydronium ion concentration, lower is the pH value. The pH of a neutral solution is 7.
- Values less than 7 on the pH scale represent an acidic solution. As the pH value increases from 7 to 14, it represents an increase in OH- ion concentration in the solution, that is, increase in the strength of alkali. Generally paper impregnated with the universal indicator is used for measuring pH.
- There are chemicals that change color at different pH values. These are called indicators. One of the most famous is Litmus. This substance turns red when the pH is less than 7 (acidic) and turns blue when the pH is greater than 7 (basic).
Salts
- A salt is formed when an acid reacts with a base and neutralisation occurs; H+ and OH- combine to form water, while the remaining ions form the salt.
- Common salts with everyday and industrial uses include sodium chloride (NaCl), sodium carbonate (Na2CO3), sodium hydrogen carbonate (NaHCO3), and sodium hydroxide (NaOH).
- In solution, salt ions normally remain dissolved. A salt crystallises when the solvent (water) is removed. Some salts are insoluble and precipitate when formed.
- The pH of a salt solution depends on the strengths of the parent acid and base:
- salt of a strong acid and a strong base → neutral (pH ≈ 7);
- salt of a strong acid and a weak base → acidic (pH < 7);
- salt of a strong base and a weak acid → basic (pH > 7).
Atomic Structure
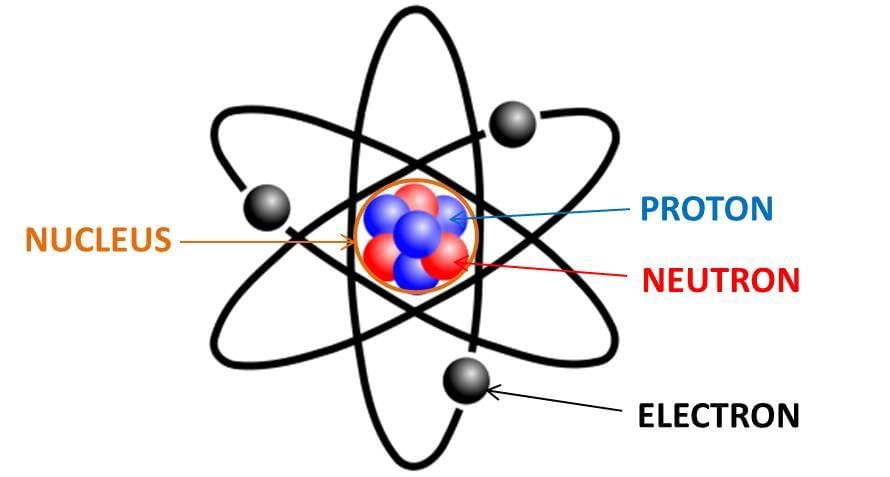
- An atom is the smallest particle of an element that can exist independently and retain the chemical properties of that element. Atoms are composed of subatomic particles: electrons, protons and neutrons.
An atom
Dalton's Atomic Theory
John Dalton proposed a theory of matter (1805) to explain chemical combination laws. The principal postulates are:
- All substances are made of tiny, indivisible particles called atoms.
- Atoms of the same element are identical in mass and properties; atoms of different elements differ.
- Atoms cannot be created, divided, or destroyed in chemical reactions.
- Atoms combine in simple whole-number ratios to form molecules (compounds).
- An atom is the smallest unit that participates in chemical reactions.
Representation of an Atom by a Symbol
- Dalton introduced the idea of using symbols to represent atoms. A chemical symbol of an element is a one- or two-letter code, usually derived from the English or Latin name. The first letter is capitalised and the second letter, if present, is lowercase.
- Examples: Hydrogen - H; Aluminium - Al; Cobalt - Co; Chlorine - Cl; Zinc - Zn.
- Certain symbols come from Latin or other classical names: iron - Fe (Ferrum), sodium - Na (Natrium), potassium - K (Kalium).
Size of the Atom / Elements
- Atoms are extremely small. The hydrogen atom, the smallest atom, has a diameter on the order of 5 × 10-11 m. Atomic radii are typically expressed in nanometres (nm), where 1 nm = 10-9 m.
Atomic Mass
- Atomic masses are measured relative to a chosen standard. Early scales used hydrogen (mass 1) or oxygen (mass 16) as standards. Since 1961, the unified scale uses the carbon-12 isotope as the standard: one atomic mass unit (1 u or 1 A.M.U.) is defined as exactly one twelfth of the mass of a carbon-12 atom.
- One atomic mass unit equals approximately 1.66 × 10-24 g.
- Relative atomic mass is a dimensionless number expressing how heavy an atom is compared with 1/12 of a carbon-12 atom.
One atomic mass unit is equal to exactly one twelfth (1/12) of the mass of a carbon-12 atom.
Molecule
- A molecule is a group of two or more atoms held together by chemical bonds. It is the smallest particle of a substance that can exist independently and retain its chemical properties.
- Molecules of an element consist of only one type of atom; many nonmetals form diatomic molecules in their elemental form (for example, O2 is diatomic oxygen). If three oxygen atoms combine, the resultant species is ozone (O3).
- The number of atoms in a molecule is called its atomicity. Atoms of different elements join in definite proportions to form molecules of compounds.
- Compounds composed of metals and non-metals often exist as charged particles called ions. A negatively charged ion is an anion, and a positively charged ion is a cation.
- A group of atoms with a net charge is a polyatomic ion.

Chemical Formulae
- A chemical formula symbolically represents the composition of a compound.
- Valency (combining capacity) of an element is used to determine how atoms combine to form compounds. Valency can be pictured as the number of bonds an atom can form.
- Binary compounds (two different elements) are written using the symbols of the constituent elements and their valencies, often using the crossover method to find the simplest whole-number ratio.
- The formula unit of an ionic compound represents the simplest whole-number ratio of cations to anions in the solid structure.
Molecular Mass and Formula Unit Mass
- The molecular mass (molar mass expressed per molecule) is the sum of atomic masses of all atoms in a molecule, expressed in atomic mass units (u).
- The formula unit mass applies to ionic substances and is the sum of atomic masses in the empirical formula unit.
- Relative atomic masses are determined with respect to a carbon-12 atom assigned a mass of 12.
Mole Concept
- A mole is a collection of particles chosen for convenience in chemical calculations. By definition, one mole of a substance contains \(6.023 \times 10^{23}\) elementary entities (atoms, molecules, ions or other particles). This constant is known as Avogadro's number.
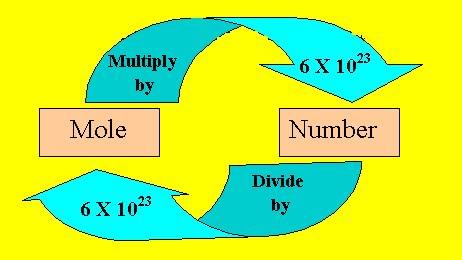
- The mass of one mole of a substance (in grams) equals its relative molecular or formula mass; this is the molar mass. At standard temperature and pressure (STP), one mole of an ideal gas occupies 22.4 L (molar volume).
- Discovery credits: J. J. Thomson discovered the electron and E. Goldstein observed positive rays (leading later to the idea of protons). Early models proposed electrons embedded in positive matter; later experiments led to new models.
Rutherford's Model of the Atom
- Rutherford's alpha-particle scattering experiment demonstrated that most of the atom's mass and all its positive charge are concentrated in a very small central region called the nucleus, with electrons moving around it. The model explained the nuclear concentration of mass but could not account for the stability and discrete spectral lines of atoms.
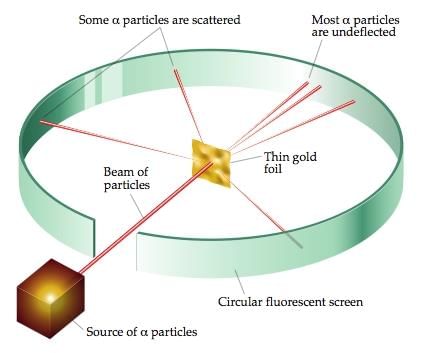
Rutherford's Alpha-particle Scattering Experiment
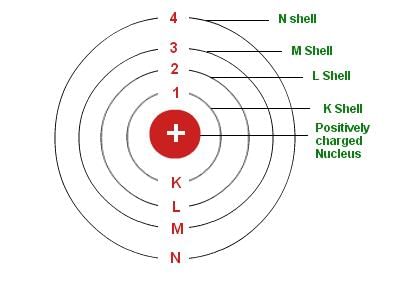
Bohr's Model
- Niels Bohr proposed that electrons orbit the nucleus in stable, discrete energy levels or shells. Electrons can move between shells by absorbing or emitting specific quanta of energy. Atoms with complete outer shells are more stable and less chemically reactive.
Neil's Bohr's Model of the Atom
Discovery of the Neutron
- J. Chadwick discovered the neutron-a neutral particle residing in the nucleus-completing the picture of the three main subatomic particles: electrons (negative), protons (positive) and neutrons (neutral).
- Because atoms contain equal numbers of protons and electrons, they are electrically neutral as a whole. The nucleus (protons + neutrons) contains most of an atom's mass while electrons occupy the surrounding space in shells labelled K, L, M, N, ...
- The maximum number of electrons that can occupy a shell with principal quantum number n is given by:
\( 2n^{2} \)
- Hence the maximum electrons per shell are:
- K-shell (n = 1): 2 electrons;
- L-shell (n = 2): 8 electrons;
- M-shell (n = 3): 18 electrons;
- N-shell (n = 4): 32 electrons, and so on.
- Electrons fill inner shells before occupying outer shells. The outermost shell (valence shell) often governs chemical behaviour.
Valency
- Valence electrons are the electrons in the outermost shell of an atom; they participate in chemical bonding.
- Elements with the same number of valence electrons display similar chemical properties. The number of valence electrons is related to the element's position in the Periodic Table (period and group).
- Valency (combining capacity) may be determined by the number of electrons an atom gains, loses or shares to attain a stable electron configuration (often achieving an octet). For a fully filled outer shell, valency is effectively zero (as for noble gases).
- Example: carbon (atomic number 6) has four valence electrons and typically shows valency 4.
Atomic Number
- The atomic number (symbol Z) is the number of protons in an atom's nucleus. In a neutral atom the atomic number also equals the number of electrons.
Mass Number
- The mass number(A) of an atom is the total number of protons and neutrons in its nucleus.
Mass number = number of protons + number of neutrons
- Isotopes are atoms of the same element (same Z) with different mass numbers (different numbers of neutrons). Isotopes have identical chemical properties but slightly different physical properties (density, melting point, boiling point).
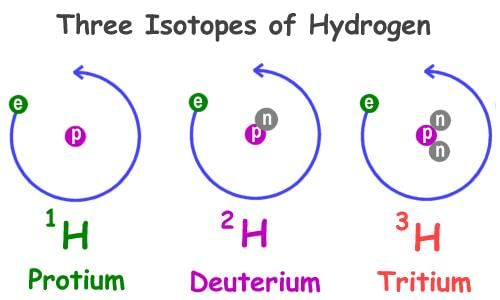
- Examples for hydrogen isotopes are: protium (¹H), deuterium (²H or D) and tritium (³H or T).
- Isobars are atoms of different elements that have the same mass number but different atomic numbers.
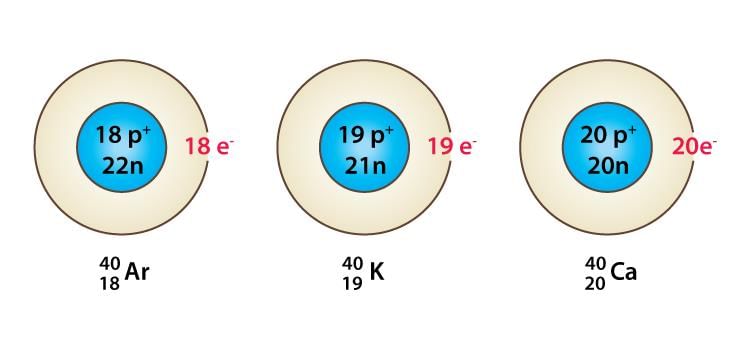
Isobars
- Isotones are atoms of different elements that have the same number of neutrons but different atomic numbers.
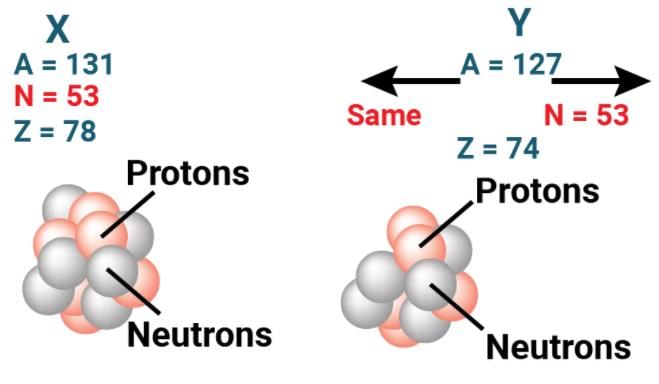
Isotones
Radioactivity
- Radioactivity is a nuclear phenomenon: the spontaneous emission of radiation from an unstable atomic nucleus.
- Ernest Rutherford studied radioactivity in detail. He found that radiation emitted from radioactive substances splits into three distinct types under magnetic fields.
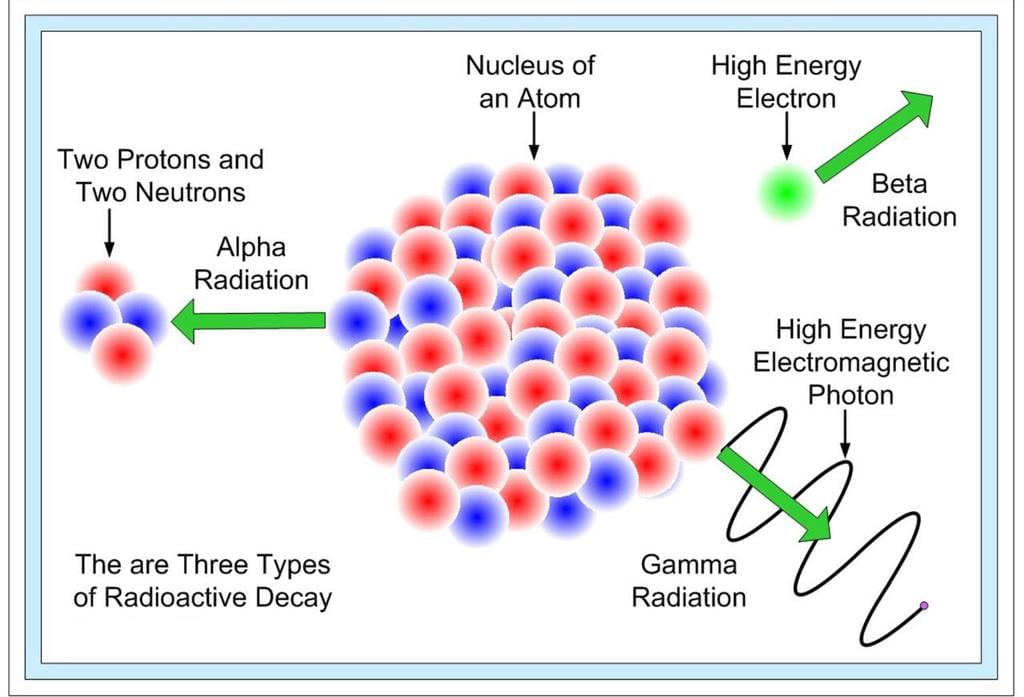
Three types of Radioactive Decay
- These three types of radiation are:
- Alpha (α) rays: positively charged particles; deflected in a direction opposite to β-rays in a magnetic field.
- Beta (β) rays: negatively charged particles; deflected in a direction opposite to α-rays.
- Gamma (γ) rays: neutral, highly penetrating electromagnetic radiation; not deflected by magnetic fields.
Final Note: The above sections present foundational concepts in acids, bases and salts and the structure of the atom. These topics form the basis for understanding chemical reactions, bonding, periodic properties and nuclear phenomena encountered in further chemistry studies.
The document NCERT Summary: Summary of Chemistry- 1 is a part of the UPSC Course Science & Technology for UPSC CSE.
All you need of UPSC at this link: UPSC
FAQs on NCERT Summary: Summary of Chemistry- 1
| 1. What are acids and bases? |  |
Ans. Acids are substances that release hydrogen ions (H+) when dissolved in water, while bases are substances that release hydroxide ions (OH-) when dissolved in water. Acids typically have a sour taste and can corrode metals, while bases have a bitter taste and feel slippery.
| 2. How do acids and bases neutralize each other? |  |
Ans. When an acid reacts with a base, they undergo a neutralization reaction where the hydrogen ions (H+) from the acid combine with the hydroxide ions (OH-) from the base to form water (H2O). This reaction results in the formation of a salt and water.
| 3. What are salts? |  |
Ans. Salts are ionic compounds composed of a metal cation and a nonmetal anion. They are formed when an acid reacts with a base through a neutralization reaction. Salts are usually solid crystals and can be classified as acidic, basic, or neutral depending on the nature of the cation and anion.
| 4. How can we differentiate between acids and bases? |  |
Ans. There are several ways to differentiate between acids and bases. One common method is by using indicators, which are substances that change color depending on the pH of the solution. For example, litmus paper turns red in the presence of an acid and blue in the presence of a base. Additionally, acids can conduct electricity, taste sour, and react with metals, while bases can conduct electricity, taste bitter, and feel slippery.
| 5. What are some common examples of acids, bases, and salts? |  |
Ans. Some common examples of acids include hydrochloric acid (HCl), sulfuric acid (H2SO4), and acetic acid (CH3COOH). Examples of bases include sodium hydroxide (NaOH), potassium hydroxide (KOH), and calcium hydroxide (Ca(OH)2). Salts like sodium chloride (NaCl), calcium carbonate (CaCO3), and potassium nitrate (KNO3) are commonly found in everyday life.
Related Searches
NCERT Summary: Summary of Chemistry- 1, video lectures, NCERT Summary: Summary of Chemistry- 1, pdf , Free, Exam, Semester Notes, NCERT Summary: Summary of Chemistry- 1, study material, Previous Year Questions with Solutions, Extra Questions, past year papers, Viva Questions, Sample Paper, practice quizzes, MCQs, Important questions, Summary, mock tests for examination, Objective type Questions, ppt, shortcuts and tricks;






