Important Formulas: p-Block Elements & Their Compounds
Trends In Properties of p-block Elements.
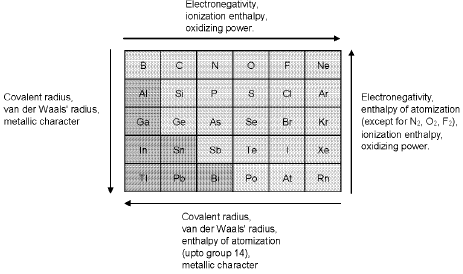
(A) Group 13 Elements : The Boron Family
General characteristics
- Members: Boron (B), aluminium (Al), gallium (Ga), indium (In) and thallium (Tl).
- Valence electronic configuration: ns2 np1.
- Common oxidation state: +3. Heavier elements can also show +1 due to the inert pair effect.
- Trends: metallic character increases down the group; size and metallic behaviour increase from B → Tl.
Oxidation state and trends in chemical reactivity
- General oxidation state is +3. Stability of lower oxidation state (+1) increases down the group because of the inert pair effect.
- Reactivity towards acids and alkalis: trivalent compounds of boron are typically covalent and react differently with acids and bases compared to heavier, more ionic congeners.
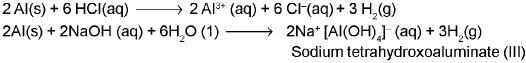
Reactivity towards halogens
The elements of group 13 form trihalides by direct combination with halogens:
2E(s) + 3X2(g) → 2EX3(s) (X = F, Cl, Br, I)
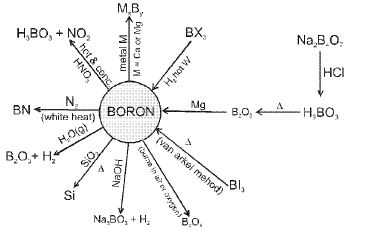
Boron: notable reactions and behaviour
Boron is the only non-metal in the group and shows a chemistry markedly different from the metals.
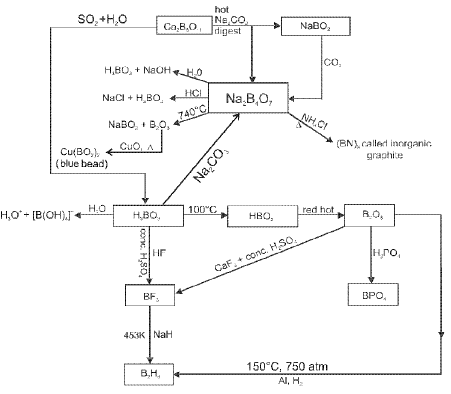
Important reactions and representative transformations involving boron and its hydrides, halides and oxides are frequently studied; these include the chemistry of boranes, boron halides and boron oxides along with Lewis acidity of boron compounds.
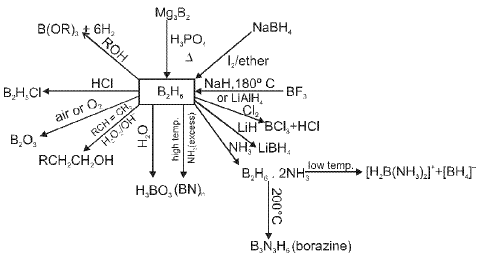
- Small amines such as NH3, CH3NH2 and (CH3)2NH cause unsymmetrical cleavage of diborane (B2H6).
- Large and strongly basic amines such as (CH3)3N and pyridine give symmetrical cleavage of diborane.
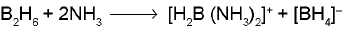
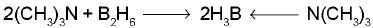
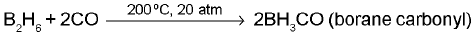
(B) Group 14 Elements : The Carbon Family
General characteristics
- Members: Carbon (C), silicon (Si), germanium (Ge), tin (Sn) and lead (Pb).
- Electronic configuration: ns2 np2.
- Common oxidation states: +4 and +2. Carbon can also show negative oxidation states in certain compounds (carbides, etc.).
- Tendency to exhibit +2 oxidation state increases down the group (Ge < Sn < Pb) due to inert pair effect.
Chemical reactivity
- Reactivity towards oxygen: members form oxides having general formulas MO and MO2 (monoxides and dioxides).
- Reactivity towards water: e.g., tin reacts with steam to form SnO2 and H2.
- Reactivity towards halogens: elements form MX2 and MX4 (X = F, Cl, Br, I); stability of dihalides increases down the group.
Anomalous behaviour of carbon
Catenation is the ability of an element to form chains with atoms of the same element. Carbon shows the highest tendency for catenation: C >> Si > Ge = Sn; Pb shows negligible catenation.
Because of catenation and pπ-pπ multiple bonding, carbon shows a wide variety of allotropes and an extensive organic chemistry.

Allotropes of carbon
Diamond: Each carbon atom is sp3 hybridised and bonded tetrahedrally to four other carbon atoms, forming a rigid three-dimensional network. C-C bond length is about 154 pm. Diamond is the hardest natural substance and is an electrical insulator.
Graphite: Each carbon is sp2 hybridised. Planar hexagonal sheets are stacked with weak van der Waals forces between layers; interlayer distance ≈ 340 pm. In-plane C-C bond length ≈ 141.5 pm. Graphite conducts electricity along the layers and is soft and slippery; used as a dry lubricant.
Fullerenes: Buckminsterfullerene (C60) has a truncated icosahedral (soccer-ball) shape made up of 20 hexagons and 12 pentagons, with 60 carbon atoms at vertices. C-C bond lengths vary (approx. 143.5 pm and 138.3 pm) due to alternating single and double bond character.
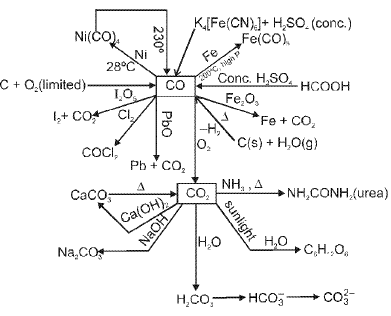
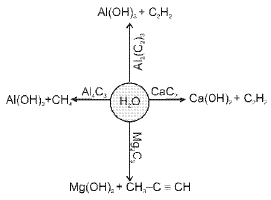
Some important reactions of CO, CO2 and metal carbides
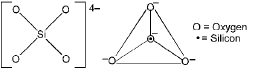
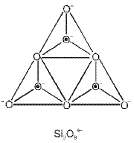
Silicates: classification
- Orthosilicates (isolated tetrahedra): SiO44- units not sharing oxygen atoms.
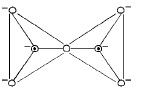
- Pyrosilicates (double tetrahedra): Two SiO4 units sharing one oxygen to give Si2O76-.
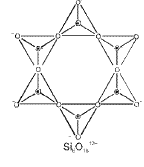
- Cyclic silicates: Tetrahedra linked to form rings.
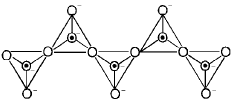
- Chain silicates: Tetrahedra share two oxygens to form single or double chains.
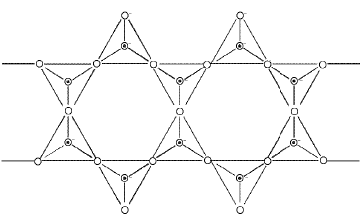
- Two-dimensional sheet silicates: Three oxygens of each SiO4 tetrahedron are shared to give sheets, general formula (Si2O5)n2n-.
- Three-dimensional framework silicates: All four oxygens of each tetrahedron are shared to form a 3D network (e.g., quartz, feldspars).
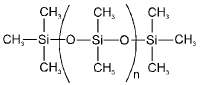
Silicones
- Silicones (polysiloxanes) are polymers containing Si-O-Si backbones with organic groups (R) attached to silicon: -[R2Si-O]-.
- Precursors used for preparing silicones include R3SiCl, R2SiCl2, and RSiCl3.
- Hydrolysis and condensation of these chlorosilanes yield linear, cyclic or cross-linked silicones. The presence of hydrocarbon groups attached to silicon imparts hydrophobicity.
- Silicones can be prepared from R3SiCl, R2SiCl2 and RSiCl3.
- Silicones from the hydrolysis of (CH3)3SiCl:
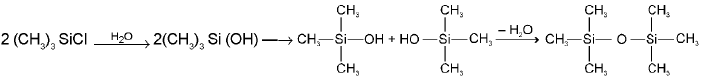
- Silicones from the hydrolysis of a mixture of (CH3)3SiCl and (CH3)2SiCl2:
- Hydrolysis of CH3SiCl3 gives complex cross-linked polymers.
- The hydrocarbon groups attached to silicon make silicones water-repellent and thermally stable.
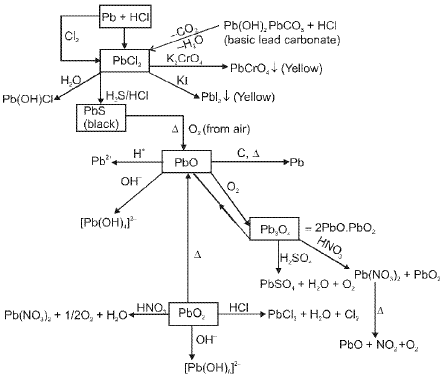
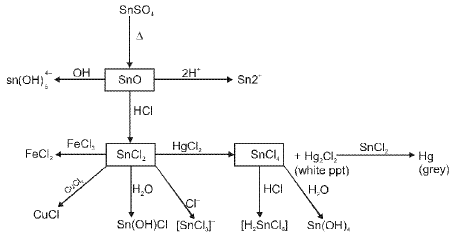
Compounds of lead and tin
Lead and tin show both +2 and +4 oxidation states; lead prefers +2 and tin shows a variable preference depending on the environment. Common compounds include oxides, halides and organometallic derivatives.
(C) Group 15 Elements : The Nitrogen Family
General characteristics
- Members: Nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb) and bismuth (Bi).
- Electronic configuration: ns2 np3.
- Radii: covalent and ionic radii increase down the group.
- Physical properties: nonmetals at the top (N and P), metalloids in the middle (As, Sb) and metal at the bottom (Bi). Metallic character increases down the group.
Chemical properties and oxidation states
- Common oxidation states: -3, +3 and +5. Nitrogen can also show +1, +2 and +4 in oxides and oxyacids.
- Stability of the +5 state decreases down the group whereas stability of the +3 state increases (inert pair effect): Bi3+ > Sb3+ > As3+; Bi5+ < Sb5+ < As5+.
Anomalous behaviour of nitrogen
- Nitrogen is small and very electronegative; it differs from other group 15 elements in several ways.
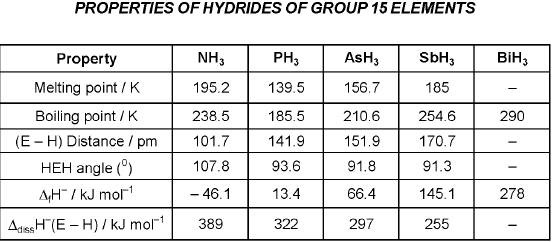
- The stability of hydrides decreases from NH3 to BiH3, and reducing character increases down the group. Basicity decreases in the sequence NH3 > PH3 > AsH3 > SbH3 > BiH3.
- Oxides in higher oxidation states are more acidic than those in lower oxidation states; acidity of oxides decreases down the group. E2O3 type oxides: N and P oxides are acidic, As and Sb oxides are amphoteric, Bi oxide is predominantly basic.
- Nitrogen does not form pentahalides (no available d orbitals). Pentahalides of heavier elements are more covalent than trihalides. Halides hydrolyse in water to give oxyacids or oxyhalides.
- These elements react with metals to form nitrides, phosphides and arsenides exhibiting -3 oxidation state, for example Ca3N2, Ca3P2, Na3As.
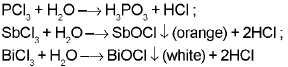
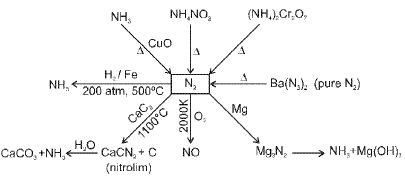
Nitrogen and its compounds
Nitrogen (N2) is a diatomic gas with a very strong N≡N triple bond; its chemistry includes industrial fixation (Haber process), manufacture of ammonia, nitric acid and many nitrogen-containing organic and inorganic compounds.
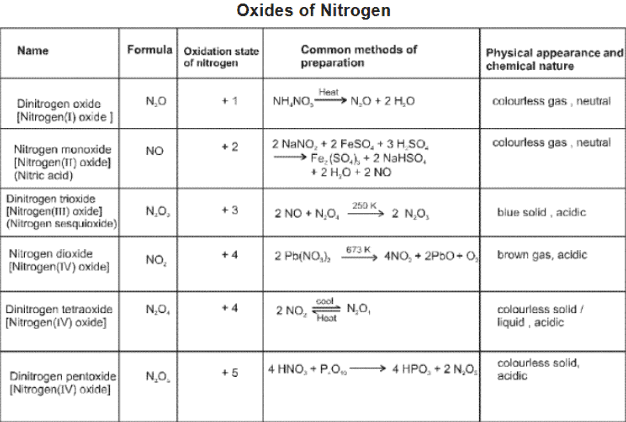
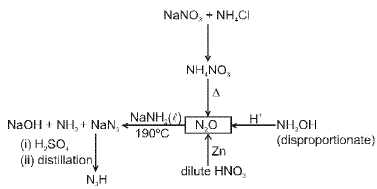
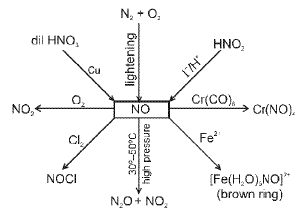
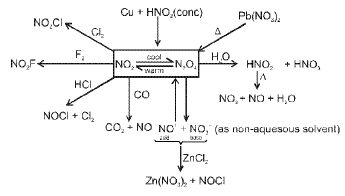
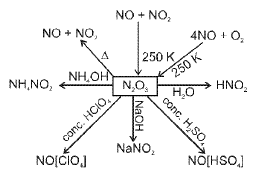
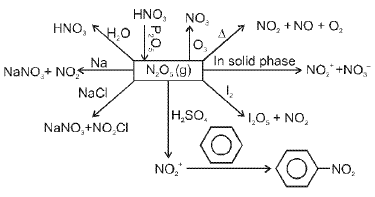
Representative reactions of tin with nitric acid:
(a) With dilute HNO3 (ammonium nitrate present):
4Sn + 10HNO3 → 4Sn(NO3)2 + NH4NO3 + 3H2O
(b) With concentrated HNO3 (oxidation producing NO2):
Sn + 4HNO3 → H2SnO3 + 4NO2 + H2O
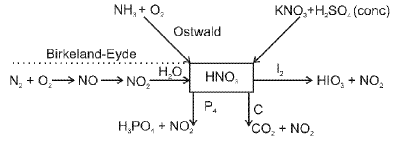
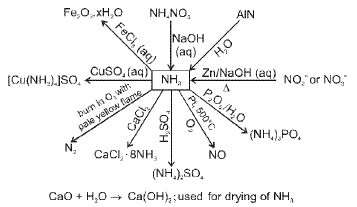
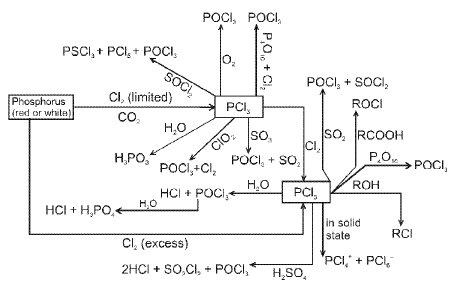
Phosphorus and its compounds
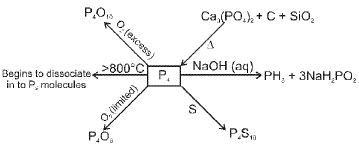
Allotropes of phosphorus include white (yellow) phosphorus, red phosphorus and black phosphorus. Thermal and pressure treatments lead to interconversion:
- White phosphorus heated in CO2 or coal gas at ~573 K gives red phosphorus.
- Heating red phosphorus in a sealed tube at 803 K yields α-black phosphorus.
- Heating white phosphorus at 473 K under high pressure gives β-black phosphorus.
Thermodynamic stability order: black > red > white.
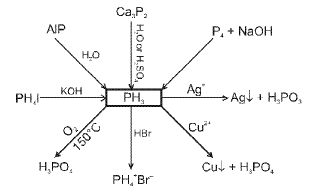
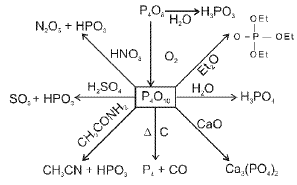
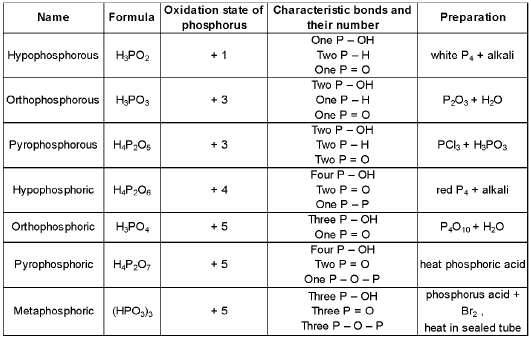
Oxoacids of phosphorus
Oxoacids of phosphorus include phosphorous acid (H3PO3, phosphorus in +3 state) and phosphoric acid (H3PO4, phosphorus in +5 state), together with related polyacids and derivatives.
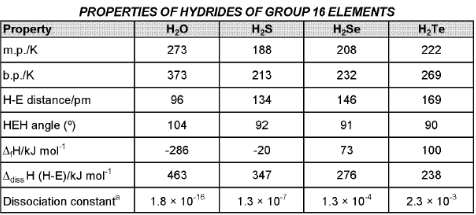
(D) Group 16 Elements : The Oxygen Family
General characteristics
- Members: Oxygen (O), sulphur (S), selenium (Se), tellurium (Te) and polonium (Po).
- Electronic configuration: ns2 np4.
- Atomic and ionic radii increase down the group because of additional shells; oxygen atom is exceptionally small.
- Oxygen and sulphur are nonmetals; selenium and tellurium are metalloids; polonium is metallic and radioactive (Po half-life ≈ 13.8 days for a specific isotope).
- Melting and boiling points increase down the group.
Catenation
Tendency for catenation decreases down the group. Sulphur prominently shows catenation (S8 rings). S-S bonds are biologically significant (e.g., disulfide bonds in cysteine residues in proteins).
Chemical properties and oxidation states
- Typical oxidation states: -2, +2, +4, +6, with -2 common for oxygen in most compounds; +4 and +6 are important for heavier congeners.
- Oxygen is anomalous because of its small size, high electronegativity and absence of d orbitals, limiting its covalency to four.
Anomalous behaviour of oxygen
- Acidic character of hydrides increases down the group: H2O < H2S < H2Se < H2Te. This trend follows decreasing H-E bond dissociation enthalpy down the group.
- Thermal stability of hydrides decreases down the group; H2O is uniquely stable.
- Hydrides except water are reducing; reducing tendency increases from H2S to H2Te.
- Reducing property of dioxide decreases from SO2 to TeO2. SO2 behaves as a reducing agent whereas TeO2 can act as an oxidising agent.
- Stability of halides generally decreases in the order F > Cl > Br > I. Steric and electronic factors make SF6 exceptionally stable.
- Monohalides of S and Se are often dimeric (e.g., S2Cl2, Se2Cl2) and can undergo disproportionation, for example: 2Se2Cl2 → SeCl4 + 3Se.
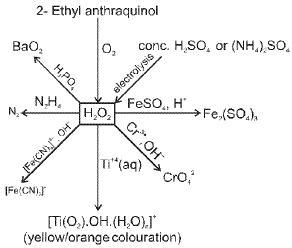
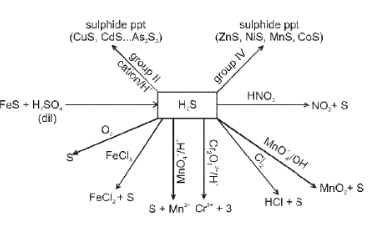
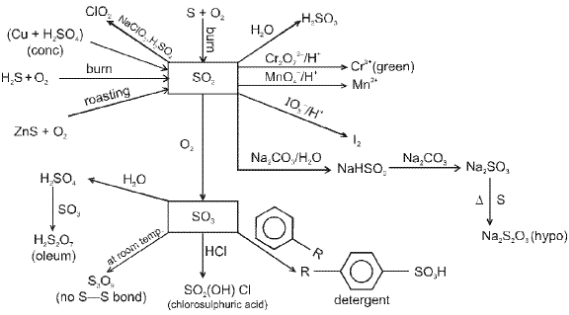
Oxygen (O2) and its compounds
Oxygen exists as a diatomic molecule O2 with a double bond and a ground state that is paramagnetic. Important compounds and reactions include formation of oxides, peroxides (O22-), superoxides (O2-) and oxoacids of heavier elements.

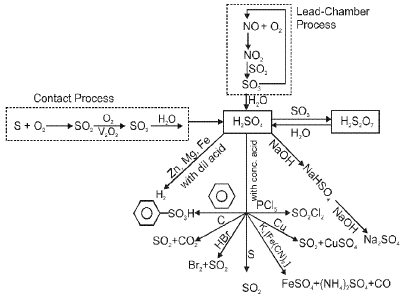
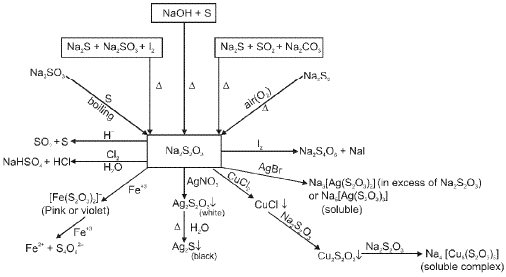
(E) Group 17 Elements : The Halogen Family
General characteristics
- Members: Fluorine (F), chlorine (Cl), bromine (Br), iodine (I) and astatine (At).
- Electronic configuration: ns2 np5.
- Halogens have the smallest atomic radii in their respective periods due to high effective nuclear charge.
- Physical states at room temperature: F and Cl are gases, Br is a liquid, I is a solid.
- Melting and boiling points increase with atomic number.
- Bond dissociation energies vary; from chlorine onwards the general order is Cl-Cl > Br-Br > F-F > I-I (note F-F is weaker due to repulsion between small atoms).
Chemical properties and oxidation states
- Halogens usually exhibit -1 oxidation state. Chlorine, bromine and iodine also show positive oxidation states (+1, +3, +5 and +7) in interhalogen and oxy-halogen compounds.
- They are strong oxidising agents (fluorine being the strongest); reactivity decreases down the group.
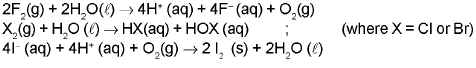

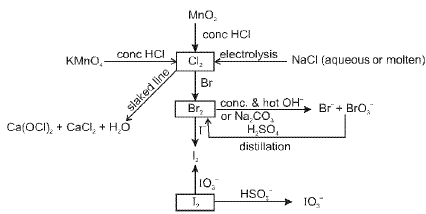
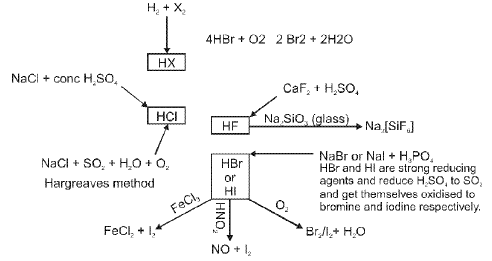
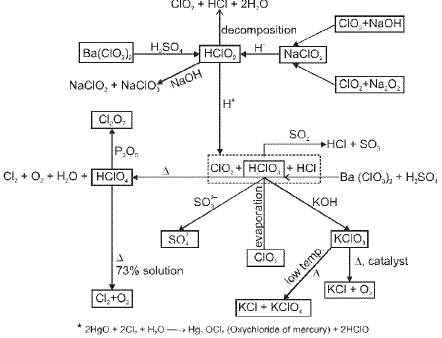
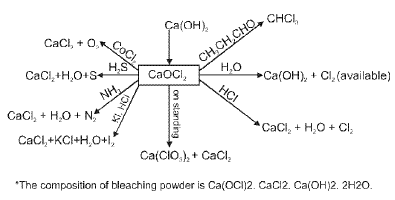
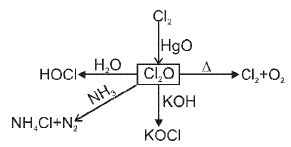
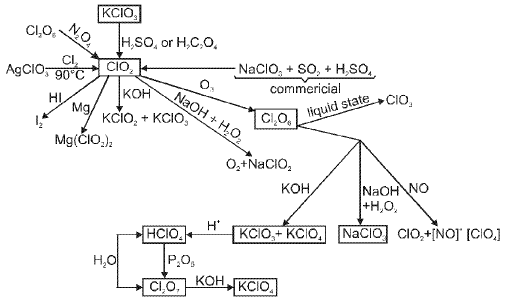
(F) Group 18 Elements : The Noble (Zero) Gas Family
General characteristics
- Members: Helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe) and radon (Rn).
- Electronic configuration (except He): ns2 np6 (He is 1s2).
- Most abundant noble gas in air is argon. Relative abundance in air: Ar > Ne > Kr > He > Xe.
- Atomic radii increase down the group.
- Physical properties: monoatomic, colourless, odourless, very low solubility in water, very low melting and boiling points due to weak dispersion forces.
Chemical properties
- Generally chemically inert because of filled valence shells, high ionisation enthalpies and positive (or less negative) electron gain enthalpies.
- Exceptions: under extreme conditions noble gases can form compounds. In 1962 Neil Bartlett prepared the first noble-gas compound by reacting PtF6 with O2 to obtain O2+PtF6-, then reasoned Xe could be oxidised similarly because the ionisation energy of Xe is similar to that of O2, leading to the isolation of XeF6 and other xenon fluorides and oxides.
Special properties and allotropes of helium
- Liquid helium exists in two forms: He I (normal liquid at 4.2 K) and He II (superfluid below 2.17 K). He II shows superfluidity - zero viscosity, extremely high thermal conductivity and ability to creep through narrow channels.
Clathrate compounds
Clathrates are inclusion compounds in which small atoms or molecules (e.g., Xe, Kr) are trapped in cavities formed by a host lattice (such as ice). Clathrates provide a method for storing noble gases, including radioactive isotopes from reactors.
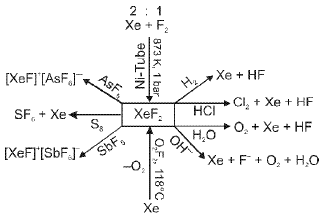
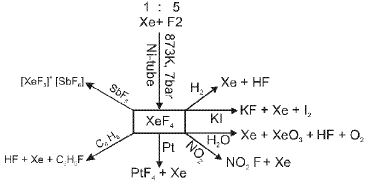
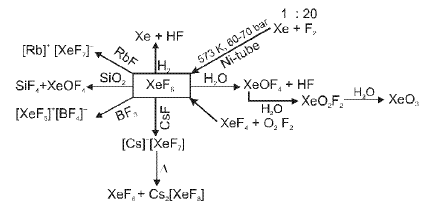
Additional Definitions And Concepts Relevant To P-block
- Inert pair effect: Tendency of the s-electrons of the valence shell to remain non-ionised or unshared in heavier p-block elements, stabilising lower oxidation states (e.g., +2 for Pb, Tl).
- Catenation: Ability of an element to form chains of its own atoms (strongest for carbon).
- Allotropy: Existence of an element in two or more different structural forms (e.g., carbon: diamond, graphite, fullerenes; phosphorus: white, red, black).
- Amphoteric oxides: Oxides that can react both as acids and bases (example: As2O3, Sb2O3).
- Lewis acidity: Many p-block elements (notably B in BF3, BCl3) act as Lewis acids because they can accept an electron pair.
Summary
This chapter summarises the important trends, characteristic reactions and formulas of p-block elements across groups 13-18. Key recurring themes are variations in oxidation states (including the inert pair effect), catenation and allotropy (especially for carbon and phosphorus), the nature of oxides and hydrides, and special reactivities such as the Lewis acidity of boron compounds and the chemistry of noble gas compounds initiated by Bartlett's work. The provided reactions, structural descriptions and illustrative images support understanding of class-12 level, examination-oriented topics in the chemistry of p-block elements.
FAQs on Important Formulas: p-Block Elements & Their Compounds
| 1. What are the key characteristics of Group 13 elements in the Boron family? |  |
| 2. How do the properties of Group 14 elements differ from those of Group 13? |  |
| 3. What are the significant trends observed in the properties of Group 15 elements? |  |
| 4. What defines the properties of Group 16 elements, the Oxygen family? |  |
| 5. What are the notable features of Group 18 elements in the Noble Gas family? |  |






