Class 10 Science: CBSE Sample Question Paper (2020-21) - 1
Class-X
Science Theory
TIME: 3 Hrs.
M.M: 80
General Instructions:
Read the following instructions very carefully and strictly follow them:
1. The question paper comprises four sections A, B, C and D. There are 36 questions in the question paper.
All questions are compulsory.
2. Section-A - Question no. 1 to 20 - all questions and parts thereof are of one mark each. These questions contain multiple-choice questions (MCQs), very short answer questions and assertion - reason type questions. Answers to these should be given in one word or one sentence.
3. Section-B - Question no. 21 to 26 are short answer type questions, carrying 2 marks each. Answers to these questions should be in the range of 30 to 50 words.
4. Section-C - Question no. 27 to 33 are short answer type questions, carrying 3 marks each. Answers to these questions should be in the range of 50 to 80 words.
5. Section-D - Question no. - 34 to 36 are long answer type questions carrying 5 marks each. Answers to these questions should be in the range of 80 to 120 words.
6. There is no overall choice. However, internal choices have been provided in some questions. A student has to attempt only one of the alternatives in such questions.
7. Wherever necessary, neat and properly labelled diagrams should be drawn.
Section A
Q.1: Give a reason why do chips manufacturers usually flush bags of chips with a gas such as nitrogen? (1 Mark)
Ans: Nitrogen displaces oxygen inside the packet, preventing oxidation of fats and oils in the chips which would make them rancid. This preserves flavour and extends shelf life.
Q.2: State two physical properties of gold which are of extreme use to jewellers. (1 Mark)
Ans: Malleability and ductility. Malleability allows gold to be beaten into thin sheets for jewellery, and ductility lets it be drawn into fine wires for intricate designs.
Q.3: Name a cyclic unsaturated carbon compound. (1 Mark)
Ans: Benzene.
Q.4: List any two observations when Ferrous Sulphate is heated in a dry test tube? (1 Mark)
Ans:
- The initial light green colour of ferrous sulphate turns to reddish-brown, indicating formation of ferric oxide or basic iron oxide.
- A colourless gas with a choking smell is evolved (sulphur dioxide), showing decomposition.
OR
Identify the products formed when 1 mL of dil. Hydrochloric acid is added to 1g of Sodium metal?
Ans: Sodium chloride and hydrogen gas. The reaction is vigorous and can be written as: 2Na + 2HCl → 2NaCl + H2.
Q.5: Which of the following is not observed in a homologous series? Give the reason for your choice. (1 Mark)
(a) Change in chemical properties
(b) Difference in -CH2 and 14u molecular mass
(c) Gradation in physical properties
(d) Same functional group
Ans: (a)
Explanation: A homologous series has members with the same functional group, so their chemical properties are similar rather than changing from member to member. What changes regularly are physical properties and molecular mass by a CH2 (14 u) unit.
Q.6: In the arrangement shown in figure, there are two coils wound on a non-conducting cylindrical rod. Initially, the key is not inserted into the circuit. Later the key is inserted and then removed shortly after. (1 Mark)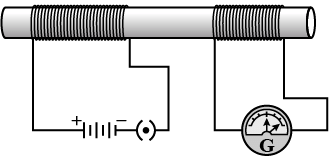
What are the two observations that can be noted from the galvanometer reading?
Ans: On inserting the key, the galvanometer shows a brief deflection in one direction; on removing the key, it shows a brief deflection in the opposite direction. These deflections are momentary because they occur only when the magnetic field through the second coil is changing.
Q.7: Draw the magnetic field lines around a straight current-carrying conductor. (1 Mark)
Ans: The magnetic field consists of concentric circles centred on the wire. The direction of the field is given by the right-hand thumb rule: if the thumb points along the current, the curled fingers show the field direction.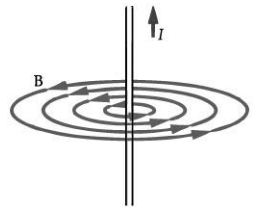
Q.8: Directions: For question number 8, two statements are given- one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below? (1 Mark)
Assertion (A): Sodium metal is stored under Kerosene.
Reason (R): Metallic sodium melts when exposed to air.
(a) Both A and R are true and R is the correct explanation of the assertion.
(b) Both A and R are true but R is not the correct explanation of the assertion.
(c) A is true but R is false.
(d) A is false but R is true
Ans: (c)
Explanation:
(i) Assertion: Sodium metal is stored under kerosene to prevent contact with air and moisture.
(ii) Reason: Metallic sodium does not simply melt on exposure to air; this statement is false.
(iii) Justification: Sodium reacts vigorously with oxygen and moisture in air to form sodium hydroxide and other products; storing it under kerosene prevents these reactions and the associated hazards.
Q.11: Directions: Q. No 11 contain five sub-parts each. You are expected to answer any four subparts in this question. (1 Mark) (ii) The difference between two consecutive members in a homologous series in alkanes in terms of molecular mass and number of atoms of elements are: (iii) The compound CH3CH(OH)CH3 belongs to which of the following homologous series? (iv) Which of the following is not the property of a homologous series? (v) Which of the following represent the name and formula of the 2nd member of a homologous series having general formula CnH2n + 2? OR Which of the following represents voltage? (a) Q.21. Tooth enamel is one of the hardest substances in our body. How does it undergo damage due to eating chocolates and sweets? What should we do to prevent it? (2 Mark) OR Q.26. "The number of trophic levels in a food chain is limited." Give a reason to justify this statement. (2 Mark) OR We do not clean natural ponds or lakes but an aquarium needs to be cleaned regularly. Why is it so? Q.27. Identify the acid and the base from which sodium chloride is obtained. Which type of salt is it? When is it called rock salt? How is rock salt formed? (3 Mark) OR (a) For the preparation of cakes, baking powder is used. If at home your mother uses baking soda instead of baking powder, how will it affect the taste of the cake and why? Q.30. (a) Budding, fragmentation and regeneration, all are considered asexual mode of reproduction. Why? (3 Mark) OR Mention the total number of chromosomes along with the sex chromosomes that are present in a human female and a human male? Explain how in sexually producing organisms the number of chromosomes in the progeny remains the same as that of the parents? Q.31. In a pea plant, the trait of flowers bearing purple colour (PP) is dominant over white colour (pp). Explain the inheritance pattern of F1 and F2 generations with the help of a cross following the rules of inheritance of traits. State the visible characters of F1 and F2 progenies? (3 Mark) Q.34. Carbon cannot reduce the oxides of sodium, magnesium and aluminium to their respective metals. Why? Where are these metals placed in the reactivity series? How are these metals obtained from their ores? Take an example to explain the process of extraction along with chemical equations. (5 Mark) Q.35. The position of certain elements in the Modern Periodic Table is shown below. Using the above table answer the following questions giving reasons in each case: (5 Mark) OR Define atomic size. Give its unit of measurement. In the modern periodic table what trend is observed in the atomic radius in a group and a period and why is it so? OR (a) Name the organs that form the excretory system in human beings.
Q.9: Directions: For question number 9, two statements are given- one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below? (1 Mark)
Assertion (A): Gas bubbles are observed when sodium carbonate is added to dilute hydrochloric acid.
Reason (R): Carbon dioxide is given off in the reaction.
(a) Both A and R are true and R is the correct explanation of the assertion.
(b) Both A and R are true but R is not the correct explanation of the assertion.
(c) A is true but R is false.
(d) A is false but R is true
Ans: (a)
Explanation:
(i) Assertion: True - bubbles are observed on adding sodium carbonate to dilute HCl.
(ii) Reason: True - carbon dioxide gas is produced in the reaction.
(iii) Justification: The reaction is Na2CO3 + 2HCl → 2NaCl + H2O + CO2, and the CO2 appears as bubbles.
Q.10: Directions: For question number 10, two statements are given- one labelled Assertion (A) and the other labelled Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below? (1 Mark)
Assertion (A): In the case of a rainbow, the light at the inner surface of the water drop gets internally reflected.
Reason (R): The angle between the refracted ray and normal to the drop surface is greater than the critical angle.
(a) Both A and R are true and R is the correct explanation of the assertion.
(b) Both A and R are true but R is not the correct explanation of the assertion.
(c) A is true but R is false.
(d) A is false but R is true.
Ans: (a)
Explanation:
(i) Assertion: True - rays inside the drop undergo internal reflection at the inner surface contributing to the rainbow.
(ii) Reason: True - for internal reflection to occur the angle of incidence inside the drop must exceed the critical angle for water-air interface.
(iii) Justification: Refraction at the first surface disperses white light; some coloured rays then meet the inner surface at angles greater than the critical angle and are internally reflected back, producing the visible rainbow.
Read the passage and answer any four questions.
A homologous series is a series of organic compounds which belong to the same family i.e. possess the same functional group) and show similar chemical properties. The members of this series are called homologous and differ from each other by the number of CH2 units in the main carbon chain.
(i) The chemical properties of which of the following compounds is similar to butane?
(a) Butyne
(b) Propene
(c) Propyne
(d) Pentane
Ans: (d)
Explanation: Pentane and butane are both alkanes (same functional group - single C-C bonds). Members of the same homologous series (alkanes) show similar chemical behaviour, so pentane is chemically similar to butane.
(a) 14 a.m.u and CH2 respectively
(b) 12 a.m.u and CH2 respectively
(c) 14 a.m.u and CH3 respectively
(d) 12 a.m.u and CH3 respectively
Ans: (a)
Explanation:
The -CH2 group has mass 12 (C) + 2 × 1 (H) = 14 a.m.u. Hence consecutive alkanes differ by one -CH2 unit and 14 a.m.u.
(a) Aldehydes
(b) Alcohols
(c) Ketones
(d) Carboxylic acids
Ans: (b)
Explanation: The compound contains an -OH (hydroxyl) group, so it belongs to the alcohol series.
(a) They show similar chemical properties.
(b) They differ by 14 units by mass.
(c) They all contain a double bond
(d) They can be represented by a general formula
Ans: (c)
Explanation: Not all homologous series contain a double bond. That is specific to series like alkenes; many homologous series (alkanes, alcohols, etc.) do not have a double bond.
(a) Methane, CH4
(b) Ethane, C2H6
(c) Ethene, C2H4
(d) Ethyne (C2H2)
Ans: (b)
Explanation: The general formula CnH2n+2 refers to alkanes; the first member (n=1) is methane (CH4) and the second (n=2) is ethane (C2H6), option (b).
Q.12. Why is the refractive index of the atmosphere different at different altitudes? (1 Mark)
Ans. The refractive index depends on air density. As altitude increases, air density decreases, so the refractive index of the atmosphere becomes smaller at higher altitudes.
Q.13. What is the magnification of the images formed by the plane mirror and why? (1 Mark)
Ans. Magnification is +1 because a plane mirror forms an image that is the same size as the object and the image is virtual and laterally inverted.
Q.14. The electronic configuration of two elements X and Y are 2, 8, 7 and 2, 8, 8, 3 respectively. Write atomic numbers of X and Y ? (1 Mark)
Ans. Atomic number of X = 2, 8, 7 = 17
Atomic number of Y = 2, 8, 8, 3 = 21
Q.15. What is the maximum resistance which can be made using five resistors each of 1/5 W ? (1 Mark)
(a) 1/5 Ω
(b) 10 Ω
(c) 5 Ω
(d) 1 Ω
Ans. (d)
Explanation.
To obtain the highest resistance, connect the resistors in series. Each resistor = 1/5 Ω, so total R = 5 × 1/5 = 1 Ω. Hence option (d).
Q.16. The proper representation of the series combination of cells obtaining maximum potential is (1 Mark)
(i)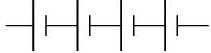
(ii)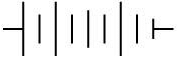
(iii)
(iv) 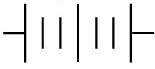
(a) (i)
(b) (ii)
(c) (iii)
(d) (iv)
Ans. (a)
Explanation.
Cells give maximum potential when connected in series with the negative terminal of one connected to the positive terminal of the next. This adds the individual emf values, giving the highest total potential.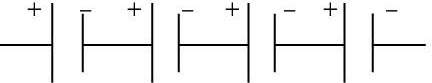

(b) Work done × Charge
(c)
(d)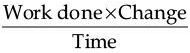
Ans. (a)
Explanation: Potential difference (voltage) = Work done / Charge. Using Charge = Current × Time, Work done = (Current × Time) × Potential difference, so the representation in (a) correctly shows voltage.
Q.17. The change in magnetic field lines in a coil is the cause of induced electric current in it. Name the underlying phenomenon. (1 Mark)
Ans. Electromagnetic induction - an emf (and hence current) is induced in a coil when the magnetic flux through it changes with time.
Q.18. Answer question numbers 18(a) to 18(d) on the basis of your understanding of the following paragraphs and the related studied concepts.
The growing size of the human population is a cause of concern for all people. The rate of birth and death in a given population will determine its size. Reproduction is the process by which organisms increase their population. The process of sexual maturation for reproduction is gradual and takes place while general body growth is still going on. Some degree of sexual maturation does not necessarily mean that the mind or body is ready for sexual acts or for having and bringing up children. Various contraceptive devices are being used by human beings to control the size of the population.
(a) List two common signs of sexual maturation in boys and girls. (1 Mark)
Ans. In boys: broadening of shoulders and appearance of facial hair. In girls: development of breasts (mammary glands) and the start of menstruation.
(b) What is the result of reckless female foeticide? (1 Mark)
Ans. It causes a shortage of females in the population, creating a serious imbalance in the male-to-female ratio and leading to social problems.
(c) Which contraceptive method changes the hormonal balance of the body? (1 Mark)
Ans. Chemical method of contraception such as oral contraceptive pills (hormonal pills).
(d) Write two factors that determine the size of a population. (1 Mark)
Ans. Birth rate and death rate.
Q.19. Veins are thin-walled and have valves. Justify. (1 Mark)
Ans. Veins carry blood at low pressure, so their walls are thinner than arteries. Valves in veins prevent backflow and ensure blood moves in one direction towards the heart, especially against gravity in the limbs.
Q.20. State the role of the pancreas in the digestion of food. (1 Mark)
Ans. The pancreas secretes pancreatic juice containing enzymes such as amylase (for starch), trypsin (for proteins) and lipase (for fats), and also bicarbonate ions to neutralise acidic chyme entering the small intestine.Section B
Ans. When sugars from chocolates and sweets remain in the mouth, oral bacteria ferment them to produce acids. If the mouth pH falls below about 5.5, these acids dissolve (demineralise) enamel and cause decay. To prevent this: brush teeth regularly with fluoride toothpaste, reduce sugary snacks between meals, rinse the mouth with water after sweets, and visit the dentist for regular check-ups.
Q.22. State the meaning of strong acids and weak acids. Give one example of each. (2 Mark)
Ans. Strong acid: An acid that ionises (dissociates) almost completely in water to give many H+ ions. Example: Hydrochloric acid (HCl).
Weak acid: An acid that ionises only partially in water, producing relatively few H+ ions. Example: Ethanoic acid (CH3COOH).
Q.23. Give the respective scientific terms used for studying :
(i) the mechanism by which variations are created and inherited and,
(ii) the development of a new type of organisms from the existing ones. (2 Mark)
Ans. (i) Genetics - the study of how traits and variations are inherited.
(ii) Evolution - the study of how new types of organisms develop from existing ones over time.
Q.24. On entering in a medium from the air, the speed of light becomes half of its value in air. Find the refractive index of that medium with respect to air. (2 Mark)
Ans. Let vair be the speed of light in air and vmedium be the speed in the medium. Given vmedium = (1/2) vair.
Refractive index µ (with respect to air) = vair / vmedium = vair / (vair / 2) = 2.
Thus the refractive index of the medium with respect to air is 2.
Q.25. List four methods of contraception used by humans? (2 Mark)
Ans. Four commonly used methods are:
(i) Mechanical barrier methods (e.g., condoms).
(ii) Chemical methods (e.g., spermicidal creams or foams).
(iii) Hormonal methods (e.g., oral contraceptive pills).
(iv) Surgical methods (e.g., tubectomy or vasectomy for permanent sterilisation).
Justify the following statement: "The use of contraceptive methods has a direct effect on the health and prosperity of a family."
Ans.
Ans.
Ans. We need to clean the aquarium because of the :
(i) absence of sufficient natural decomposers and ecological cycles in a closed artificial tank.
(ii) stagnancy of water and accumulation of waste, which would otherwise be broken down in a natural ecosystem.Section C
Ans. Acid - Hydrochloric acid (HCl).
Base - Sodium hydroxide (NaOH).
Type of salt - Neutral salt (formed by neutralisation of an acid by a base).
It is called rock salt (halite) when it occurs as large crystalline deposits often mixed with impurities and forming brownish crystals.
Rock salt is formed by the evaporation and drying up of seas or saline lakes, leaving behind crystalline deposits of sodium chloride.
(b) How is baking soda converted into baking powder?
(c) What makes the cake soft and spongy?
Ans.
(a) If baking soda (sodium bicarbonate) is used without an acid present, it can decompose on heating to form sodium carbonate (Na2CO3), giving a bitter taste to the cake.
(b) Baking powder is made by mixing baking soda with a mild edible acid (e.g., tartaric acid) and a drying agent; on heating the acid and base react to release CO2.
(c) The CO2 gas liberated during baking forms bubbles in the batter, making the cake soft and spongy.
Q.28. (i) Name the element with atomic number 17? (3 Mark)
(ii) To which period does it belong?
(iii)To which group does it belong?
(iv) Write its electronic configuration.
Ans. (i) Chlorine.
(ii) 3rd period.
(iii) 17th group (halogens).
(iv) 2, 8, 7.
Q.29. Three resistors of 5 Ω, 10 Ω and 15 Ω are connected in series and the combination is connected to the battery of 30 V. Ammeter and voltmeter are connected in the circuit. Draw a circuit diagram to connect all the devices in the proper correct order. What are the current flowing and potential difference across 10 Ω resistance? (3 Mark)
Ans.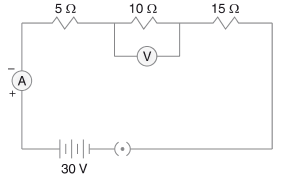
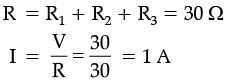
Solution:
Total resistance in series = 5 + 10 + 15 = 30 Ω.
Current I = V / R = 30 V / 30 Ω = 1 A.
Potential difference across 10 Ω = I × R = 1 A × 10 Ω = 10 V.
Therefore: Current through the circuit = 1 A; Voltage across 10 Ω resistor = 10 V.
(b) With the help of neat diagrams, explain the process of regeneration in Planaria?
Ans. (a) These are asexual because they involve only one parent, do not require fusion of gametes, and produce offspring genetically identical to the parent through mitotic cell divisions.
(b) In Planaria regeneration, if the body is cut into pieces, each piece can regrow the missing parts and develop into a complete worm. Specialised cells divide and differentiate to replace lost structures, so one original organism can give rise to several complete organisms by regeneration.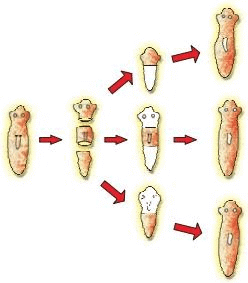
Ans. Chromosome numbers:
Human somatic cells have 46 chromosomes in total (23 pairs). Of these, 22 pairs are autosomes and 1 pair is the sex chromosomes.
In a human male: 22 pairs of autosomes + XY sex chromosomes (total 46).
In a human female: 22 pairs of autosomes + XX sex chromosomes (total 46).
How the number is maintained:
During gamete formation (meiosis), the chromosome number is halved so gametes are haploid (23 chromosomes). When a male gamete (sperm) and a female gamete (egg) fuse at fertilisation, the diploid number (46) is restored in the zygote. This keeps the chromosome number constant across generations.
Detailed Answer
Ans. Cross: Pure-breeding purple (PP) × pure-breeding white (pp):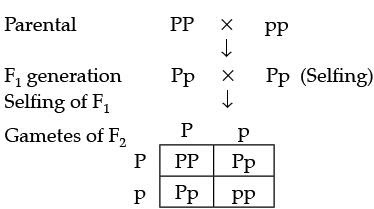
F1 generation: All offspring are Pp (heterozygous) and show the dominant purple colour - so all flowers are purple (phenotype 100% purple).
F2 generation: When F1 individuals (Pp) are self-pollinated, the genotypic ratio obtained is 1 PP : 2 Pp : 1 pp, giving a phenotypic ratio of 3 purple : 1 white. Thus three plants show purple flowers and one shows white.
Q.32. The resistance of a wire of 0.01 cm radius is 10 Ω. If the resistivity of the material of the wire is 50 × 10-8 W.m, find the length of the wire. (3 Mark)
Ans. Given: radius r = 0.01 cm = 0.01 × 10-2 m = 1.0 × 10-4 m.
Resistivity ρ = 50 × 10-8 Ω·m = 5.0 × 10-7 Ω·m.
Resistance R = 10 Ω.
Resistance formula: R = ρL / A, where A = πr2.
So L = R × A / ρ = R × π r2 / ρ.
Substitute values: L = 10 × π × (1.0 × 10-4)2 / (5.0 × 10-7) = 10 × π × 1.0 × 10-8 / 5.0 × 10-7.
Compute: (1.0 × 10-8) / (5.0 × 10-7) = 1/50 = 0.02.
Thus L = 10 × π × 0.02 = 0.2 π ≈ 0.628 m.
Therefore, the length of the wire is approximately 0.628 m.
Q.33. What is a solenoid? Draw the field lines of the magnetic field produced on passing a current through and around a current-carrying solenoid. (3 Mark)
Ans. Definition: A solenoid is a long coil of insulated conducting wire with many turns wound closely together in the shape of a cylinder.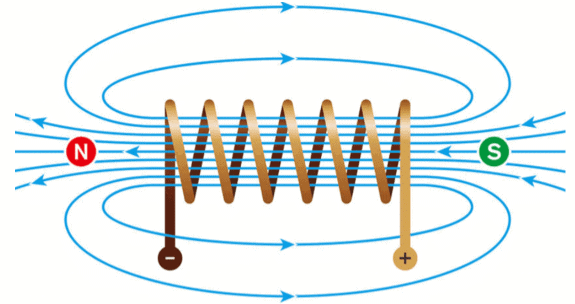
Section D
Ans.
For example, extraction of aluminium from its ore (bauxite) involves purification to obtain alumina (Al2O3) followed by electrolysis of molten alumina (Hall-Héroult process):
At cathode: Al3+ + 3e- → Al (aluminium metal)
At anode: 2O2- → O2 + 4e-
This electrolytic process yields pure aluminium metal and oxygen gas.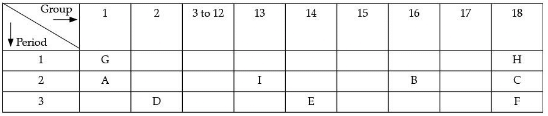
(i) Which element will form only covalent compounds?
(ii) Which element is a non-metal with valency 2?
(iii) Which element is a metal with valency 2?
(iv) Out of H, C and F which has the largest atomic size.
(v) To which family does H, C and F belong.
Ans. (i) Element E is silicon. It will form covalent bonds because it has four electrons in its outermost shell and typically shares electrons to complete its octet.
(ii) Non-metal with valency 2 is B, which is oxygen.
(iii) Element D is a metal with valency 2 (magnesium). Its low electronegativity makes it likely to lose two electrons to form Mg2+.
(iv) Element F has the largest atomic size. Element F is argon; it occupies more principal energy levels than H and C, so its atomic radius is the largest among the three.
(v) Elements H, C and F belong to Group number 18 in the given table (noble gases) as shown; these elements have their outer shells complete and are chemically stable, placing them in the noble gas family according to the table provided.
Ans. Atomic size (atomic radius) is the distance from the centre of the nucleus to the outermost electron shell of an atom.
Unit: Angstrom (Å), where 1 Å = 10-10 m.
Trend across a period (left to right): Atomic radius decreases because nuclear charge increases and pulls electrons closer, reducing size.
Trend down a group: Atomic radius increases because electrons occupy higher principal energy levels (more shells), making atoms larger despite increased nuclear charge.
Reasons:
Across a period, increased effective nuclear charge draws electrons inward. Down a group, added shells outweigh increased nuclear attraction, increasing size.
Q.36. (a) Why is there a difference in the rate of breathing between aquatic organisms and terrestrial organisms? Explain.
(b) Draw a diagram of the human respiratory system and label - pharynx, trachea, lungs, diaphragm and alveolar sac on it. (5 Mark)
Ans. (a) Terrestrial organisms breathe air, which contains a high concentration of dissolved oxygen and is easy to obtain. Aquatic organisms rely on the small amount of oxygen dissolved in water, which is much less available than in air. Therefore aquatic organisms often have a higher rate of breathing or specialised structures (gills) to extract enough oxygen from water.
(b) Diagram of the human respiratory system :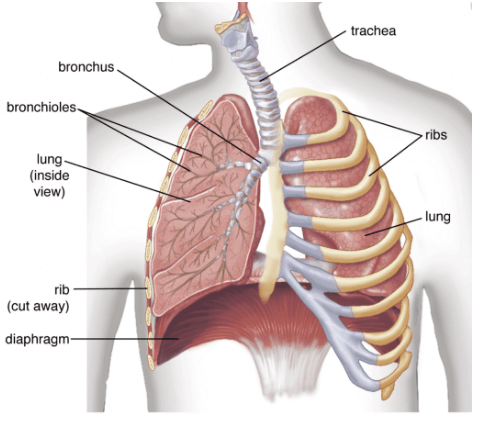
(b) Describe in brief how urine is produced in the human body.
Ans. (a) The human excretory system comprises a pair of kidneys, a pair of ureters, a urinary bladder and a urethra.
(b) Urine formation involves three main steps:
(i) Glomerular filtration: Blood is filtered in the glomerulus; water, nitrogenous wastes (like urea), glucose, amino acids and salts enter Bowman's capsule as filtrate.
(ii) Tubular reabsorption: Useful substances (glucose, certain ions, most of the water) are reabsorbed from the tubule back into the blood via surrounding capillaries.
(iii) Secretion: Additional wastes and excess ions are secreted into the tubule; the remaining fluid becomes urine, which is collected into collecting ducts, passes into the ureter and is stored in the urinary bladder before being expelled through the urethra.
FAQs on Class 10 Science: CBSE Sample Question Paper (2020-21) - 1
| 1. What is the duration of the Class X Science Theory exam? |  |
| 2. How many marks is the Class X Science Theory exam worth? |  |
| 3. What is the format of the Class X Science Theory exam? |  |
| 4. Is the Class X Science Theory exam conducted by CBSE? |  |
| 5. Are sample question papers available for the Class X Science Theory exam? |  |






