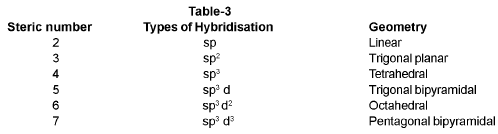
Important Formulas: Chemical Bonding
Chemical Bond : In the process each atom attains a stable outer electronic configuration of inert gases. Ionic or Electrovalent Bond : The formation of an ionic compound depends primarily upon the ease of formation of positive and negative ions from the respective neutral atoms and on the arrangement of these ions in the crystalline lattice of the solid.
Ionic Bonding - Conditions and Basics
- Essential conditions for formation of ionic compounds:
- Large difference in electronegativity between the two combining elements.
- Low ionization enthalpy for the electropositive element (M(g) → M+(g) + e-).
- Highly negative electron gain enthalpy for the electronegative element (X(g) + e- → X-(g)).
- Large (negative) lattice enthalpy for the resulting ionic solid (M+(g) + X-(g) → MX(s)).
Lattice Enthalpy
Definition: The lattice enthalpy of an ionic solid is defined as the energy required to completely separate one mole of a solid ionic compound into its gaseous constituent ions.
Factors affecting lattice energy:
- Lattice energy depends on the interionic distance and the charges on the ions. Qualitatively, lattice energy increases as the interionic distance decreases.
- Lattice energy ∝ Z+·Z-, where Z+ and Z- are the magnitudes of the ionic charges.
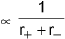
Determination of Lattice Energy - Born-Haber Cycle
Born-Haber cycle: A thermochemical cycle, based on Hess's law, that interrelates the successive enthalpy changes involved in the formation of an ionic compound from its elements in their standard states. It is used to calculate lattice enthalpies using measurable quantities such as ionization energy, electron affinity and enthalpies of atomization and formation.
Hydration and Hydrolysis
Hydration: When ionic salts dissolve in water they produce ions and the resulting solution conducts electricity. Small cations with large charge density, such as Li+, are strongly hydrated; the hydrated radius of Li+ becomes large and its mobility in solution is reduced. Large cations such as Cs+ are less hydrated and therefore often move faster in solution and conduct more readily.
Hydrolysis: Hydrolysis means reaction with water molecules leading to breaking or redistribution of O-H bonds to form H+ and OH- or to other rearrangements. Hydrolysis of covalent halides often occurs by either:
- Coordinate bond formation - typically in halides of atoms having vacant (often d) orbitals.
- Hydrogen-bond formation - for example, in some nitrogen trihalides the hydrogen-bonded pathway assists hydrolysis.
General Properties of Ionic Compounds
- Physical state: At room temperature ionic compounds are usually solids; they conduct electricity when molten or in aqueous solution (where ions are mobile) but not in the solid state (where ions are fixed in a lattice).
- Isomerism and isomorphism: Simple ionic compounds do not generally show isomerism, but many ionic salts show isomorphism - different compounds crystallise in the same structure (e.g., FeSO4·7H2O and MgSO4·7H2O).
- Electrical conductivity: Ionic solids conduct in molten state and in aqueous solution because ions are free to move.
- Solubility: Ionic compounds tend to be soluble in polar solvents such as water which have a high dielectric constant.
Covalent Character in Ionic Compounds - Fajan's Rules
Fajan's observation: Greater polarisation (distortion of the anion electron cloud by the cation) increases covalent character in an ionic compound.

Fajan's rules that govern covalent character:
- Small cation size → large polarising power → increased covalency.
- Large anion size → more easily polarised → increased covalency.
- Greater positive charge on cation → greater polarisation → increased covalency.
- Greater negative charge on anion → greater polarisability → increased covalency.
- Cations with pseudo-noble-gas electron configurations (e.g., that result from d-electron involvement) often polarise anions more and increase covalent character.
Covalent Bonding
Definition: A covalent bond is formed by sharing of valence electrons between atoms to make molecules. Example: formation of Cl2 by sharing one electron from each chlorine atom to form a shared electron pair.
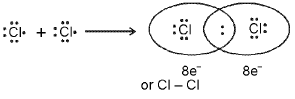
Important conditions for covalent bond formation:
- Each covalent bond results from sharing an electron pair between atoms.
- Each combining atom usually contributes at least one electron to the shared pair.
- As a result of sharing, atoms may achieve noble-gas electronic configurations in their outer shells.
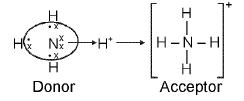
Coordinate (Dative) Bond
Definition: A coordinate bond is a covalent bond in which both electrons of the shared pair are contributed by the same atom, although the shared pair is subsequently shared by both atoms.
- Examples and illustrations:
- Ammonium ion, NH4+, where a lone pair on NH3 is donated to H+.
- Ozone, O3, has resonance structures where coordinate bonding is used to describe electron distribution.
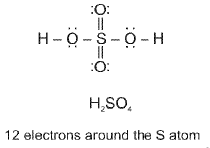
- Other examples include H2SO4, HNO3, H3O+, N2O and complex ions such as [Cu(NH3)4]2+.
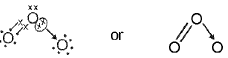
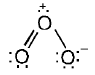
Formal Charge
Formal charge: A bookkeeping tool used to assign charges to atoms in Lewis structures and to help choose the most reasonable resonance structure (the structure with the smallest magnitude of formal charges and with negative charges on the more electronegative atoms is usually preferred).

Limitations of the Octet Rule
- Incomplete octet: Some compounds have central atoms with less than eight electrons, for example LiCl, BeH2, BF3, AlCl3.
- Odd-electron molecules: Molecules with an odd number of electrons such as nitric oxide, NO, and nitrogen dioxide, NO2, cannot be described by simple octet arrangements.
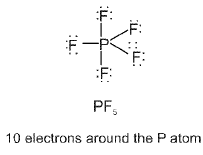
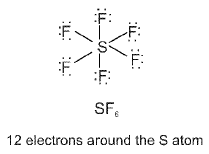
- Expanded octet: Elements of the third period and beyond (for example P, S, Cl) can have more than eight electrons around the central atom (expanded octet) in species such as SF6, PCl5, etc.
- Other drawbacks: Noble gases (e.g., xenon and krypton) form compounds such as XeF2, KrF2, XeOF2, which the octet rule does not anticipate; the octet rule also does not explain molecular shapes or relative stabilities and energies of molecules.
Valence Bond Theory (VBT)
Essence of VBT: According to valence bond theory, a covalent bond forms when half-filled atomic orbitals of two atoms overlap and the electrons with opposite spins pair up in the overlap region. VBT emphasises the importance of orbital overlap and spin pairing.
Orbital Overlap - σ and π Bonds
Orbital overlap concept: A covalent bond results from the overlap of atomic orbitals containing unpaired electrons of opposite spin.
Types of overlap:
- Sigma (σ) bond: Formed by end-to-end (head-on) overlap of orbitals along the internuclear axis. σ bonds can arise from s-s, s-p and p-p end-to-end overlaps.
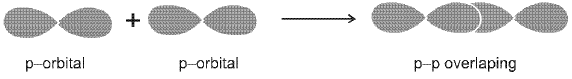
- Pi (π) bond: Formed by side-to-side overlap of parallel p orbitals above and below the internuclear axis; π bonds occur in addition to a σ bond in double and triple bonds.

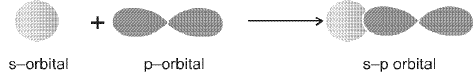
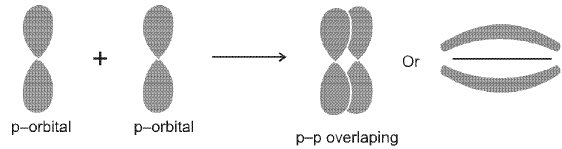
Relative strength: σ bonds usually have greater orbital overlap and are stronger than π bonds, which have less overlap.
Valence Shell Electron Pair Repulsion (VSEPR) Theory
Main postulates:
- The shape of a molecule depends on the number of valence-shell electron pairs (bonded and lone pairs) around the central atom.
- Electron pairs in the valence shell repel each other because their electron clouds are negatively charged.
- Electron pairs arrange themselves in space so as to minimise repulsion and maximise the distance between them.
- The valence shell is treated as a sphere and electron pairs are assumed to localise on the surface at maximum separation.
- Multiple bonds are treated as one electron domain (a double or triple bond acts like a single "super" pair) for determining geometry.
- If resonance structures exist, the VSEPR model may be applied to any representative structure to predict the shape.
Order of repulsion: lone pair-lone pair (lp-lp) > lone pair-bond pair (lp-bp) > bond pair-bond pair (bp-bp).
Hybridisation
Salient features of hybridisation:
- The number of hybrid orbitals equals the number of atomic orbitals combined to form them.
- All hybrid orbitals produced are equivalent in energy and shape.
- Hybrid orbitals usually form stronger and more directional bonds than pure atomic orbitals.
- Hybrid orbitals are oriented in specific directions in space to minimise electron-pair repulsions, so the type of hybridisation helps determine molecular geometry.
Important conditions for hybridisation:
- Only orbitals present in the valence shell participate in hybridisation.
- Orbitals undergoing hybridisation should have comparable energies.
- Promotion of electrons is not a necessary precondition for hybridisation.
- It is orbitals that hybridise, not the electrons themselves.
Determination of hybridisation - Steric number rule (Gillespie):
Steric number of an atom = number of atoms bonded to that atom + number of lone pair(s) on that atom.
Hybridisation Involving d-Orbitals
Type of d-orbitals involved: For some atoms (third period and beyond) d orbitals may participate in hybridisation (for example sp3d, sp3d2), changing geometry and bonding possibilities.
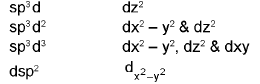
Molecular Orbital Theory (MOT)
Historical note: Developed by F. Hund and R. S. Mulliken in 1932.
Basic principles:
- Molecular orbitals (MOs) are formed by combination of atomic orbitals (AOs) of comparable energy and proper symmetry.
- An electron in an AO is influenced by one nucleus; an electron in an MO is influenced by two or more nuclei - atomic orbitals are monocentric while molecular orbitals are polycentric.
- The number of MOs formed equals the number of combining AOs; when two AOs combine they form one bonding and one antibonding MO.
- MOs are filled according to the Aufbau principle, Pauli exclusion principle and Hund's rule. The exact order of MO energies (especially of σ2p and π2p) is determined experimentally for each molecule.
Conditions for effective combination of AOs:
- Combining AOs should have the same or nearly the same energy.
- Combining AOs must have the same symmetry with respect to the molecular axis.
- Combining AOs should overlap effectively.
Energy Level Diagrams and Bond Order
Energy order for O2 and F2 MOs:
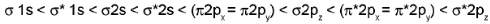
Characteristic feature: For O2 and F2 the σ2pz MO lies higher in energy than the π2px, π2py MOs.
Bond order (b.o.):
b.o. = 1/2 (Nb - Na)
Where Nb is the number of electrons in bonding MOs and Na is the number in antibonding MOs. Positive bond order indicates a stable bond; zero or negative bond order indicates instability.
Relation to bond properties:
- Integral bond order values 1, 2, 3 correspond approximately to single, double and triple bonds respectively.
- Bond length generally decreases as bond order increases.
- If all MOs are doubly occupied, the molecule is diamagnetic; unpaired electrons give paramagnetism (for example O2 is paramagnetic).
Dipole Moment
Definition: Dipole moment μ = q × d, where q is the magnitude of separated charge and d is the distance between charges.
Units: Dipole moment is expressed in Debye (D):
- 1 D = 3.33564 × 10-30 C·m
- 1 Debye = 1 × 10-18 e.s.u.·cm
Example representations and vector addition of bond dipoles determine the molecular dipole. When the vector sum is non-zero the molecule has a net dipole moment.

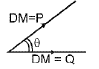
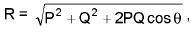
Resonance
Definition: Resonance is the phenomenon in which two or more Lewis structures (which differ only in the positions of electrons, not atoms) can be written for the same compound. The true structure is a resonance hybrid - a weighted average of the contributing structures.
Example: Ozone, O3, is represented by two equivalent resonance structures whose hybrid gives an intermediate bond order and equal O-O bond lengths.
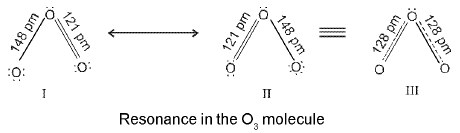
Resonance hybrid: The actual electron distribution obtained by combining all valid resonance structures without violating covalence rules.
Hydrogen Bonding
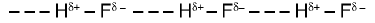
Conditions required for hydrogen bonding:
- Hydrogen must be covalently bonded to a highly electronegative atom (F, O or N).
- The electronegative atom should be small in size (so its lone pairs are localised and available for bonding).
- A lone pair of electrons must be present on the electronegative atom that can interact with H.
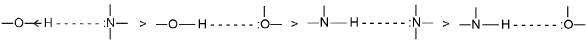
Types of hydrogen bonding:
- Intramolecular hydrogen bonding: Hydrogen bonding occurs within the same molecule when a hydrogen atom lies between two electronegative atoms; commonly leads to formation of a 5- or 6-membered ring. Intramolecular H-bonding often lowers the boiling point compared with the corresponding intermolecularly hydrogen-bonded isomer.
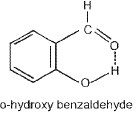
Necessary conditions for intramolecular H-bonding:
- The hydrogen-bonded ring should be planar.
- A 5- or 6-membered ring is preferred (less strain).
- Interacting atoms must be positioned to minimise strain on ring closure.
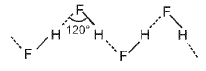
- Intermolecular hydrogen bonding: Hydrogen bonds formed between different molecules of the same or different compounds (e.g., in water and in hydrogen fluoride).
Examples:
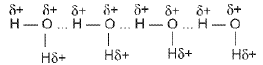
In HF the hydrogen bonds link molecules into chains (HF)n in both solid and liquid states.
Intermolecular Forces (Van der Waals Forces)
Overview: Intermolecular attractions that hold molecules together are weaker than covalent or ionic bonds and include:
- Ion-dipole attractions
- Dipole-dipole attractions
- Ion-induced dipole attractions
- Dipole-induced dipole attractions
- Instantaneous dipole-induced dipole attractions (London dispersion forces)
- Strength of van der Waals forces generally increases with molecular mass and surface area.
- Greater van der Waals forces generally lead to higher boiling points.
Metallic Bond
Models for metallic bonding:
- Electron-sea model: Metal atoms release valence electrons to form a delocalised "sea" of electrons that surrounds positive metal ions; this explains electrical conductivity, malleability and ductility.
- Band model: In solids, atomic orbitals form continuous energy bands (valence and conduction bands); the overlap and partial filling of bands explain metallic electrical conductivity and other properties.
Some Special Bonding Situations
- Electron-deficient bonding: Some compounds contain bonds that have fewer electrons than expected for two-centre two-electron bonds. Examples include three-centre two-electron (3c-2e) bonds in diborane, B2H6, and bridging hydrides in metal clusters and some main-group compounds (e.g., Al2(CH3)6, solid BeH2).
- Back bonding (π back-donation): Occurs when one atom with a lone pair (often a second-period element like O, N or F bound to carbon or boron) donates electron density into an empty orbital (often a p or d orbital) of the bonded atom. Back bonding strengthens bonding and shortens bond length. Example: in BF3 the F lone pairs can donate into empty boron p orbital to some extent.
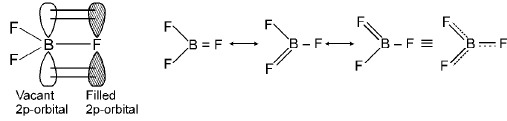
Extent of back bonding in boron trihalides: BF3 > BCl3 > BBr3 (fluorine's strong p→p donation stabilises partial back bonding more effectively).






